Research Article
Volume 2 Issue 2 - 2018
Alkaliphilic Bacterial Consortium Used in the Removal of Mixed Reactive Dyes for the Treatment of Textile Effluent
Department of Biotechnology Stella Maris College, Cathedral road, Chennai-86, Tamil Nadu, India
*Corresponding Author: Veena Gayathri Krishnaswamy, Department of Biotechnology Stella Maris College, Cathedral road, Chennai-86, Tamil Nadu, India.
Received: January 28, 2018; Published: March 29, 2018
Abstract
An alkaliphilic bacterial consortium aerobically decolorized the synthetic textile effluent containing the mixed azo reactive dyes (50 mg/L) with 91.8% efficiency at 37°C pH 8. The decolorization of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) were optimized through a broad range of pH (8-10), temperature (28-45°C) and dye concentrations (50-250 mg/L) within 5 days. Bench scale bioreactor studies were performed with synthetic wastewater and their physicochemical parameters were analyzed and ultimately with real time synthetic wastewater. The effect of azodyes and its degraded products were tested against Brassica nigra (Mustard) and Trigonella foenum- graecum (Fenugreek). The results presented in the study demonstrated the application of the alkaliphilic bacterial consortium in the removal of mixed reactive dyes present in the textile effluent which would be difficult to perform by conventional mesophilic bacterial strains.
Keywords: Wastewater; Textile Effluent; Azo Reactive Dyes; Bacterial Consortium; Decolorization; Bioreactor
Introduction
Textile effluent discharged is becoming a major source of environmental pollution in India. Azo dyes are recalcitrant in nature are not readily degraded by the conventional methods because of their structural complexity under alkaline conditions. More than 10-15% dyes do not bind to fiber during color processing and are released into the wastewater causing serious environmental pollution (Rajeswari., et al. 2014). Pollution caused by dye effluents is mainly due to the durability of dyes in wastewater, color fastness, stability, and resistance of dyes to degradation (Madhuri., et al. 2014). Color is an observable pollutant. Color in effluent has now been considered a pollutant that needs to be treated before discharging it. These dyes are resistant to degradation by native microflora, causing their bioaccumulation and biomagnification (Kurade., et al. 2016).
Its removal has been the target of great attention on the last few years, not only because of the toxicity of dyes but also due to its visibility (Karthik., et al. 2014). Synthetic dyes are easy to synthesize, soluble in water, versatile in color and very fast in cloth coloration as compared to the natural dyes, therefore textile industries prefer them over the natural dyes (Ajay Kumar., et al. 2016). Conventional physico-chemical methods such as adsorption with activated carbon, coagulation, precipitation, solvent extraction, membrane filtration, chemical oxidation, ozonation, and flocculation have been used to treat dye containing effluents; however, most of these methods is expensive or require additional chemicals (Deepa., et al. 2013). Decolourization and degradation of the synthetic dye using microbes is an environmental friendly and cost effective substitute, which generate less amount of sludge and the operation are simpler and more attractive to degrade all the dyes to different traditional treatment methods (Wookeun., et al. 2014, Ashok pandey 2007).
Recently Chen., et al. (2010) has described bacterial strains that display good growth of aerobic or agitation culture. Anaerobic reduction implies decolourization of the dyes to usually colorless but potentially harmful aromatic amines. Aromatic amines are generally not further degraded under anaerobic conditions. For several aromatic amines, this can be achieved by biodegradation under aerobic conditions (Sriram., et al. 2013). In some industries pre-treatment was done aerobically before being discharged. But aerobic treatment of textile effluents containing azo dyes by an activated sludge is resistant to biodegradation. Textile effluent has low Biological Oxygen Demand/Chemical Oxygen Demand ratio, hence it is necessary to give a pre-treatment before biological process (Abdul., et al. 2014). Conservative method of using mesophilic microbes to remove the mixed dyes may be difficult to treat the textile effluent, because of its alkaline nature.
Naturally occurring alkaline environments, such as carbonate springs, alkaline soils, and soda lakes, are characterized by their high values (pH 8.0-11.0) due to the presence of high concentration of sodium carbonate salts, which are generally formed by evaporative concentration (Arunachalam., et al. 2015). Extremophiles are a potent source of extremozymes, which show utmost stability under extreme conditions. Scientific interest in extremophilic micro-organisms has recently increased to understand the biochemical mechanisms involved under extreme conditions because of possible biotechnological use of enzymes and molecules from such organisms (Vaishali., et al. 2012).
Alkaliphiles are a group of extremophilic organisms that can survive at pH 9.0 and above, and can be isolated from normal environments. Phytotoxicity is the most studied parameter amongst testing for the toxicity assays. An assessment of the ecological and genetic impact of the environmental pollutants on the plant populations is of great importance as plants are important commercial products and are consumed by humans. Moreover, plants may be used as biosensors of genetic toxicity of the environmental pollutants (Rashid., et al. 2014).
Hence, the present study focuses on the treatment of synthetic textile effluent wastewater by a moderately alkaliphilic bacterial consortium isolated from textile effluent contaminated site. The enriched alkaliphilic consortium was studied for removal of different concentrations of mixed reactive dyes in Synthetic Wastewater (SWW). The growth conditions for the decolourization of mixed reactive dyes by the bacterial consortium were optimized (pH and temperature).The synthetic textile effluent wastewater was studied for removal of mixed dyes by the moderately alkaliphilic bacterial consortium at different dilutions. Their physico-chemical parameters like Chemical Oxygen Demand (COD), Biochemical Oxygen Demand (BOD), Mixed Liquid Suspended Solids (MLSS), Mixed Liquid Volatile Suspended Solids (MLVSS), Phosphate and Nitrogen were studied. The intermediates produced during the treatment were evaluated using FTIR analysis. Phytotoxicity assay of the mixed reactive dyes was tested on the plants Brassicanigra (Mustard) and Trigonellafoenum-graecum (Fenugreek). Further, the synthetic textile effluent was treated with a laboratory scale bioreactor.
Materials and Methods
Dyes and Chemicals
The textile dyes (C.I. RR 195 -Reactive Red 195, C.I. RBL 1 -Reactive Brown 23and C.I. RBR 23-Reactive Black 1) was purchased from India mart. All other chemicals used for mineral salts medium and synthetic wastewater preparation were of analytical grade and purchased from Merck, India.
The textile dyes (C.I. RR 195 -Reactive Red 195, C.I. RBL 1 -Reactive Brown 23and C.I. RBR 23-Reactive Black 1) was purchased from India mart. All other chemicals used for mineral salts medium and synthetic wastewater preparation were of analytical grade and purchased from Merck, India.
Bacterial Consortium and Culture Conditions
The bacterial consortium contained (Acinetobacter lwoffii, Acinetobacter albensis and Enterobacter cloacae) was isolated from soil samples of textile effluent contaminated sites from Manellore Industrial Effluent Treatment Company Ltd, Manellore. The bacterial consortium was enriched in Mineral Salts Medium (MSM) amended with 50 ppm concentrations of each of these mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23).
The bacterial consortium contained (Acinetobacter lwoffii, Acinetobacter albensis and Enterobacter cloacae) was isolated from soil samples of textile effluent contaminated sites from Manellore Industrial Effluent Treatment Company Ltd, Manellore. The bacterial consortium was enriched in Mineral Salts Medium (MSM) amended with 50 ppm concentrations of each of these mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23).
Graphical Abstract
The composition of the mineral salts medium used for enrichment and decolourization was as follows: Disodium hydrogen phosphate (Na2HPO4) 12.8 g/l; Potassium dihydrogen phosphate (KH2PO4) 3 g/l; Ammonium chloride (NH4Cl) 1 g/l; Sodium chloride (NaCl) 0.5 g/l; 0.05M Magnesium sulphate (MgSO4) 10 ml/l; 0.01M Calcium chloride (CaCl2) 10ml/l and 20% glucose at pH 8.5 (Tarun and Singh, 2012). The selected isolate was then purified by streaking on nutrient agar added with 100 mg/L of the dyes. The single colony pure cultures were stored in 15 % glycerol at -20°C.
To study the growth of the bacterial consortium with the mixed dyes present in the synthetic waste water (SWW) they were inoculated in 12g of NaCl, 0.049g NaPO4. 12H2O, 0.024g NaNO3, 0.255g KHSO4, and 1g of yeast were added for a 1L solution. This composition was found to be more similar to real textile wastewater (dos Santos., et al. 2007)
Bacterial Consortium Growth on Mixed Reactive Azo Dyes
To study the growth of the isolated bacterial consortium on SWW with the mixed reactive azo dyes (C.I. RR 195, C.I. RBL 1, and C.I.RBR 23), 50 mg/l of the individual dyes was mixed and added to 100 ml of SWW. 1 ml of pre-cultured bacterial consortium was added to each of the flasks containing SWW and incubated in a shaker at 37°C. Growth OD at 540 nm was taken till the 5th day and growth of the bacterial consortium was determined to observe the growth pattern on the removal of the mixed dyes.
To study the growth of the isolated bacterial consortium on SWW with the mixed reactive azo dyes (C.I. RR 195, C.I. RBL 1, and C.I.RBR 23), 50 mg/l of the individual dyes was mixed and added to 100 ml of SWW. 1 ml of pre-cultured bacterial consortium was added to each of the flasks containing SWW and incubated in a shaker at 37°C. Growth OD at 540 nm was taken till the 5th day and growth of the bacterial consortium was determined to observe the growth pattern on the removal of the mixed dyes.
Decolourization Assay
Assay was performed for mixed azo dye decolourisation (Pokharia and Ahluwalia. 2013). To study the decolourization of reactive azo dyes, a volume of 1ml of pre-cultured bacterial consortium was added to 50 ml of the SWW and supplemented with different concentrations (100, 200, 300, 400, 500 ppm) of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23). SWW added with reactive azo dyes alone served as control (abiotic control). The bio-decolourization of the mixed reactive dye (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) by the bacterial consortium was observed at 24h intervals of 5 days. In order to monitor the decolourization process, the samples were withdrawn periodically, centrifuged at 10,000 rpm for 15 min and decolourization was measured using UV/Vis spectra (Hitachi) at the corresponding λ max of the dye and was compared with the uninoculated control. All decolourization experiments were carried out in duplicate.
Assay was performed for mixed azo dye decolourisation (Pokharia and Ahluwalia. 2013). To study the decolourization of reactive azo dyes, a volume of 1ml of pre-cultured bacterial consortium was added to 50 ml of the SWW and supplemented with different concentrations (100, 200, 300, 400, 500 ppm) of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23). SWW added with reactive azo dyes alone served as control (abiotic control). The bio-decolourization of the mixed reactive dye (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) by the bacterial consortium was observed at 24h intervals of 5 days. In order to monitor the decolourization process, the samples were withdrawn periodically, centrifuged at 10,000 rpm for 15 min and decolourization was measured using UV/Vis spectra (Hitachi) at the corresponding λ max of the dye and was compared with the uninoculated control. All decolourization experiments were carried out in duplicate.
Effect of pH and Temperature on the decolourization of mixed dyes
In order to study the effect of pH and temperature, the sterilized SWW was amended with 50 ppm of each of the (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) dyes. The medium was maintained at different pH: 8, 8.5, 9, 9.5 and 10 respectively. A volume of 1 ml of overnight culture was inoculated in the flasks and incubated in a shaker at 36 ̊C. The effect of temperature was studied by inoculating overnight culture and incubating in a shaker at 22°C, 37°C, and 45°C. The measurement of decolourization of the total dye concentration and total protein estimation was performed every 24 hour interval for 5 days.
In order to study the effect of pH and temperature, the sterilized SWW was amended with 50 ppm of each of the (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) dyes. The medium was maintained at different pH: 8, 8.5, 9, 9.5 and 10 respectively. A volume of 1 ml of overnight culture was inoculated in the flasks and incubated in a shaker at 36 ̊C. The effect of temperature was studied by inoculating overnight culture and incubating in a shaker at 22°C, 37°C, and 45°C. The measurement of decolourization of the total dye concentration and total protein estimation was performed every 24 hour interval for 5 days.
Chemical Oxygen Demand Studies (COD)
COD determination was made according to the standard methods for examination of water and waste water (APHA 2017) 0.4 g of HgSO4 was placed in a reflux flask. 20 ml of the sample or an aliquot of sample diluted to 20 ml with distilled water was added and mixed well. Then, pumice stone or glass beads was added, followed by 10 ml of std. K2Cr207. 30 ml of H2SO4 containing Ag2SO4 were added slowly. The flask was connected to a condenser. Contents were refluxed for a minimum of 2 hrs. The condenser was cooled and washed down with distilled water and diluted for a minimum of 150 ml, cooled and titrated with excess of K2Cr2O7 using 0.1N ferrous ammonium sulphate as ferroin indicator. Sharp color change of blue-green to wine-red indicated the end point or completion of the titration. Blank was refluxed in the same manner using distilled water instead of sample.
COD determination was made according to the standard methods for examination of water and waste water (APHA 2017) 0.4 g of HgSO4 was placed in a reflux flask. 20 ml of the sample or an aliquot of sample diluted to 20 ml with distilled water was added and mixed well. Then, pumice stone or glass beads was added, followed by 10 ml of std. K2Cr207. 30 ml of H2SO4 containing Ag2SO4 were added slowly. The flask was connected to a condenser. Contents were refluxed for a minimum of 2 hrs. The condenser was cooled and washed down with distilled water and diluted for a minimum of 150 ml, cooled and titrated with excess of K2Cr2O7 using 0.1N ferrous ammonium sulphate as ferroin indicator. Sharp color change of blue-green to wine-red indicated the end point or completion of the titration. Blank was refluxed in the same manner using distilled water instead of sample.
Biological Oxygen Demand Studies (BOD) (APHA 2017)
The required volume of distilled water was aerated in a container by bubbling compressed air for 1-2 days to attain Dissolved Oxygen (DO) saturation. The temperature was maintained near 20°C. To each liter of diluted water, 1 ml of each of the phosphate buffer, magnesium sulphate, calcium chloride, and ferric chloride solutions was added and mixed well. In the case of the wastes which are not expected to have sufficient bacterial population, seeds were added to the diluted water. One bottle was kept for decolourization of the initial DO and 2 bottles were incubated at 20°C for 5 days. Care was taken that the bottles have a water seal. A blank was prepared for duplicate by siphoning plain diluted water (without seed) to measure the O2 consumption of dilution water. DO of the bottles kept for immediate DO determination and blank was fixed by adding 2 ml MnSO4 followed by 2 ml of alkali-iodide-azide as described in the estimation of DO. DO in the sample and in the blank were determined on initial day and after 5 days.
The required volume of distilled water was aerated in a container by bubbling compressed air for 1-2 days to attain Dissolved Oxygen (DO) saturation. The temperature was maintained near 20°C. To each liter of diluted water, 1 ml of each of the phosphate buffer, magnesium sulphate, calcium chloride, and ferric chloride solutions was added and mixed well. In the case of the wastes which are not expected to have sufficient bacterial population, seeds were added to the diluted water. One bottle was kept for decolourization of the initial DO and 2 bottles were incubated at 20°C for 5 days. Care was taken that the bottles have a water seal. A blank was prepared for duplicate by siphoning plain diluted water (without seed) to measure the O2 consumption of dilution water. DO of the bottles kept for immediate DO determination and blank was fixed by adding 2 ml MnSO4 followed by 2 ml of alkali-iodide-azide as described in the estimation of DO. DO in the sample and in the blank were determined on initial day and after 5 days.
Mixed Liquid Suspended Solids (MLSS) and Mixed Liquid Volatile Suspended Solids (MLVSS) Studies (APHA 2017)
The initial weight of the two crucibles was measured. 20 ml of reactor sample was taken and filtered in a 0.45 µm membrane filter assembly.10 ml of filtrate was taken in one crucible and 10 ml of unfiltered sample in one crucible. Then, the crucibles were kept in the hot water bath for complete evaporation. The crucibles were placed in a hot air oven at 150°C for 1 hr. The crucibles were kept in desiccator for 15 mins for cooling. The final weight of the crucibles was measured. Then, the crucibles were kept in a muffle furnace at 550°C for 1 hr. The crucibles were cooled in the desiccator for 15 minutes and the final weight of the crucible was measured.
The initial weight of the two crucibles was measured. 20 ml of reactor sample was taken and filtered in a 0.45 µm membrane filter assembly.10 ml of filtrate was taken in one crucible and 10 ml of unfiltered sample in one crucible. Then, the crucibles were kept in the hot water bath for complete evaporation. The crucibles were placed in a hot air oven at 150°C for 1 hr. The crucibles were kept in desiccator for 15 mins for cooling. The final weight of the crucibles was measured. Then, the crucibles were kept in a muffle furnace at 550°C for 1 hr. The crucibles were cooled in the desiccator for 15 minutes and the final weight of the crucible was measured.
Estimation of Total Phosphate Content in Synthetic Wastewater
The phosphate uptake in the wastewater sample were quantified by stannous chloride reduced molybdophosphoric acid blue method. The soluble phosphate content in the culture medium was estimated after 24, 42, and 72h of incubation by using the stannous chloride, calorimetric method (Saxena, 1994) and (APHA, 2017). After every 24h, 10 ml of the agitated sample was drawn from the series of individual flask and transferred into the centrifuge tubes of 15ml capacity under aseptic conditions. Then the tubes containing samples were centrifuged at 10,000 rpm at 15 min and the clear supernatant was used for soluble phosphate estimation at 690 nm by using spectrophotometer (UV-VIS Hitachi - U3210). Phosphate uptake efficiency (E) was calculated using the formula, E = [(I-F)/I] x 100 Where, I and F are the initial and final concentrations of phosphorous respectively. An efficiency value of 100% was obtained when no phosphate appeared in the water sample (i.e., F = 0).
The phosphate uptake in the wastewater sample were quantified by stannous chloride reduced molybdophosphoric acid blue method. The soluble phosphate content in the culture medium was estimated after 24, 42, and 72h of incubation by using the stannous chloride, calorimetric method (Saxena, 1994) and (APHA, 2017). After every 24h, 10 ml of the agitated sample was drawn from the series of individual flask and transferred into the centrifuge tubes of 15ml capacity under aseptic conditions. Then the tubes containing samples were centrifuged at 10,000 rpm at 15 min and the clear supernatant was used for soluble phosphate estimation at 690 nm by using spectrophotometer (UV-VIS Hitachi - U3210). Phosphate uptake efficiency (E) was calculated using the formula, E = [(I-F)/I] x 100 Where, I and F are the initial and final concentrations of phosphorous respectively. An efficiency value of 100% was obtained when no phosphate appeared in the water sample (i.e., F = 0).
Total Kjeldahl Nitrogen Estimation (TKN) (APHA 2017)
A suitable volume of well mixed sample was taken in kjeldahl flask. To that, 50 ml of digestion reagent was added. If the suspended solids concentration is high, large quantities of reagents were added to keep salt to acid ratio at 0.8. Digestion was continued for another 30 mins. After the sample appeared clear, to ensure complete decomposition/destruction of organic matter, it was cooled and diluted with distilled water to 150 or 300 ml depending on capacity of the flask. The flask was placed in its proper position in the distillation apparatus and heat was turned on. 0.5 ml phenolphthalein reagent was added, followed by sodium hydroxide-sodium thiosulphate reagent till pH rose just above 8.3. Post distillation, 200 ml of distillate was collected in 50 ml of boric acid. Plain boric acid was used for colorimetric estimation, and indicated boric acid for titrimetric estimation. The tip of the condenser was extended well below the level of boric acid solution. After distillation was complete, the flask containing distillate was removed first and then heat was put off to avoid back suction. The concentration of ammonia was measured by nesslerization as directed in ammonia estimation guidelines (APHA 2017). The distillate was titrated with 0.02 N H2SO4 till the indicator turned into a pale lavender color. A blank was also carried through all the steps and necessary corrections were applied.
A suitable volume of well mixed sample was taken in kjeldahl flask. To that, 50 ml of digestion reagent was added. If the suspended solids concentration is high, large quantities of reagents were added to keep salt to acid ratio at 0.8. Digestion was continued for another 30 mins. After the sample appeared clear, to ensure complete decomposition/destruction of organic matter, it was cooled and diluted with distilled water to 150 or 300 ml depending on capacity of the flask. The flask was placed in its proper position in the distillation apparatus and heat was turned on. 0.5 ml phenolphthalein reagent was added, followed by sodium hydroxide-sodium thiosulphate reagent till pH rose just above 8.3. Post distillation, 200 ml of distillate was collected in 50 ml of boric acid. Plain boric acid was used for colorimetric estimation, and indicated boric acid for titrimetric estimation. The tip of the condenser was extended well below the level of boric acid solution. After distillation was complete, the flask containing distillate was removed first and then heat was put off to avoid back suction. The concentration of ammonia was measured by nesslerization as directed in ammonia estimation guidelines (APHA 2017). The distillate was titrated with 0.02 N H2SO4 till the indicator turned into a pale lavender color. A blank was also carried through all the steps and necessary corrections were applied.
Analysis of Metabolites during the Decolourization Study
FTIR Spectroscopy Studies
Decolourization of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) by bacterial consortium was analyzed using FTIR. For this, 100 ml of culture broth was taken at 0 hr and 72 hr, centrifuged at 10,000g for 15 mins and the degraded metabolites were extracted from supernatant using equal volumes of ethyl acetate. The extracts were dried over anhydrous Na2SO4 and evaporated to dryness in a rotary evaporator (Arun Prasad., et al. 2013). The samples were mixed with KBr (1:20; 0.02 g of sample with KBr at a final weight of 0.4 g), ground, desorbed at 60°C for 24 hr and pressed to obtain IR- transparent pellets. The absorbance FT-IR spectra of the samples were recorded using an FTIR instrument model (IR affinity-1 (SHIMADZU)). The spectrum was collected within a scanning range of 400-4000 cm-1. The instrument was first calibrated for background signal scanning with a control sample of pure KBr and then the experimental samples were scanned. The FT-IR spectrum of the control was finally subtracted from the spectra of the non-degraded and degraded dyes.
FTIR Spectroscopy Studies
Decolourization of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) by bacterial consortium was analyzed using FTIR. For this, 100 ml of culture broth was taken at 0 hr and 72 hr, centrifuged at 10,000g for 15 mins and the degraded metabolites were extracted from supernatant using equal volumes of ethyl acetate. The extracts were dried over anhydrous Na2SO4 and evaporated to dryness in a rotary evaporator (Arun Prasad., et al. 2013). The samples were mixed with KBr (1:20; 0.02 g of sample with KBr at a final weight of 0.4 g), ground, desorbed at 60°C for 24 hr and pressed to obtain IR- transparent pellets. The absorbance FT-IR spectra of the samples were recorded using an FTIR instrument model (IR affinity-1 (SHIMADZU)). The spectrum was collected within a scanning range of 400-4000 cm-1. The instrument was first calibrated for background signal scanning with a control sample of pure KBr and then the experimental samples were scanned. The FT-IR spectrum of the control was finally subtracted from the spectra of the non-degraded and degraded dyes.
Scanning Electron Microscopy
The sample preparation for Scanning Electron Microscopy (SEM) was carried out according to the method of Prior and Perkins (1974). The isolated bacterial strains were grown individually on MSM for 24h. The bacterial strains in the mineral salts medium were centrifuged at 8000g for 10min and the pellets were immediately re-suspended in 2% glutaraldehyde with 0.05M phosphate buffer and 4% sucrose (pH 7.3). Fixation was obtained overnight at 4°C. After 24 hours the pellets were centrifuged at 8000g for 10min, washed 4 times with distilled water, placed on aluminum foil. The samples were then dehydrated with series of gradient ethanol (i.e 10%, 20%, 30% till 90%), air dried and finally the dried flakes were coated with platinum and examined under SEM (JEOL JSM-6360).
The sample preparation for Scanning Electron Microscopy (SEM) was carried out according to the method of Prior and Perkins (1974). The isolated bacterial strains were grown individually on MSM for 24h. The bacterial strains in the mineral salts medium were centrifuged at 8000g for 10min and the pellets were immediately re-suspended in 2% glutaraldehyde with 0.05M phosphate buffer and 4% sucrose (pH 7.3). Fixation was obtained overnight at 4°C. After 24 hours the pellets were centrifuged at 8000g for 10min, washed 4 times with distilled water, placed on aluminum foil. The samples were then dehydrated with series of gradient ethanol (i.e 10%, 20%, 30% till 90%), air dried and finally the dried flakes were coated with platinum and examined under SEM (JEOL JSM-6360).
Phytotoxicity Study
The phytotoxicity study was carried out at room temperature using B. nigra (Mustard) and T. foenum graecum (Fenugreek) (10 seeds of each), by adding separately 5 ml of untreated and treated synthetic wastewater and mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23). The control set was carried out using distilled water at the same time. Germination (%) and the length of plumule and radicle were recorded after 7 days (Jadhav., et al. 2010).
The phytotoxicity study was carried out at room temperature using B. nigra (Mustard) and T. foenum graecum (Fenugreek) (10 seeds of each), by adding separately 5 ml of untreated and treated synthetic wastewater and mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23). The control set was carried out using distilled water at the same time. Germination (%) and the length of plumule and radicle were recorded after 7 days (Jadhav., et al. 2010).
Bench Scale Reactor Studies With Synthetic Textile Effluent Wastewater
A bioreactor of 3L capacity was used for laboratory study in the treatment of textile effluent contaminated wastewater. 12g of Na Cl, 0.049g Na3PO4. 12H2O, 0.024g NaNO3, 0.255g KHSO4, and 1gof yeast were added for a 1L solution. This composition was chosen to be more similar to real waste water (dos Santos., et al. 2007). The bacterial consortium was acclimatized in the synthetic textile containing wastewater for 5 days for scaling up the volume of consortium. The bioreactor was filled with 2L of the synthetic textile effluent containing wastewater to which 100ml of bacterial consortium was added. Temperature was maintained at 37°C and the solution was continuously aerated with constant agitation using stirrer at 300 rpm. The samples from the reactor were analyzed for chemical parameters such as decolourization and COD and total protein every 24 hours (Pokharia., et al. 2013).
A bioreactor of 3L capacity was used for laboratory study in the treatment of textile effluent contaminated wastewater. 12g of Na Cl, 0.049g Na3PO4. 12H2O, 0.024g NaNO3, 0.255g KHSO4, and 1gof yeast were added for a 1L solution. This composition was chosen to be more similar to real waste water (dos Santos., et al. 2007). The bacterial consortium was acclimatized in the synthetic textile containing wastewater for 5 days for scaling up the volume of consortium. The bioreactor was filled with 2L of the synthetic textile effluent containing wastewater to which 100ml of bacterial consortium was added. Temperature was maintained at 37°C and the solution was continuously aerated with constant agitation using stirrer at 300 rpm. The samples from the reactor were analyzed for chemical parameters such as decolourization and COD and total protein every 24 hours (Pokharia., et al. 2013).
Results
Decolorization of Mixed Dyes (C.I.RR 195, C.I.RBL 1 and C.I.RBR 23) bythe Bacterial Consortium at Different Concentrations
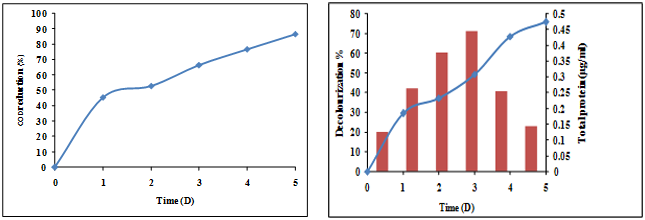
The ability of the isolated bacterial consortium to degrade C.I.RR 195, C.I.RBL 1 and C.I.RBR 23 was studied at 50 ppm to 250 ppm concentrations (50 ppm, 100 ppm, 150 ppm, 200 ppm and 250 ppm) in the SWW. Decolourization of mixed reactive dyes by the isolated consortium at different concentrations is represented in the Figure 1. The decolorization at 50 ppm concentrations varied from 25.7% (1st day) to 91.8% at the end of the 5th day. Once the concentration was increased to 100 ppm, the decolourization efficiency started to decrease to 87% (5th day).
The ability of the isolated bacterial consortium to degrade C.I.RR 195, C.I.RBL 1 and C.I.RBR 23 was studied at 50 ppm to 250 ppm concentrations (50 ppm, 100 ppm, 150 ppm, 200 ppm and 250 ppm) in the SWW. Decolourization of mixed reactive dyes by the isolated consortium at different concentrations is represented in the Figure 1. The decolorization at 50 ppm concentrations varied from 25.7% (1st day) to 91.8% at the end of the 5th day. Once the concentration was increased to 100 ppm, the decolourization efficiency started to decrease to 87% (5th day).
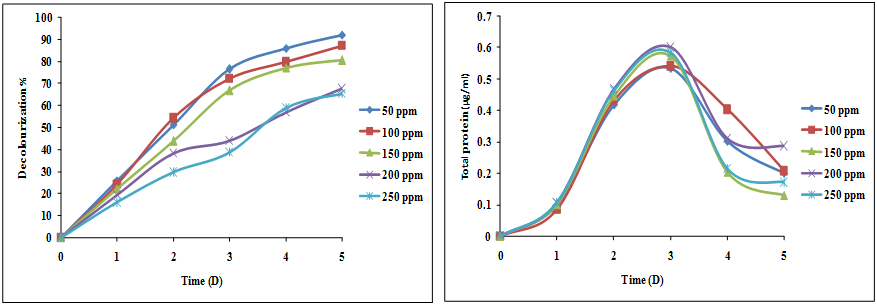
Figure 1: Decolourization of various concentrations of mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23).
Effect of pH and Temperature on the decolourization of C.I. RR 195, C.I.RBL 1 and C.I.RBR 23 (50 ppm)
Alkaline pH plays an important role in the metabolism of alkaliphilic bacteria. This study was carried out to determine the effect of pH (pH 8, pH 8.5, pH 9, pH 9.5, and pH 10) on the decolourization of C.I. RR 195, C.I. RBL 1 and C.I. RBR 23 dyes at optimum concentration of 50 ppm respectively in SWW. It was observed that at pH 8, the degradation percentage was found to be 83.2% by the end of the 5th day, represented by Figure 2. Minimum decolourization was found to be 68.3% at pH 10. The optimum pH was found to be 8 for maximum removal of dyes. Similar studies have reported on the effect of pH (7, 8 and 9) on decolourization of four reactive dyes (Reactive yellow, Reactive red, Reactive brilliant red and Reactive brilliant blue) at 50 mg/l concentrations of dyes with 20% inoculum. The percentage of removal of the mixed dyes increased from increase in incubation period at pH 9. The maximum removal 80%, 73%, 63% and 63.5% dyes respectively were found at pH 9 by Bacillus spp. At pH 7 and 8 percentage removal of dye decreased when compared to pH 9, (Maulin Shah., et al. 2014).
Alkaline pH plays an important role in the metabolism of alkaliphilic bacteria. This study was carried out to determine the effect of pH (pH 8, pH 8.5, pH 9, pH 9.5, and pH 10) on the decolourization of C.I. RR 195, C.I. RBL 1 and C.I. RBR 23 dyes at optimum concentration of 50 ppm respectively in SWW. It was observed that at pH 8, the degradation percentage was found to be 83.2% by the end of the 5th day, represented by Figure 2. Minimum decolourization was found to be 68.3% at pH 10. The optimum pH was found to be 8 for maximum removal of dyes. Similar studies have reported on the effect of pH (7, 8 and 9) on decolourization of four reactive dyes (Reactive yellow, Reactive red, Reactive brilliant red and Reactive brilliant blue) at 50 mg/l concentrations of dyes with 20% inoculum. The percentage of removal of the mixed dyes increased from increase in incubation period at pH 9. The maximum removal 80%, 73%, 63% and 63.5% dyes respectively were found at pH 9 by Bacillus spp. At pH 7 and 8 percentage removal of dye decreased when compared to pH 9, (Maulin Shah., et al. 2014).
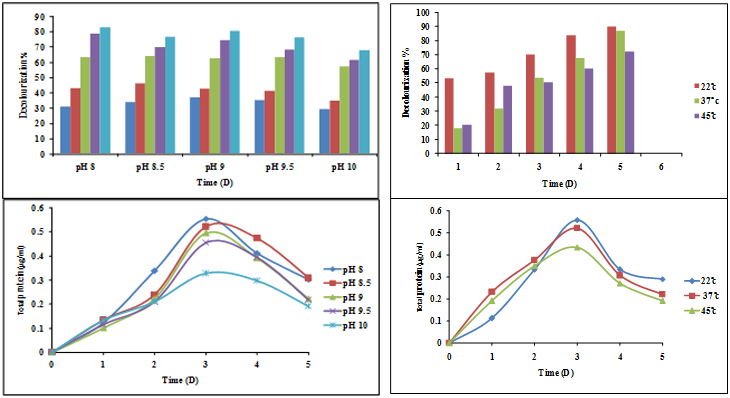
Figure 2: Effect of pH and temperature on decolourization of mixed reactive dyes
C.I. RR 195, C.I. RBL 1 and C.I. RBR 23 and total protein content (50 ppm).
Effect of temperature (28°C, 37°C and 45°C) was studied by inoculating the isolated bacterial consortium in SWW containing C.I. RR 195, C.I. RBL 1 and C.I. RBR 23 dyes at an optimum concentration of 50 ppm respectively. At 28°C, enhanced bacterial consortium showed maximum decolourization of the mixed dyes which was found to be 90% by the end of the 5th day, represented by Figure 2. In the present work optimization studies were carried out at different temperatures (28°C, 37°C and 45°C) out of which only 28°C enhanced the growth of the bacterial consortium and showed maximum decolourization of the mixed dyes which was found to be 90% by the end of the 5th day. Decolorizing activity significantly decreased from increase in the temperature from 37°C to 45°C. The minimum decolourization of mixed dyes was found to be 72.5% at 45°C. Pratiksha Pradhan., et al. (2012) has reported the optimum temperature for dye decolourization of Reactive red 31 and Reactive yellow 81 as 35°C by Streptococcus sp.
Reduction of COD/BOD and its Biomass Content (MLSS AND MLVSS) and Removal of TKN/Phosphate
Synthetic textile effluent in the present study was evaluated for the reduction in COD and BOD values. At every 24-hour interval their physico-chemical parameters were analyzed. A pattern of reduction in parameters was observed, which showed a continuous reduction from the first day and a maximum reduction observed at the end of fifth day of incubation. The reduction of COD and BOD was found to be 1840 mg/L and 1760 mg/L (1st day) to 350 mg/L and 80 mg/L at the end of the 5th day respectively. The biomass content (MLSS and MLVSS) was found to reduce from 1000 mg/L and 890 mg/L (1st day) to 950 mg/L and 950 mg/L by the end of the 5th day respectively. During the period of treatment for diluted SWW (80:20), it could be noted that the bacterial consortium utilized the mixed dyes and it was also interpreted by showing the reduction in TKN/P nutrients. Initial TKN/P values were (168 mg/L) and P (13.31 mg/L), which reduced to 125.7 mg/L and 2.32 mg/L respectively by the end of the 5th day, represented in Figure 3.
Synthetic textile effluent in the present study was evaluated for the reduction in COD and BOD values. At every 24-hour interval their physico-chemical parameters were analyzed. A pattern of reduction in parameters was observed, which showed a continuous reduction from the first day and a maximum reduction observed at the end of fifth day of incubation. The reduction of COD and BOD was found to be 1840 mg/L and 1760 mg/L (1st day) to 350 mg/L and 80 mg/L at the end of the 5th day respectively. The biomass content (MLSS and MLVSS) was found to reduce from 1000 mg/L and 890 mg/L (1st day) to 950 mg/L and 950 mg/L by the end of the 5th day respectively. During the period of treatment for diluted SWW (80:20), it could be noted that the bacterial consortium utilized the mixed dyes and it was also interpreted by showing the reduction in TKN/P nutrients. Initial TKN/P values were (168 mg/L) and P (13.31 mg/L), which reduced to 125.7 mg/L and 2.32 mg/L respectively by the end of the 5th day, represented in Figure 3.
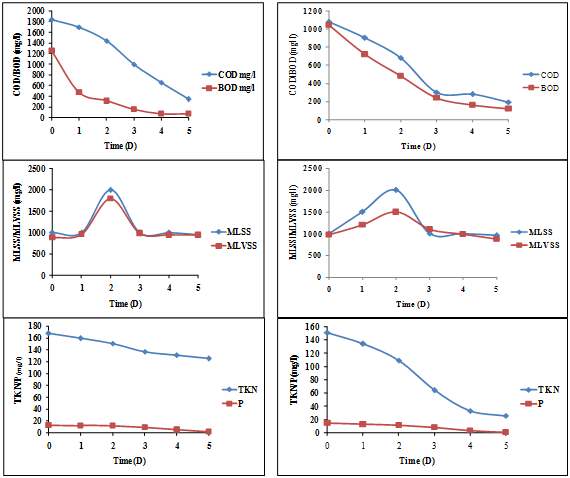
Figure 3: Reduction of COD/BOD and its biomass content (MLSS and MLVSS)
and removal of TKN/P in diluted SWW (80:20) and (50:50)
It could be noted that the initial COD and BOD concentration was as high as 1080 mg/L and 1040 mg/L, which reduced to 190 mg/L and 120 mg/L by the end of the 5th day. The biomass content (MLSS and MLVSS) was recorded as 1000 mg/L and 980mg/L (1st day) and reduced to 960 mg/L and 880 mg/L respectively by the end of the 5th day. During the period of treatment with the diluted SWW (50:50), it was noted that the bacterial consortium utilized the mixed dyes and it was also corroborated by the reduction in TKN/P nutrients. Initial TKN/P values were 151.2 mg/L and 15.2 mg/L (1st day), which reduced to 25.8 mg/L and 1.25 mg/L respectively by the end of the 5th day, represented in Figure 3.
Anju Francis., et al. (2016) reported 86% COD removal in moving bed biofilm reactor using Box Behnken method with carriers inoculated with Microbacterium marinilacus, isolated from textile sludge after fluidized bed Fenton pre-treatment. Abioye., et al. (2014) reported that the COD and BOD reduction was found to be 2060 mg/l to 1430 mg/l and 824 mg/l to 572 mg/l respectively after 10 days of incubation by bacterial consortium and he also reported COD and BOD decrease after 15 days of incubation which is 2060 mg/l to 1300 mg/l and 824 mg/l to 520 mg/l respectively. They also reported P and TKN reduction from 2.64 mg/l to 2.5 mg/l and 256.8 mg/l to 53.2 mg/l respectively after 10 days of incubation, 2.64 mg/l to 2.38 mg/l (P), 256.8 mg/l to 30.3 mg/l (TKN) after 13 days of incubation and 2.64 mg/l to 2.38 mg/l (P), 256.8 mg/l to 30.3 mg/l (TKN) after 15 days of incubation by bacterial consortium.
Reduction of COD/BOD and its Biomass Content (MLSS AND MLVSS) and Removal of TKN/Phosphate in SWW
In real time SWW without dilution, the batch study results showed that the initial COD and BOD of wastewater was found to be 1640 mg/L and 1200 mg/L, which reduced to 620 mg/L and 140 mg/L respectively. The growth of the bacterial consortium by utilizing the dyes was further proved by MLSS and MLVSS values of 1000 mg/L and 950 mg/L (1st day), which was reduced to 1000 mg/L and 890 mg/L in the wastewater by the end of the 5th day respectively. During the period of treatment for the raw SWW (100%), it could be noted that the bacterial consortium utilized the mixed dyes and a reduction in TKN/P nutrients also proved this. Initial TKN/P values were 274.4 mg/L and 16.4 mg/L (1st day), which decreased to 188.9 mg/L and 0.08 mg/L respectively by the 5th day, represented in Figure 4.
In real time SWW without dilution, the batch study results showed that the initial COD and BOD of wastewater was found to be 1640 mg/L and 1200 mg/L, which reduced to 620 mg/L and 140 mg/L respectively. The growth of the bacterial consortium by utilizing the dyes was further proved by MLSS and MLVSS values of 1000 mg/L and 950 mg/L (1st day), which was reduced to 1000 mg/L and 890 mg/L in the wastewater by the end of the 5th day respectively. During the period of treatment for the raw SWW (100%), it could be noted that the bacterial consortium utilized the mixed dyes and a reduction in TKN/P nutrients also proved this. Initial TKN/P values were 274.4 mg/L and 16.4 mg/L (1st day), which decreased to 188.9 mg/L and 0.08 mg/L respectively by the 5th day, represented in Figure 4.
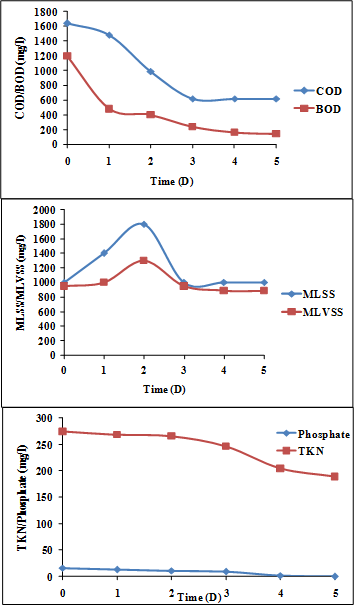
Figure 4: Reduction of COD/BOD and its biomass content (MLSS and MLVSS)
and removal of TKN/P in diluted SWW (100%).
Analysis of Metabolites during the Decolourization Study
FTIR Spectroscopy Results
The FTIR spectrum of the mixed azo reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) used as a control and decolourized mixed reactive azo dyes (72 hrs) were compared for evaluating the production of metabolites during the decolourization. The spectrum of the control dye (Figure 5) displayed a peak at 3442.12 cm-1 and 2348 cm-1 indicating the presence of N-H stretching. The 1614.49 cm-1 peak indicates the presence of (-N = N-) azo group. The peak at 1393.63 cm-1 is present due to C-H bending. The peak at 1115.87 cm-1 indicates the presence of C-F halogen. The peak at 662.58 cm-1 indicates the presence of C-Br group.
FTIR Spectroscopy Results
The FTIR spectrum of the mixed azo reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) used as a control and decolourized mixed reactive azo dyes (72 hrs) were compared for evaluating the production of metabolites during the decolourization. The spectrum of the control dye (Figure 5) displayed a peak at 3442.12 cm-1 and 2348 cm-1 indicating the presence of N-H stretching. The 1614.49 cm-1 peak indicates the presence of (-N = N-) azo group. The peak at 1393.63 cm-1 is present due to C-H bending. The peak at 1115.87 cm-1 indicates the presence of C-F halogen. The peak at 662.58 cm-1 indicates the presence of C-Br group.
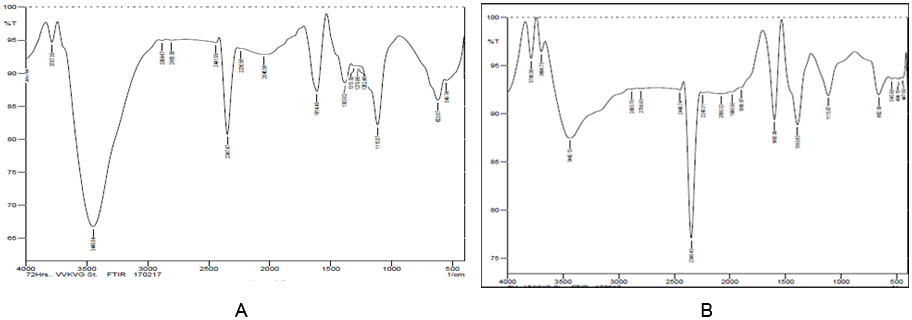
Figure 5: FTIR spectrum of degraded metabolites by the bacterial consortium
A. 0 hr and B. 72 hr (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23).
A. 0 hr and B. 72 hr (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23).
After decolourization, the FTIR spectrum of extracted metabolites (72 hr) (Figure 5) showed a considerable change in the position of peaks as compared to the spectrum of the control dye. A new peak at 3449.84 cm-1 was found due to the presence of N-H stretching in the extracted metabolites. A new narrow peak at 2347.47 cm-1 indicated the presence of carbon dioxide asymmetric stretching. The peak at 622.07 cm-1 indicates the presence of C-Br stretching found in the FTIR spectrum. The reduction of peaks at 1600.99 cm-1 indicates the reductive cleavage of C.I. RR 195, C.I. RBL 1 and C.I. RBR 23 dyes at azo bond position (-N = N-) when compared to the control dyes spectrum. The peak at 1383.02 cm-1was found due to degradation of primary amine. The two peak values (1600.99 cm-1 and 1383.02 cm-1) in the FTIR spectrum proved the changes in functional groups in the mixed reactive dyes. The breakage of the aromatic ring of the mixed reactive azo dyes indicated the decolourization of mixed reactive azo dyes by the bacterial consortium under alkaline conditions.
Harshad., et al. (2015) reported the FTIR spectrum of extracted products after decolourization of dye RB 172 showing variation in the position of peaks when compared to control dye spectrum. The disappearance of peaks at 1620.29 cm-1 and 1598 cm-1 indicated the reductive cleavage of dye RB 172 at the azo bond position. The peak at 2974.86 cm-1 indicates the C-H stretching of alkanes while the peak at 2833.71 cm-1 shows C-H stretching of ethers. The peak obtained at 1442.05 cm-1 is due to C-H deformation of alkanes. In addition, peak at 1246.96 cm-1 shows O-NO2 vibrations of nitrates while the peak at 1067.06 cm-1 suggests C-OH stretching to primary alcohols. These changes in the FTIR spectrum are clear evidence for the degradation of dye RB 172 into simpler molecules, like aliphatic amines and carboxylic acids.
Phytotoxicity study on mixed reactive dyes (c.i. rr 195, c.i. rbl 1 and c.i. rbr 23) and its degraded metabolites
Seed germination and plant growth bioassay are the most common techniques used to evaluate the phytotoxicity of the dyes. Thus, the primary aim was to assess the phytotoxicity of the dyes and its degraded metabolites produced by the bacterial consortium. Germination of mustard and fenugreek (B. nigra and T. foenum graecum) seeds was less with mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) as compared to its degradation metabolites and plain water (Figure 6). The length of plumule and radicle were significantly affected by mixed reactive dyes than its degraded metabolites (Table 1), indicating the toxic nature of the dyes compared to their degraded metabolites. Ajay Kumar., et al. (2015) reported that in the phytotoxicity study, the 3000 ppm of RR 198 has caused inhibition of germination of P. mungo and C. arietinum seeds by 50% and 60% respectively and the length of radicle and plumule were decreased by 65% and 70 % in comparison to control respectively. The degraded sample has not shown any inhibitory effect on seed germination and the length of radicle and plumule decreased by 15% and 10% respectively.
Seed germination and plant growth bioassay are the most common techniques used to evaluate the phytotoxicity of the dyes. Thus, the primary aim was to assess the phytotoxicity of the dyes and its degraded metabolites produced by the bacterial consortium. Germination of mustard and fenugreek (B. nigra and T. foenum graecum) seeds was less with mixed reactive dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) as compared to its degradation metabolites and plain water (Figure 6). The length of plumule and radicle were significantly affected by mixed reactive dyes than its degraded metabolites (Table 1), indicating the toxic nature of the dyes compared to their degraded metabolites. Ajay Kumar., et al. (2015) reported that in the phytotoxicity study, the 3000 ppm of RR 198 has caused inhibition of germination of P. mungo and C. arietinum seeds by 50% and 60% respectively and the length of radicle and plumule were decreased by 65% and 70 % in comparison to control respectively. The degraded sample has not shown any inhibitory effect on seed germination and the length of radicle and plumule decreased by 15% and 10% respectively.

Figure 6: Phytotoxicity studies of mixed dyes and their extracted metabolites
(a. Control b. Mixed dyes c. Extracted metabolites).
| Parameter studied | Water | Mixed reactive dyes | Extracted metabolites |
| Germination (%) | 100 | 60 | 100 |
| Plumule (cm) | 7.23 | 3.55 | 7.12 |
| Radicle (cm) | 9.70 | 5.76 | 9.53 |
a. B. nigra (Mustard)
| Parameter studied | Water | Mixed reactive dyes | Extracted metabolites |
| Germination (%) | 100 | 45 | 88.43 |
| Plumule (cm) | 6.98 | 3.23 | 5.76 |
| Radicle (cm) | 4.26 | 2.20 | 4.19 |
b. T. foenum graecum (Fenugreek)
Table 1: Phytotoxicity study on mixed reactive dyes and its degraded metabolites.
Table 1: Phytotoxicity study on mixed reactive dyes and its degraded metabolites.
Sacle Bioreactor Studies with Synthetic Textile Effluent Wastewater
The isolated bacterial consortium which was able to degrade mixed reactive dyes azo dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) under alkaline conditions was used in the treatment of textile effluent containing contaminated wastewater in a lab-scale bioreactor using 50 ppm of mixed dyes (Figure 7). The color removal was found to be 75.9% and the COD reduction was found to be 86.7% at the end of the 5th day (Figure 7). In the present study, bacterial strains were isolated from the consortium of which three bacterial strains were identified by 16s rRNA sequencing as Acinetobacter lwoffii, Acinetobacter albensis and Enterobacter cloacae (data not shown), which could decolourize the mixed reactive azo dyes up to 250 mg/L in the SWW. Similar studies were carried out by Rajaganesh., et al. 2014 with bacterial consortium which contained Bacillus subtilis, Pseudomonas aeruginosa, Pseudomonas fluorescens, Serratia martestens and Escheriachia coli from textile dye effluents to evaluate the potential on decolourization of Reactive red M5B, Reactive blue 19, Reactive yellow 44, Reactive orange M2R and Reactive green 19 azo dyes in MSM. While the present study contained three moderately alkaliphilic bacterial isolates in the consortium which were able to remove 250 mg/L of the mixed dyes. However, Rashid., et al. (2014) showed that decolourization of azo dyes up to 200 ppm was possible using 6 isolates under non-alkaliphilic conditions.
The isolated bacterial consortium which was able to degrade mixed reactive dyes azo dyes (C.I. RR 195, C.I. RBL 1 and C.I. RBR 23) under alkaline conditions was used in the treatment of textile effluent containing contaminated wastewater in a lab-scale bioreactor using 50 ppm of mixed dyes (Figure 7). The color removal was found to be 75.9% and the COD reduction was found to be 86.7% at the end of the 5th day (Figure 7). In the present study, bacterial strains were isolated from the consortium of which three bacterial strains were identified by 16s rRNA sequencing as Acinetobacter lwoffii, Acinetobacter albensis and Enterobacter cloacae (data not shown), which could decolourize the mixed reactive azo dyes up to 250 mg/L in the SWW. Similar studies were carried out by Rajaganesh., et al. 2014 with bacterial consortium which contained Bacillus subtilis, Pseudomonas aeruginosa, Pseudomonas fluorescens, Serratia martestens and Escheriachia coli from textile dye effluents to evaluate the potential on decolourization of Reactive red M5B, Reactive blue 19, Reactive yellow 44, Reactive orange M2R and Reactive green 19 azo dyes in MSM. While the present study contained three moderately alkaliphilic bacterial isolates in the consortium which were able to remove 250 mg/L of the mixed dyes. However, Rashid., et al. (2014) showed that decolourization of azo dyes up to 200 ppm was possible using 6 isolates under non-alkaliphilic conditions.
Discussion
The degradation of azo dyes was found effective at 5th day and it decreased as concentration of dye was increased. The decolourisation was enhanced at pH of 9 (Maulin Shah., et al. 2014). It was observed that the degradation by Bacillus sps was efficient at 28ºC but Pratiksha Pradhan., et al. (2012), results showed, the degradation was efficient at 37ºC. The decrease in the COD, BOD and the toxicity OD dyes and its metabolites were also studied and it showed increase in length of plumule and radicle than in seeds treated with azo dyes. This shows that the biological degradation of azo dyes also reduces the toxicity of the dyes.
Conclusion
Application of traditional wastewater treatment requires enormous cost and continuous input of chemicals which becomes uneconomical and causes further environmental damage. This work reports that an enriched bacterial consortium can efficiently decolourize 50 ppm of mixed reactive azo dyes efficiently up to 91.8% within 5 days. The bacterial consortium exhibited maximum decolourization ability of mixed reactive azo dyes at pH 8 at 28°C. Maximum decolourization of mixed reactive dyes by bacterial consortium was exhibited. The physico-chemical parameters such as COD, BOD, MLSS and MLVSS, TKN and P played an important role for the decolourization of mixed reactive azo dyes by the bacterial consortium. The phytotoxicity study and the lab-scale reactor study of real time SWW act as important applications to test the removal of mixed azo dyes by the isolated alkaliphilic bacterial consortium. This shows that the isolated bacterial consortium has enormous potential to degrade the textile dyes and resolve the problem of high COD/BOD content present in the industrial application.
Acknowledgements
The authors would like to acknowledge the management of Stella Maris College for the financial support provided by them and our special acknowledgements to our Sister Principal Jasintha Quadras for her moral supportto carry out the research work.
The authors would like to acknowledge the management of Stella Maris College for the financial support provided by them and our special acknowledgements to our Sister Principal Jasintha Quadras for her moral supportto carry out the research work.
References
- Abioye OP., et al. “Biological Treatment of Textile Effluent Using Candida zeylanoides and Saccharomyces cerevisiae isolated from Soil”. Advances in Biology (2014): 4.
- American Public Health Association. In: Standard Methods for the Examination of Water and Wastewater, 23rd ed. APHA, Washington, DC (2017).
- Arun Prasad AS and Bhaskara Rao KV. “Aerobic biodegradation of azo dye Acid Black-24 by Bacillus halodurans”. Journal of Environmental Biology 35.3 (2014): 549-554.
- Basha S., et al. “Microbial bioremediation of heavy metals from textile industry dye effluents using isolated bacterial strains”. International Journal of Current Microbiology and Applied Sciences3.5 (2014): 785-794.
- Chinnathambi A. “Industrial Important Enzymes from Alkaliphiles–An Overview”. Biosciences Biotechnology Research Asia 12.3 (2015): 2007-2016.
- Deepa K., et al. “Bioremoval of Direct Red from aqueous solution by Pseudomonas putida and its adsorption isotherms and kinetics”. Ecological engineering 58 (2013): 207-213.
- Dos Santos AB., et al. “Review paper on current technologies for decolourisation of textile wastewaters: perspectives for anaerobic biotechnology”. Bio resource technology 98.12 (2007): 2369-2385.
- Francis A and Sosamony KJ. “Treatment of Pre-treated Textile Wastewater using Moving Bed Bio-film Reactor”. Procedia Technology 24 (2016): 248-255.
- Guo H., et al. “Bioremediation of heavy metals by growing hyperaccumulaor endophytic bacterium Bacillus sp. L14”. Bioresource Technology 101.22 (2010): 8599-8605.
- Jadhav JP., et al. “Evaluation of the efficacy of a bacterial consortium for the removal of color, reduction of heavy metals, and toxicity from textile dye effluent”. Bio resource Technology 101.1 (2010): 165-173.
- Jamalpure MS Trupti. “Studies on the alkaline amylase from an alkaliphilic Streptomyces species NCL 716”. Division of Biochemical Sciences National Chemical Laboratory (2012).
- Karthik V., et al. “An overview of treatments for the removal of textile dyes”. Journal of Chemical and Pharmaceutical Sciences 7.4 (2014): 301-307.
- Kaveh R. Transcriptional responses of soybean (Glycine max) and thale cress (Arabidopsis thaliana) (2014) plants exposed to different classes of environmental contaminants. Temple University.
- Khan S and Abdul M. “Environmental and health effects of textile industry wastewater”. Environmental Deterioration and Human Health (2014): 55-71.
- Kurade MB., et al. “Biodegradation and detoxification of textile dye Disperse Red 54 by Brevibacillus laterosporus and determination of its metabolic fate”. Journal of Bioscience and Bioengineering 121.4 (2016): 442-449.
- Lade H., et al. “Biodegradation and detoxification of textile azo dyes by bacterial consortium under sequential microaerophilic/aerobic processes”. EXCLI journal 14 (2015): 158-174.
- Pandey A., et al. “Bacterial decolorization and degradation of azo dyes”. International Bio deterioration & Biodegradation 59.2 (2007): 73-84.
- Pandey., et al. “Microbial Decolorization and Degradation of Reactive Red 198 Azo Dye by a Newly Isolated Alkaligenes Species”. Proceedings of the National Academy of Sciences India Section B: Biological Sciences 86.4 (2016): 805-815.
- Pokharia A and Sarabjeet SA. “Isolation and screening of dye decolorizing bacterial isolates from contaminated sites”. Textiles and Light Industrial Science and Technology (2013).
- Pokharia Anamika and Sarabjeet Singh Ahluwalia. “Isolation and screening of dye decolorizing bacterial isolates from contaminated sites”. Textiles and Light Industrial Science and Technology (2013).
- Pradhan P and Harsh Dev K. “Degradation of azo and triphenyl methane dye by the bacteria isolated from local industrial waste”. International Journal of Current Research and Review 4.20 (2012): 39-49.
- Prior RB and Perkins RL. “Artifacts induced by preparation for SEM in Proteus mirabilis exposed to carbenicillin”. Canadian Journal of Microbiology 20.5 (1974): 794-795.
- Rahmad N., et al. “Comparative proteomic analysis of different developmental stages of the edible mushroom Termitomyces heimii”. Biological research 47.1 (2014): 30.
- Rajeswari K., et al. “Biodegradation of Reactive Dyes by an isolated bacterium Lysinibacillus Sphae Ricus Rsv-1”. The IIOAB Journal 5.3 (2014): 19.
- Sahasrabudhe., et al. “Decolorization and detoxification of sulfonated toxic diazo dye CI Direct Red 81 by Enterococcus faecalis YZ 66”. Journal of Health Science and Engineering 12.1 (2014): 151.
- Saxena MM. Environmental Analysis – Water, Soil and Air. Agro Botanical Publishers (India) (1994).
- Shah Maulin P. “An Innovative Approach to Biodegradation of Textile Dye (Remazol Black B) by Bacillus Spp”. International Journal of Environmental Bioremediation & Biodegradation 1.2 (2013): 43-48.
- Sriram N., et al. “Biological degradation of Reactive dyes by using bacteria isolated from dye effluent contaminated soil”. Middle–East Journal of Scientific Research 17.12 (2013): 1695-1700.
Citation:
Velvizhi M and Veena Gayathri Krishnaswamy. “Alkaliphilic Bacterial Consortium Used in the Removal of Mixed Reactive
Dyes for the Treatment of Textile Effluent”. Clinical Biotechnology and Microbiology 2.2 (2018): 317-330.
Copyright: © 2018 Velvizhi M and Veena Gayathri Krishnaswamy. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.






























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.