Research Article
Volume 2 Issue 6 - 2018
Screening of the Yeast Phenols and Flavonoids by Many Analytical Methods
1Botany & Microbiology Department, Faculty of Science, Assiut University, PO.Box. 71516, Assiut, Egypt
2Department of Pharmacognosy, Faculty of Pharmacy, Assiut University, PO Box.71516, Assiut, Egypt
2Department of Pharmacognosy, Faculty of Pharmacy, Assiut University, PO Box.71516, Assiut, Egypt
*Corresponding Author: Eman M Mohamed, Botany & Microbiology Department, Faculty of Science, Assiut University, PO.Box. 71516,
Assiut, Egypt.
Received: September 07, 2018; Published: September 27, 2018
Abstract
Screening 80 yeast methanolic extracts their flavonoids producivity by many analytical methods includes spectrophotometer at 362 nm, ammonium test and TLC analysis. Spectrophotometric reading recorded that the flavonoids contents ranged between 14.9 ± 1-105.8 ± 4.3 µg/g DW, classified into three categories includes 20, 24 and 36 are high, moderate and low productivity, respectively. Ammonium test cleared that each yeast extract spot has espicall kinds of flavonoids appears by distinguished color includes yellow, orange, yellow green and brown, the color intensty depend up on the amounts of flavonoids. Select the deep yellow spots for confirmed by TLC with using rutin and quercetin as standard. The highest flavonoids producers were tested for production of phenols by HPLC analysis using gallic acid as standard, recorded gallic acid ranged between 1000-23500 µg/L. Seven phenols recorded by GC/MS in the highest flavonoids Diutina rugosa MH333102 strain.
Keywords: Yeast; Phenols; Flavonoids; Spectrophotometrically; Ammonium test; TLC; HPLC; GC/MS analysis; Gallic acid; Quercetin and Rutin
Introduction & Background
Since ancient human civilizations yeast are known and used in bread and alcoholic beverage production [De-Oca., et al. 2016]. Saccharomyces cerevisiae and many other benefit yeast are applied on bio-technological, industrial and commercial scales for production of many fermented products such as baking, single cell proteins, fermented foods, alcoholic beverages, ethanol production and biofuel, research (chemical, biological, and genetic as a model organism), enzymes production which use in (paper, skin and tissue industries), flavoring agents, pharmacology, medicine, bioremediation, animal feeding and soil fertility [De- Vuyst & Neysens, 2005, Daniel., et al. 2009, Kurtzman., et al. 2011, N’guessan., et al. 2011, Pham., et al. 2011, Chan., et al. 2012]. Phenols and flavonoids are secondary bioactive metabolites synthesized in each living cells in animals and plant kingdom such as higher plants, bacteria, yeast, fungi, and algae [Huynh., et al. 2014, De- Carvalho., et al. 2016]. It derived from two metabolic pathways includes shikimic acid (aromatic amino acids) and Acetyl Coenzyme A, ACOA pathways. Flavonoids have over 9,000 compounds are known and classified according to their numbers of carbon atoms [Turner, 1971, Pandey., et al. 2016, Sarker & Nahar, 2012].
Phenols and flavonoids are classified according to their molecules into free phenols stored in the cell vacuoles, conjugated phenols and structural bound phenols in cell wall through several covalent bonds. Free phenols compounds can be effectively extracted by conventional techniques, while several hydrolysis processes have been used to enhance the release of bound phenols. Fermentation has been considered as one of the best processes to obtain extracts with a high quality and a high activity, using economically and environmental friendly techniques. Extraction of phenols and flavonoids from plants occurs by several methods including physical, physicochemical, and chemical techniques such as organic solvent, ultrasound-assisted, microwave-assisted, cold pressing and supercritical fluid. All these techniques have low yield and it can't release the cell walls bound phenols. Pretreatments by hydrolyzed firstly by enzymes, acids or an alkaline step prior to conventional extraction could be used to maximize the extraction yield but these methods are toxic and have negative effects on the environment. Plant cell walls were degradation by fungal enzymes for release the ponded phenols through fermentation of the basal natural food substrate, the phenols and flavonoids amounts were increased by phenols found naturally in the food substrates (free, conjugated with other molecules and degradative bonded from cell walls) and amount synthesize by fungal pathways or transformed amount by fungal enzymes. Simple phenols, non-benefit flavonoids and complex toxic polyphenols are transformed and converted by fermentation to more bioactive, soluble stabile, detoxified and accessible products by microbial enzymes [Hynh., et al. 2014]
Many authors recorded that the flavonoids have colors and high aromatic value, it also have wide range of applications in many industries such as cosmetic, pharmaceutical, medicine, chemistry and food industry [Sarker & Nahar 2012, De- Carvalho., et al. 2016].
Flavonoids play importance rolls in the living cell life cycles, act as coloring agents, protective layers against pathogens microbes, insect and herbivorous animal, stress of unfavorable environmental conditions (strong light and UV radiation as photoreceptors, low/high temperature, ozone, heavy metals, drought, elicitors or inductor). Also it act as a physiological regulator for enhancement the symbiotic nitrogen fixation and chemical messenger. Flavonoids are providing color, fragrance, the taste of the fruits, flowers, and seeds for pollination and seed transmission, [Percival, 1998, Mierziak., et al. 2014, De-Carvalho., et al. 2016].
Flavonoids in medicine and pharmacology act as natural antioxidants, anticancer, anti-inflammatory, antihypertension, antiobesity, anti-cardiovascular diseases, antidiabites, antiallergic and antimicrobial, it used in prevent and treatments diarrheal and numerous human diseases [Manzoni & Rollini, 2011, Hynh., et al. 2014, Stankovic., et al. 2014].
Flavonoids in cosmetic make to improved skin hydration, smoothen surface and induce growing skin cells, restore its antibacterial barrier, protective, astringent and anti-edema properties. They also used in the treatment of acne, blackheads, dandruff, prevent baldness and wrinkles and slow down the aging processes [Mierziak., et al. 2014].
This investigation has been designed to studies many aims includes primary screening of the yeast bioactive metabolites especially phenols and flavonoids of 80 yeast methanolic extracts by many analytical methods includes spectrophotometer, ammonium test and TLC analysis. Confirmation and estimation the phenols and flavonoids by HPLC analysis and known the phenols fractionation in the highest yeast strain recorded by flowered analytical methods.
Materials and Methods
Collection of yeast samples and cultivations
Eighty yeast isolates and strains were collected from different sources at Assiut Governorate [Eman., et al. 2018a].
Eighty yeast isolates and strains were collected from different sources at Assiut Governorate [Eman., et al. 2018a].
Yeast propagation media
Yeast isolates are re-cultivated on YMEPG medium (Yeast Extract Malt Extract Peptone Glucose) medium. The medium was autoclaved at 121°C for 20 minutes, cooled to approximately 45°C and adjusted to pH 3.7 [Wickerham, 1951].
Yeast isolates are re-cultivated on YMEPG medium (Yeast Extract Malt Extract Peptone Glucose) medium. The medium was autoclaved at 121°C for 20 minutes, cooled to approximately 45°C and adjusted to pH 3.7 [Wickerham, 1951].
Production media
YMEPG medium the amount of glucose here is 100 g/L, this media prepare in two-step the first is preparing all media content except glucose to dissolved in half a liter of dist. Water and then autoclaved at 121°C for 20 minutes. The second is to dissolve glucose in the other half liter of dist. Water then autoclaved at 115°C for 60 minutes. A loop full of yeast inoculum was taken from a pure culture of the yeast isolate grown on slants and inoculated into 50 ml of sterilized propagation media then incubated for 48 hours at 27οC on a shaker with 140 rpm. Take 15 ml of previous yeast propagation media and transfer it into 150 ml production media then incubated for 72 hours at 28 ± 22οC on a shaker. Centrifuge each broth cultures for 15 min at 5000rpm, the cell mass drying on air and weight [Wickerham, 1951].
YMEPG medium the amount of glucose here is 100 g/L, this media prepare in two-step the first is preparing all media content except glucose to dissolved in half a liter of dist. Water and then autoclaved at 121°C for 20 minutes. The second is to dissolve glucose in the other half liter of dist. Water then autoclaved at 115°C for 60 minutes. A loop full of yeast inoculum was taken from a pure culture of the yeast isolate grown on slants and inoculated into 50 ml of sterilized propagation media then incubated for 48 hours at 27οC on a shaker with 140 rpm. Take 15 ml of previous yeast propagation media and transfer it into 150 ml production media then incubated for 72 hours at 28 ± 22οC on a shaker. Centrifuge each broth cultures for 15 min at 5000rpm, the cell mass drying on air and weight [Wickerham, 1951].
Extraction of tested yeast
Centrifugation yeast cultures for obtained their biomass and homogenize with 40 ml methanol 98 pure in a high-speed blender at 16.000 rpm. Leave homogenized mixture in a shaker for overnight then filtrate and concentrated the extract by drying. Mixture was filtered through Whatman filter paper No.2 and dried over anhydrous Na2SO4. The extracts were driedand stored in a dark glass vials for further investigation [Kaur., et al. 2009, Stanković, 2011, Eman, 2012, Lallianrawna., et al. 2013, Bag., et al. 2015].
Centrifugation yeast cultures for obtained their biomass and homogenize with 40 ml methanol 98 pure in a high-speed blender at 16.000 rpm. Leave homogenized mixture in a shaker for overnight then filtrate and concentrated the extract by drying. Mixture was filtered through Whatman filter paper No.2 and dried over anhydrous Na2SO4. The extracts were driedand stored in a dark glass vials for further investigation [Kaur., et al. 2009, Stanković, 2011, Eman, 2012, Lallianrawna., et al. 2013, Bag., et al. 2015].
Primary screening of the flavonoids productivity by 80 yeast methanolic extract by three analytical methods
Sodium hydroxide NaOH test
The extract was treated with a few drops of. Formation of intense yellow color, which becomes color less on addition of few drops of dilute hydrochloric acid, indicates the presence of coumarin andflavonoids [Vimalkumar., et al. 2014].
The extract was treated with a few drops of. Formation of intense yellow color, which becomes color less on addition of few drops of dilute hydrochloric acid, indicates the presence of coumarin andflavonoids [Vimalkumar., et al. 2014].
Ammonia test
One mL from each yeast extract is spotted on filter papers No.3 and expose to concentrated ammonia vapor each spot appeared by distinguished color depend up on the kind of the flavonoids found in the yeast extract and comparing the formed color with the spot of extract on filter paper which not exposed to ammonia vapors and recorded the flavonoids color [Vimalkumar., et al. 2014].
One mL from each yeast extract is spotted on filter papers No.3 and expose to concentrated ammonia vapor each spot appeared by distinguished color depend up on the kind of the flavonoids found in the yeast extract and comparing the formed color with the spot of extract on filter paper which not exposed to ammonia vapors and recorded the flavonoids color [Vimalkumar., et al. 2014].
TLC analysis
Secondary screening of flavonoids by Thin Layer Chromatographic analysis (TLC) and using rutin and quercetin as slandered material. All reagents and chemicals were purchased from Sigma-Aldrich. All solvents were HPLC grade and were used as such.The solvents used were freshly distilled before use. Analytical by TLC was carried out on alumina sheets pre-coated with silica gel, Merk, Kiesel gel 60 F254 60 F254 (5×2cm×0.2mm) solvent system (8:2) and (7:3) Dichloromethane: Methanol spraying with H2SO4
Secondary screening of flavonoids by Thin Layer Chromatographic analysis (TLC) and using rutin and quercetin as slandered material. All reagents and chemicals were purchased from Sigma-Aldrich. All solvents were HPLC grade and were used as such.The solvents used were freshly distilled before use. Analytical by TLC was carried out on alumina sheets pre-coated with silica gel, Merk, Kiesel gel 60 F254 60 F254 (5×2cm×0.2mm) solvent system (8:2) and (7:3) Dichloromethane: Methanol spraying with H2SO4
Spectrophotometric analysis
Total flavonoids in each yeast methanolic extracts was determined with NaOH reagent using qurcetetin and rutin as a standard the absorbance was measured at 362nm versus blank sample on a spectrophotometer and expressed in terms of equivalent (µg/g DW extract). Yeast extracts were dissolved in a known volume of methanol leave for 10 min.Absorbance (AU) reading was made in Triplicate [Mabry., et al. 1970, Lombard., et al. 2002, Pe´rez-Gregorio., et al. 2010].
Total flavonoids in each yeast methanolic extracts was determined with NaOH reagent using qurcetetin and rutin as a standard the absorbance was measured at 362nm versus blank sample on a spectrophotometer and expressed in terms of equivalent (µg/g DW extract). Yeast extracts were dissolved in a known volume of methanol leave for 10 min.Absorbance (AU) reading was made in Triplicate [Mabry., et al. 1970, Lombard., et al. 2002, Pe´rez-Gregorio., et al. 2010].
HPLC analysis
HPLC used for estimation the phenols contents [Lombard., et al. 2002, Stanković, 2011] were used. In this study the highest flavonoids recorded by the flowered analytical methods were confirmed by HPLC. Five yeast methanolic extracts were selected and performed by Agilent HPLC analysis, model 6890 N/5975B (Agilent Technologies, Palo Alto, CA, USA) at the analytical Chemistry Unit, ACAL, Chemistry Department, Faculty of Science, Assiut University, Assiut, Egypt.
HPLC used for estimation the phenols contents [Lombard., et al. 2002, Stanković, 2011] were used. In this study the highest flavonoids recorded by the flowered analytical methods were confirmed by HPLC. Five yeast methanolic extracts were selected and performed by Agilent HPLC analysis, model 6890 N/5975B (Agilent Technologies, Palo Alto, CA, USA) at the analytical Chemistry Unit, ACAL, Chemistry Department, Faculty of Science, Assiut University, Assiut, Egypt.
GC/MS Analysis
Chemical profile of the phenols found in yeast strain Diutina rugosa MH333102, UMC13566 methanolic extract of the highest flavonoids and antibacterial bioactive was estimated by GC/MS analysis. Apparatus:GC-MS (7890A-5975B); Column: DB-5ms; GC-Conditions. Oven program: 40°C for 2 min; then 10°C/min to 150°C for 3 min; then 10°C/min to 220°C for 6 min; then 15°C/min to 280°C for 28 min; Run Time 61 min and 2 min (Post Run) 260°C. Flow program: 0.5 mL/min for 10.9 min; then 1 mL/min per min to 1 mL/min for 30 min.
Chemical profile of the phenols found in yeast strain Diutina rugosa MH333102, UMC13566 methanolic extract of the highest flavonoids and antibacterial bioactive was estimated by GC/MS analysis. Apparatus:GC-MS (7890A-5975B); Column: DB-5ms; GC-Conditions. Oven program: 40°C for 2 min; then 10°C/min to 150°C for 3 min; then 10°C/min to 220°C for 6 min; then 15°C/min to 280°C for 28 min; Run Time 61 min and 2 min (Post Run) 260°C. Flow program: 0.5 mL/min for 10.9 min; then 1 mL/min per min to 1 mL/min for 30 min.
Statistical analysis
All experimental measurements were carried out in triplicate and are expressed as the average of three analyses ± standard deviation.
All experimental measurements were carried out in triplicate and are expressed as the average of three analyses ± standard deviation.
Results and Discussion
Yeast flavonoids screened by many analytical methods
Results showing that the primary screening of the yeast ability to produce of phenols and flavonoids by 80 yeast methanolic extract were tested by detected by ammonium test, TLC, and spectrophotometer at 362 nm [Table 1 & 2 and Figure 1 & 2a-c]. Secondary screening was applied by the same methods on the highest yeast producers [Table 2 & Figure 3]. The results were confirmed by HPLC and GC/MS analysis [Table 3 & Figure 4a-g & 5].
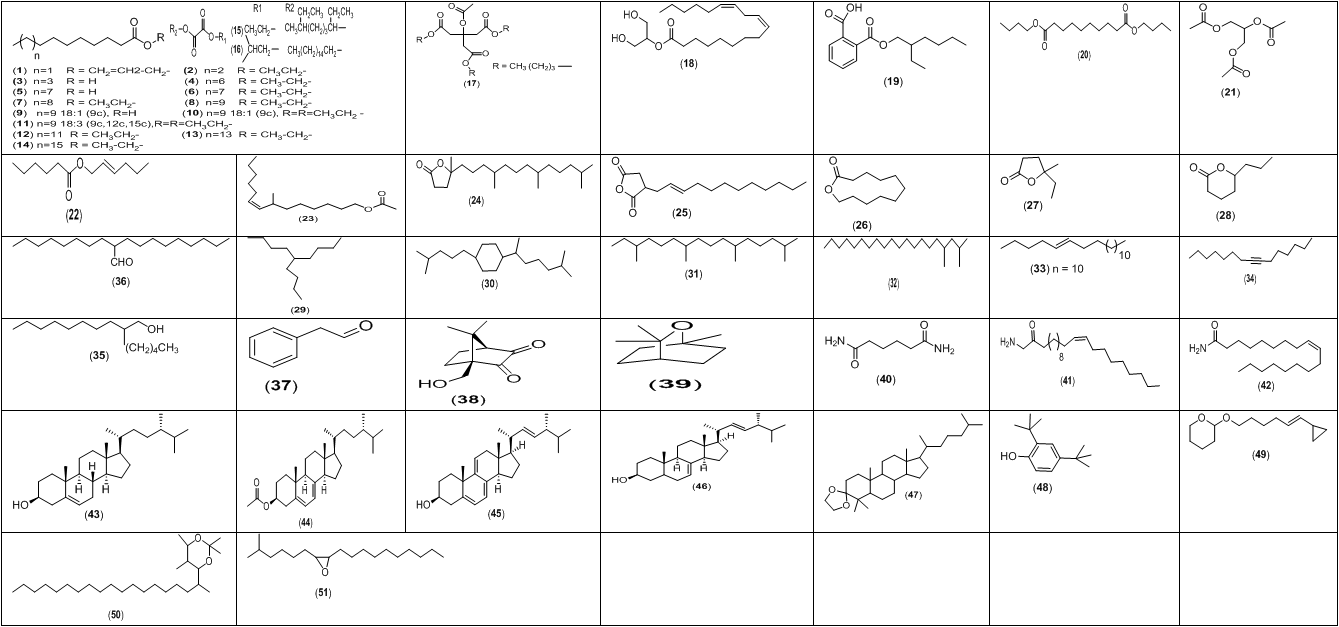
Figure 1: Chemical structures of the main aroma metabolites detected in ethanolic extract of P. ostreatus by GC/ MS analysis.
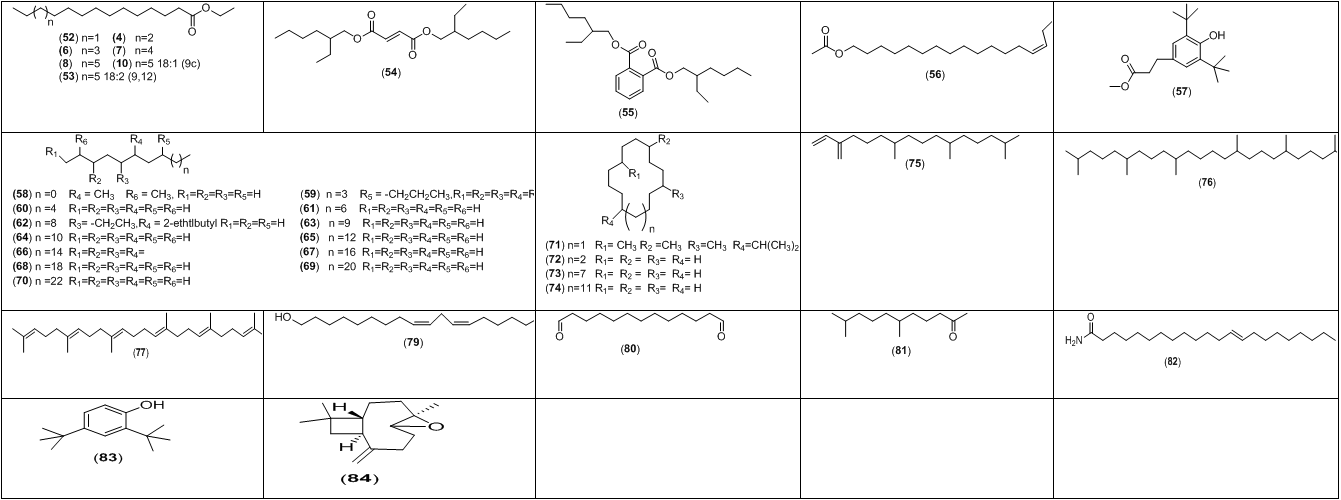
Figure 2: Chemical structures of the main aroma metabolites detected in ethanolic extract of P. ostreatus spent.
| Detected by aroma metabolites | Basidium | Detected by aroma metabolites | Basidium |
| 9,12-octadecadienoic [Cis-linoleic] Fatty acid | 29.19 | butyl-citrate Ester | 0.53 |
| 9,12-octadecadienoic acid-2-hydroxy-1-(hydroxymethyl)ethyl ester Ester | 5.04 | 6-tetradecanesulfonic acid, butyl ester Ester | 0.50 |
| hexadecanoic acid, ethyl ester Ester | 3.34 | tetradecanoic [myristic acid] Fatty acid | 0.32 |
| didecyl phthalate Ester | 1.68 | 2,2-dideutero-heptadecanalAldehyde | 0.21 |
| 2-ethylhexyl methacrylate Ester | 1.53 | cyclohexanecarboxylic acid,2,2-dimethyl propyl ester Ester | 0.30 |
| 3,4,6,7-tetramethylidenebicyclo [3.2.1] octan-2-exo-olAlcohol | 1.50 | 1-heneicosyl formate Ester | 0.18 |
| ergost-5,8(14)-dien-3-ol Sterol | 1.48 | l-proline,N-ethoxycarbonyl-,butyl ester Ester | 0.10 |
| Dihexylphthalate Ester | 1.41 | sulfurous acid, hexyl octyl ester Ester | 0.10 |
| pentadecanoic acid Fatty acid | 1.36 | 1R-4-acetamido-2,3-cis-epoxy-cyclohexanolAlcohol | 0.10 |
| N-1, N-1-dimethyl-N-2-N-pentylformamidineAmidine | 1.21 | 7-hexyl- docosane Ketone | 0.09 |
| proopyl-trans-4- [trans-4-(trans-4- propylcyclohexyl) cyclohexyl] cyclo hexanecarboxylate Ester | 1.16 | Undecane Alkane | 0.08 |
| 1,2-benzenedicarboxylic acid, butyl-8-methyl nonyl ester Ester | 0.41 | Octadecane Ketone | 0.08 |
| hexadecanoic acid,2-hydroxy-1- (hydroxyl methyl) ethyl ester Estersr | 0.39 | Heptadecane Ketone | 0.08 |
| Hexatriacontane Ketone | 0.35 | 1,2-benzenedicarboxylic acid, bis(2-methyl propyl) ester Ester | 0.06 |
| Dodecane Ketone | 0.25 | 1,2-benzenedicarboxylic acid,dibutylester Ester | 0.06 |
| 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi-1,3-dioxolane Ketone | 0.24 | methyl-N-hydroxybenzenecarboximidoate Ester | 0.06 |
| 1,2-benzenedicarboxylic acid, ditridecylester Ester | 0.23 | sulfurous acid, butyl dodecyl ester Ester | 0.05 |
| Pentadecane Ketone | 0.22 | methyl-2,8-dimethylundecanoate Ester | 0.05 |
| 3-ethyltetracosaneAlkane | 0.19 | pelargonic [nonanoic acid] Fatty acid | 0.04 |
| tridecane Ketone | 0.19 | benzeneacetic acid Acid | 0.03 |
| 9-octylheptadecaneKetone | 0.18 | D-xenialactol Sterol | 0.02 |
| 4,5,6,7,8,8,9,10-octahydro-1,3,4-trimethoxy-4b,8,8-trimethyl-2-(1-methylethyl)-9-phenanthrenolAlcohol | 0.16 | 5-pentyl-1, 3-benzenediolAlcohol | 0.02 |
| butanoic acid, -ethyl-ester Ester | 0.17 | N-methylmaleamic acidAcid | 0.02 |
| Triacontane Ketone | 0.14 | 7,9-di-tert-butyl-1-oxaspiro [4.5] deca-6, 9-diene-2, 8-dione Ketone | 0.01 |
| Tetratriacontane Ketone | 0.13 | 5-(acetyloxy)- 2-pentanone Ketone | 0.01 |
| 2,13-octadecadien-1-ol Alcohol | 0.13 |
Table 1: IUPAC name and mass fractions of aroma metabolites detected in P. ostreatus basidium recorded by GC/MS analysis according to (Eman and Farghaly 2014).
| Detected by aroma Metabolites | Spent | Detected by aroma Metabolites | Spent |
| 2-butenedioic acid -, bis(2-ethylhexyl)-ester (54) Ester | 36 | Eicosane (69) Ketone | 0.7 |
| ethyl-tetradecananoate (52) Ester | 6.1 | Cycloeicosane (73) Ketone | 0.7 |
| 1,2-benzenedicarboxylic acid bis (2-ethyl hexyl)ester (55) Ester | 4.6 | 2,6,10-trimethyl,14-ethylene-14-pentadecne (75) Ketone | 0.7 |
| 11,15-tetramethyl-2-hexadecen-1-ol. (78) Alcohol | 3.7 | 2,6,10,15,19,23-hexamethyl-tetracosane (76) Alkan | 0.7 |
| 9,12-octadecadienoic-ethyl-ester(53) Ester | 2.8 | 2,6,10,14,18,22-tetracosahexaene (77) Alkan | 0.4 |
| cyclotetracosane(72) Ketone | 2.4 | 9,12-octadecadien-1-ol. (79) Alcohol | 0.4 |
| cyclopentadecane(74) Ketone | 2.4 | benzenepropanoic acid,3,5-bis (1,1-dimethyl ethyl)-4-hydroxy-,methyl-ester(57) Ester | 0.3 |
| 6,10,14-trimethyl-2-pentadecanone(81)Ketone | 1.9 | tridecan-edial(80)Aldehyde | 0.3 |
| 3,8-dimethyl-decane(61) Ketone | 1.1 | 14-octadecen-1-ol-acetate(56)Ester | 0.3 |
| 3,8-dimethyl-decane(61) Ketone | 1.1 | 1,7,11-trimethyl-4-(1-methylethyl)-cyclotetradecane(71) Ketone | 0.3 |
| tetradecane(63) Ketone | 1.0 | triacontane(58) Ketone | 0.1 |
| 6-propyltridecane(62)Alkane | 1.0 | dotriacontane(59) Ketone | 0.1 |
| 13-docosen-amide(82)Amide | 0.8 | caryophylleneoxide(83)Sterol | 0.1 |
| 3-ethyl-5-(2-ethylbutyl)octadecane(67) Ketone | 0.7 |
Table 2: IUPAC name and mass fractions of aroma metabolites detected in P. ostreatus spent recorded by GC/MS analysis.
| Detected by aroma metabolites | Substrate | Basidium | Detected by aroma metabolites | Substrate | Spent | |
| ergosta-5,7,22-trien-3β-ol(44) Sterol | 1.7 | 47.21 | 2,4-bis(1,1-dimethylethyl) phenol (48) Phenol | 1.3 | 1.3 | |
| Linoleic-ethyl-ester (11) Ester | 11.0 | 18.53 | ethyloctadecanoate(8) Ester | 1.2 | 0.2 | |
| hexadecanoic [palmitic acid] (5) Fatty acid | 2.4 | 6.98 | ethylheptadecanoate(7) Ester | 0.2 | 0.3 | |
| 9-octadecenamide [cis-linoleic amide](40) Amide | 1.6 | 6.21 | Detected metabolites by GC/MS | Substrate | Basidium | Spent |
| dodecanoic [lauric acid] (3) Fatty acid | 0.3 | 0.21 | Ethylpentadecanoate (4) Ester | 1.5 | 0.4 | 1.3 |
| 5-eicosene (33) Ketone | 0.8 | 0.1 | Ethylhexadecanoate (6) Ester | 11.1 | 3.3 | 6.1 |
| 9-octadecenoic [Oleic] (9) Fatty acid | 0.06 | 0.1 | Ethyloleate (10) Ester | 0.2 | 1.0 | 2.9 |
| ergosta-5,7,9(11),22-tetraen-3β-ol(45) Sterol | 1.0 | 1.0 | Legends IUPAC (chemical structure of the metabolites by recorded GC/MS analysis and drawing by Khallaf) Classified according to the bioactive chemical groups and mass fraction. |
|||
| Detected by aroma metabolites | Basidium | Spent | ||||
| hexadecane (70) Ketone | 0.4 | 0.8 | ||||
| Nonadecane (68 ) Ketone | 0.1 | 0.7 | ||||
| docosane(66) Ketone | 0.3 | 0.6 | ||||
| heptacosane(65) Ketone | 0.4 | 0.5 | ||||
| tetracosane(64) Ketone | 0.1 | 0.5 | ||||
| octacosane(60) Ketone | 0.1 | 0.3 | ||||
Table 3: Comparative studding of the IUPAC name and mass fractions of aroma metabolites found and detected in both samples of (substrates and basidium), (basidium and spent), (substrate and spent) and (substrate, basidium and spent) in P. ostreatus recorded by GC/MS analysis.
Simple reagent test by alkaline reagent test (sodium hydroxide NaOH test)
The extract was treated with a few drops of NaOH formation of intense yellow color, which becomes colorless on the addition of few drops of dilute hydrochloric acid, indicates the presence of coumarin andflavonoids by various degrees of yellow colors
The extract was treated with a few drops of NaOH formation of intense yellow color, which becomes colorless on the addition of few drops of dilute hydrochloric acid, indicates the presence of coumarin andflavonoids by various degrees of yellow colors
Spectrophotometric analysis
A total flavonoids in 80 yeast methanolic extracts was determined with NaOH reagent using qurcetetin and rutin as a standard, the absorbance was measured by spectrophotometer at 362 nm, the flavonoids were determined by (µg/g DW). Screening results clearing that the yeast flavonoids fluctuated between total flavonoids fluctuated between 14.9±1 to 105.8±4.3 µg/g DW and classified into three categories includes ≥51, 50.9-31and ≤30.9 µg/g DW are 20, 24 and 36 were high, moderate and low, respectively [Table 1-3 and Figure 1 & 2a-c].
A total flavonoids in 80 yeast methanolic extracts was determined with NaOH reagent using qurcetetin and rutin as a standard, the absorbance was measured by spectrophotometer at 362 nm, the flavonoids were determined by (µg/g DW). Screening results clearing that the yeast flavonoids fluctuated between total flavonoids fluctuated between 14.9±1 to 105.8±4.3 µg/g DW and classified into three categories includes ≥51, 50.9-31and ≤30.9 µg/g DW are 20, 24 and 36 were high, moderate and low, respectively [Table 1-3 and Figure 1 & 2a-c].
Ammonia test
Screening the production of flavonoids by 80 yeast methanolic extract were employed by ammonia test and spreayed the spot by ammonia vapor each spot appear by distinguished color with a yellow color degree which flactoweted between faintly yellow, deep yellow, orange, yellow-green and brown [Table 3 and Figure 3].
Screening the production of flavonoids by 80 yeast methanolic extract were employed by ammonia test and spreayed the spot by ammonia vapor each spot appear by distinguished color with a yellow color degree which flactoweted between faintly yellow, deep yellow, orange, yellow-green and brown [Table 3 and Figure 3].
TLC analysis
Select the most flavonoids producers are reconfirmed many times and clear that the recorded results found on TLC plate described in
HPLC AnalysisSelect the most flavonoids producers are reconfirmed many times and clear that the recorded results found on TLC plate described in
The five highest total phenols and flavonoids producers yeast methanolic extract were confirmed by HPLC and recorded that Gallic 100-23500 µg/L, rutin and quercetin are recorded 1-50 µg/L [Table 3 and Figure 4a-g].
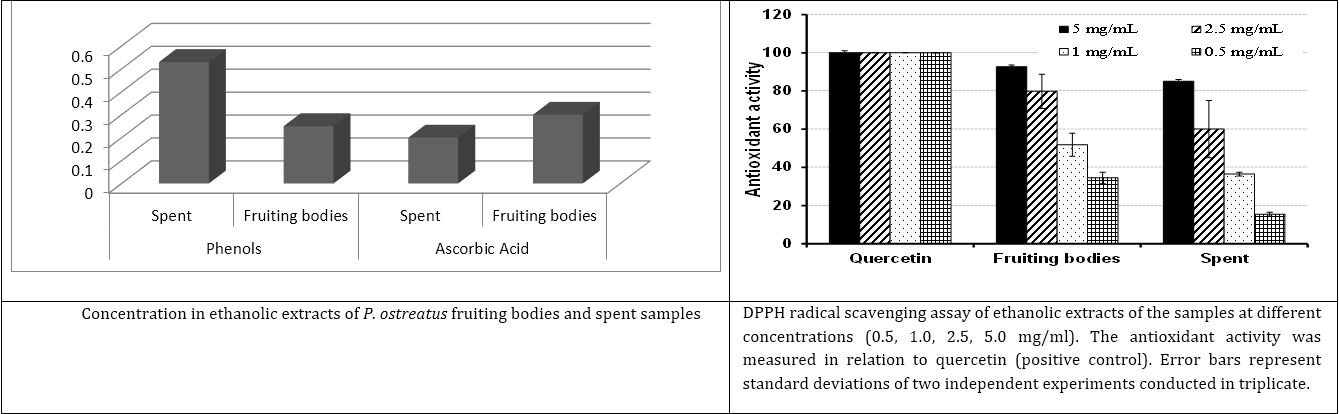
Figure 4: Total phenols (μg/100g DW) and ascorbic acids (g/100g DW) recorded
as antioxidant metabolites in P. ostreatus fruiting bodies and spent samples.
Yeast antibacterial activity
The relationships between phenols and bioassay test un-cleared may be the antibacterial bioactive metabolites in the yeast extracts depend upon other metabolites other than the phenols and flavonoids. Each yeast kinds have metabolic profile completely differ from sample to another'saccording to [Eman., et al. 2018b].
The relationships between phenols and bioassay test un-cleared may be the antibacterial bioactive metabolites in the yeast extracts depend upon other metabolites other than the phenols and flavonoids. Each yeast kinds have metabolic profile completely differ from sample to another'saccording to [Eman., et al. 2018b].
Numerous studies and Authors recorded and clearing that the phenols and flavonoids are synthesis and distributed in all members and groups of the plant kingdom and it includes higher plants, algae, mushrooms, bacteria, filamentous fungi and yeast. Many kinds of phenols and flavonoids were produced by many bacterial genus includes Bacillus pumilus produced Gallic acid, catechin, epicatechin. B. subtilisproducedchlorogenic acid, naringin, Daidzein, genistein. Lactobacillus acidophilus produced gallic acid. L. johnsonii, L. reuteri, produced phenols & flavonoids. L. acidophilus produced sinapic, caffeic, p-coumaric & ferulic acids. L. plantarum produced quercetin. L. delbrueckii supsp. lactis produced daidzein, genistein [Huynh., et al. 2014].
Yeast Saccharomyces cerevisiae was studied and recorded phenols contents ranged between 234-317mg/L [Li., et al. 2017], and riches by flavonoids [Du., et al. 2011, Kumar & Pandey, 2013, Bartosz & Bartosz, 2014], and syringic acid, p-coumaric acid, ferulic acid recorded by [Huynh., et al. 2014]. Other genus of yeast are studied and recorded Cryptococcus flavus, R. glutamic, Wickerhamomyces anomalous were producedphenols content 0.2-0.6 mg/L and flavonoids, which act as anti-aging metabolites such as ascorbic acid "vitamin E or vitamin C", 𝛼-tocopherol and CoQalone [Coghe., et al. 2004, Moore., et al. 2007, Restucci., et al. 2011].
Entophytic filamentous fungi produced phenols and flavonoids [De-Carvalho., et al. 2016]. [Huynh., et al. 2014] recorded the production of phenols and flavonoids by fungal fermentations such as Aspergillus oryzae produced daidzein, genistein, Gallic, gallocatechin, epigallocatechin, epicatechin, 3-p-coumaroyl quinic acid and kaempferol-rutinoside. Monascus purpureus produced daidzein and genistein. Aspergillus oryzae var. effuses produced chlorogenic, ferulic, p-coumaric and caffeic acid. Aspergillus niger producedchlorogenic, ferulic, coumaric and caffeic acids. Rhizopus oryzae produced ferulic, hydroxybenzoic, caffeic, chlorogenic and vanillin. Rhizopus oligosporus produced daidzein and genistein.
Many Authors reported that the edible mushrooms have high contents of phenols and flavonoids. Mushrooms have 0.40 mg/g phenols [Gezer., et al. 2006], 130 mushrooms studied and recorded as a phenols and flavonoids production [Ferreira., et al. 2009], Lentinus edodes producedellagic acid [Huynh., et al. 2014], different genus edible produced 0.40-2.21 mg/g phenols range [Rashidi & Yang, 2016], Pleurotus ostreatus produced phenols and flavonoids [Eman ., et al. 2018c].
Numerous studies are detected and estimated phenols and flavonoids by higher plants, [De-Carvalho., et al. 2016] includes 39 medicinal & culinary herbs produced 0.23-17.5 mg/g FW or 0.1- 4.4% [Zheng & Wang, 2001], Six traditional medicine plants produced phenols 14-50 & flavonoids15.7-68.0 mg/g [Jara., et al. 2013] and 66 medicine plants produced 1.8-166.3 mg/g phenols [Maslennikov., et al. 2014]. 3.8, GC/MS Analysis
| Tested Samples | Dine | Phenol | Acid | Aldehyde | Amide | Alkane | Sterol | Alcohol | Fatty acids | ketones | Esters | Total No. | References |
| Total detected metabolites | 1 | 1 | 2 | 2 | 2 | 5 | 5 | 7 | 7 | 33 | 33 | 98 | In this investigation & in Eman & Farghaly2014 |
| Basidium | 1 | - | 2 | 1 | - | 2 | 2 | 5 | 4 | 13 | 21 | 51 | Eman & Farghaly2014 |
| Spent | - | - | - | 1 | 1 | 3 | 1 | 2 | - | 13 | 6 | 27 | In this investigation |
| Substrate & basidium | - | - | - | - | 1 | - | 2 | - | 3 | 1 | 1 | 8 | In this investigation & in Eman & Farghaly2014 |
| Basidium & spent | - | - | - | - | - | - | - | - | - | 6 | - | 6 | In this investigation & in Eman & Farghaly2014 |
| Spent & substrates | - | 1 | - | - | - | - | - | - | - | - | 2 | 3 | In this investigation |
| Substrate, basidium & spent | - | - | - | - | - | - | - | - | - | - | 3 | 3 | In this investigation & in Eman & Farghaly2014 |
Table 4: Summarized the results recorded in tables 1-3.
| Tested Mushrooms | Tested samples | Total phenols | References |
| Pleurotus ostreatus | Spent | 0.53µg/100g | In this investigation |
| P. ostreatus | Fruiting bodies | 0.25µg/100g | In this investigation |
| P. ostreatus | Fruiting bodies | 0.44 | Rashidi & Yang, 2016 |
| Coriolis versicolor | Fruiting bodies | 23.28 | Mau., et al. 2002 |
| Ganoderma lucidum | Fruiting bodies | 47.25 | Mau., et al. 2002 |
| G. lucidum antler | Fruiting bodies | 55.96 | Mau., et al. 2002 |
| G. tsugae | Fruiting bodies | 51.28 | Mau., et al. 2002 |
| P. ostreatus | Fruiting bodies | 0.39 | Rashidi & Yang, 2016 |
| P. sajor-caju | Fruiting bodies | 2.21 | Rashidi & Yang, 2016 |
| Agaricus bisporus | Fruiting bodies | 0.63 | Rashidi & Yang, 2016 |
| Hypzigus marmoreus | Fruiting bodies | 0.67 | Rashidi & Yang, 2016 |
| Volvariella volvacea | Fruiting bodies | 0.73 | Rashidi & Yang, 2016 |
| Flammulina velutipes | Fruiting bodies | 0.75 | Rashidi & Yang, 2016 |
| Pleurotus eryngii | Fruiting bodies | 0.44 | Rashidi & Yang, 2016 |
| Hericium erinaceus | Fruiting bodies | 0.46 | Rashidi & Yang, 2016 |
| Lentinula edodes | Fruiting bodies | 0.46 | Rashidi & Yang, 2016 |
| Ramaria flava | Fruiting bodies | 0.40 | Gezer., et al. 2006 |
| Agaricus bisporus | Fruiting bodies | 0.40 | Nagy., et al. 2017 |
| Pleurotus ostreatus ascorbic acid = 0.3±0.01 g/100g DW in fruiting bodies and = 0.2±0.1 g/100g DW in spent | |||
Table 5: Ascorbic acids and total phenols recorded as antioxidant metabolites in P. ostreatus fruiting bodies and spent samples comparative with the results recorded by many Author's.
GC/MS (analysis showing that the all information’s (common name, CID, GC/MS % & retention time, related to phenols classes, IUPAC name, molecular formula and molecular weight (g/mol), bioactivity and chemical structure, according to Pub Chem citation) about ten phenols and flavonoids detected by different analytical methods in methanolic extract of the highest flavonoids contents Diutina rugosa MH333102 strain, especially seven phenols fractions were detected by GC/MS included (pyrocatechol; resorcinol; α.-methyl-α-propylcyclopropanemethanol; aloxiprin; 3,4,7,7-Tetrahydro-3-Methyl-2(3H)-benzo-furanone; p-cumenol and butaxamine, were 6.4, 6.4, 1.7, 1.4, 0.9, 0.9, and 0.02%, respectively
| Methanolic Extracts mg/ml | 5 | 2.5 | 1 | 0.5 | References |
| Standard quercetin | 100±1 | 100±2 | 100±1 | 100 ±3 | In this investigation |
| Fruiting bodies | 93±1 | 80 ± 9 | 52 ± 6 | 34 ± 3 | |
| Spent | 85±8 | 60 ± 15 | 36 ± 1 | 15 ± 1 | |
| Other mushrooms tested by different solvent | µg/mg-1 | Standard | Antioxidant% DPPH assay | At µg/ml | |
| Ramaria Flava (Schaeff) Quél | 94.7 | 160 | Gezer., et al. 2006 | ||
| 39.83 | Pyrocatechol | ||||
| 8.27 | Quercetin | 98.9 | 160 | ||
| Tocopherol | 99.2% | 160 | |||
| Trametes versicolor Acetone extract | 50.9 | 54.9 | 500 | Kamiyama., et al. 2013 | |
| T. versicolor Methanol extract | 33.9 | 40.2 | 500 | ||
| T. versicolor n-hexane extract | 29.5 | 29.5 | 500 | ||
| T. versicolor Chloroform extract | 15.2 | 15.2 | 500 | ||
| Black & red ear mushroom Methanol extract | 100 | 0.1 | |||
| Phellinus Quél Ethanol extract | High | ||||
| Coprinus comatus | 47.0 | 20 | |||
| Pleurotus sajor-caju Hot water extract | 89.29 | 1000 | Rashidi & Yang, 2016 |
Table 6: The antioxidant bioactivity in P. ostreatus fruiting bodies, spent samples and flavonoids (quercetin positive control) as a standard material tested by using DPPH radical scavenging assay in this investigation and many references. Error bars represent standard deviations of two independent experiments conducted in triplicate.
The recorded metabolites included catechol used in the production of the photographic developer, perfumes, pharmaceuticals agents such as urushiols for treatment the skin-irritation, catecholamines or phenethylamine hormones which act as neurotransmitters; pesticides, flavors, and fragrances includes vanillin (methylated catechol) using as (catechol-monoethyl-ether or guethol, is converted to ethylvanillin, a component of chocolate confectioneries. 3-Trans-isocamphylcyclo hexanol. Piperonal, a flowery scent, is prepared from the methylene diether of catechol followed by condensation with glyoxal and decarboxylation. Α.-methyl-α-propyl-cyclo-propane-methanol. Resorcinol is natural phenols, 2% solution used as a spray has been used with marked effect in hay fever and in a whooping cough. In the latter disease, 0.6 mL of the 2% solution has been given internally. It can be included as an anti-dandruff agent in shampoo or in sunscreen cosmetics. It has also been employed in the treatment of gastric ulcers in doses of 125 to 250 mg in pills and is said to be analgesic and hemostatic in its action. In large doses, it is a poison, causing giddiness, deafness, salivation, sweating, and convulsions, medicated soaps (parasol). Aloxiprin formed from a dimer of aspirin and act as anti-inflammatory, antipyretic and analgesic drug. It is a chemical compound of aluminum hydroxide and aspirin. Aspirin or acetylsalicylic acid act as antipain, antifever, inflammation. anti-ischaemic strokes, and anti-blood clots anti-colorectal and anticancer. Cuminol or (4-(1-hydroxy-1-methylethyl)-phenol)is phenols derived by hydroxylation from p-Cymene (1-Methyl-4-(propane-2-yr) benzeneor alkylbenzene) related to a monoterpene.
Many Authors studied the production of many kinds of phenols in wine during fermentation such as catechin10–126μg/L, gallic acid 11.92–53.01 mg/kg, protocatechuic acid 75.12–179.03 [Coghe., et al. 2004, Moore., et al. 2007, Restucci., et al. 2011, Alkan., et al. 2017, Huynh., et al. 2014].
[De Carvalho., et al. 2016] reviewed and reported the production of phenols by many entophytic fungi includes alterperylenol act as antibacterial; vincristine; p-chlorocinnamide, isocoumarin, chlorogenic acid (5-O-caffeoylquinic acid) which act as antioxidant and has inhibitory effect on herbivores and other pathogens, improving the resistance of the plants;macrosporin and 3-Omethylalaternin which act as anti-inflammatory, cytotoxic and antimicrobial; desmethyldiaportinol and altersolanol A act as cytotoxic and antimicrobial; capsaicin act as anti-tumors, anti-strong pain;luteolin act as antioxidant; excelsione act as anti-leukemia and antimicrobial;cytosporones B & C, phomopsin A-C act as antimicrobial dicerandrol C and mycophenolic acid and (E)-7-(2-hydroxy-4-(hydroxymethyl)phenyl)-2-methyloct-6-enoic acid; cytosporone C and dothiorelone B, together with seven new compounds act as cytotoxic; (2,4,7-trioxa-bicyclo and heptan-3-yl) phenol act as antimicrobial and antifungal activities against different human pathogens; pestalol A-E, 4-hydroxyphenethyl, 2-4hydroxy phenyl) acetate and p-Hydroxyphenethyl aceitc acid methyl ester act as antihuman cancer cell lines, anti-influenza virus and anti-tuberculosis; phomodione, usnic acid and cercosporamida act as antifungal and antibacterial; tyrosol act as antioxidant; tyrosol act as antihuman pathogen; tyrosol and diphenyl ether 2,4-dihydroxy-2’,6-diacetoxy-3’-methoxy-5’-methyl-diphenyl ether act as anticancer and antifungal and nigrosphaerin A, isochromene derivative, methyl-4-hydroxybenzoate and tyrosol.
Chemical profile of the phenolic compounds produced by six commercial Saccharomyces cerevisiae strains through the fermentation of Kiwifruit detected by HPLC and recorded that caffeic acid 1.1-3.0, caftaric acid 0.3-1.0, catechin 0.2-0.41, gallic acid 0.17-0.4, chlorogenic acid 1.0, coumaric acid 0.1, ellagic acid 0.3-1.0, epicatechin 0.8-2.0, ferulic acid 1.0-1.1, protocatechuic acid 0.14-1.0, proanthocyanidins B2 0.21-0.5 and total phenols 234-317 mg/L [Li., et al. 2017].
Conclusions
Yeast act as natural sources of phenols and flavonoids which have antibacterial, antioxidant and other many bioactive activities. Yeast is promising and interesting for academic research as well as for industry, medicine, and natural pharmacology. Chemical profile of phenols and flavonoids differ from yeast to another depend up on the genus, species, the source of isolation, substrate or fermentation medium and methods of detection. Yeast needs further investigations to discovering their secrets. There are no interests to declare 'Declarations of interest: none'.
References
- Akalın H., et al. “Determination of some individual phenolic compounds and antioxidant capacity of mead produced from different types of honey”. Journal of the Institute of Brewing 123(2017): 167-174.
- Bag, GC. et al. “Assessment of total flavonoid content and antioxidant activity of methanolic rhizome extract of three Hedychium species of Manipur Valley”. International Journal of Pharmaceutical Sciences Review and Research30.1(2015): 154-159 .
- Bartosz, IS., et al. “Effect of antioxidants supplementation on aging and longevity”. Research International. Review Article (2014): 1-17.
- Chan GF., et al. “Genome sequence of Pichia kudriavzevii M12, a potential producer of bio-ethanol and phytase”. Eukaryot Cell11.10(2012): 1300-1301.
- Coghe S., et al. “Ferulic acid release and 4-vinyl guaiacol formation during brewing and fermentation: Indications for feruloyl esterase activity in Saccharomyces cerevisiae”. Journal of Agricultural and Food Chemistry 52 (2004): 602-608.
- Daniel HM., et al. “Yeast diversity of Ghanaian cocoa bean heap fermentations”. FEMS Yeast Research 9 (2009): 774-78.
- De-Carvalho., et al. “Importance and implications of the production of phenolic secondary metabolites by endophytic fungi: A Mini-Review”. Medicinal Chemistry(2016): 259-271.
- De-Oca., et al. “description, and structure. Chapter 2”. Universidad Autónoma Del Estado de México (2016).
- De Vuyst L., et al. “The sourdough microflora: biodiversity and metabolic interactions”. Trends in Food Science & Technology(2016):43-56.
- Du F., et al. “Advances in the microbial heterologous production of flavonoids”.African Journal Microbiology Research 5.18(2011): 2566-2574.
- Eman MM., et al. “Chemical profile, agaritine, and selenium content”. Brazilian Archives of Biology and Technology 55.6(2011): 911-920 .
- Eman MM., et al. “Effect source, media and techniques" of isolation on kind of isolated yeast and molecular technique identification”. In press (2018).
- Eman, MM., et al. “Antibacterial activity of yeasts, which may act as a novel source of antimicrobial agents and GC/MS analysis of Rhodotorula mucilaginosa AUMC13565. Journal of General and applied Microbiology (2018).
- Eman MM., et al. “Aroma and antioxidant metabolites in Pleurotus ostreatus mushroom detected by GC/MS analysis, and antioxidant bioactivty confirmed by DPPH free radical scavenging assay. Food Chemistry (2018).
- Ferreira IC., et al. “Antioxidants in wild mushrooms Article Literature Review”. Current Medicinal Chemistry 16.12(2009): 543-1560 .
- Gezer K., et al. “Free radical scavenging capacity and antimicrobial of wild mushrooms from Turkey”. African Journal of Biotechnology 5.20(2006): 1924-1928.
- Huynh NT., et al. “Improved release and metabolism of flavonoids by steered fermentation processes: A Review” International Journal of Molecular Sciences 15(2014): 19369-19388.
- Jara-Palacios MJ., et al. “Study of Zalema grape pomace: Phenolic composition and biological effects in Caenorhabditis elegans.”Journal Agricultural & Food Chemistry 61 (2013): 5114-5121.
- Kaur B., et al. “Production and evaluation of physicochemical properties of red pigment from Monascus purpureus MTCC 410.” Internet Journal Microbiology 7(2009): 1-7.
- Kumar S., et al. “Chemistry and biological activities of flavonoids: an overview Review Article”. Scientific World Journal(2013): 1-16.
- Kurtzman CP., et al. “The yeasts, a taxonomic study. 1. 5Th edition”. Elsevier (2011).
- Lallianrawna S., et al. “Determination of total phenolic content, total flavonoid content and total antioxidant capacity of Ageratina adenophora (Spreng). King & H. Rob. Science Vision, 13.4(2013): 149-156.
- Li S., et al. “Effects of six commercial Saccharomyces cerevisiae strains on phenolic attributes, antioxidant activity, and aroma of Kiwifruit (Actinidia deliciosa cv.) wine. Research Article”. Bio-Med Research International (2017): 1-10.
- Lombard KA., et al. “Flavonoids quantification in the extract by spectrophotometric and high-performance liquid chromatography analysis”. Hortscienc37(2012): 682-685.
- Mabry TJ., et al. “The systematic identification of flavonoids. Springer Verlag, New York (1970).
- Maslennikov PV., et al. “The content of phenolic compounds in medicinal plants of a botanical garden (Kaliningrad Oblast)”. Akademii Nauk, Seriya Biologicheskaya 5(2013): 551-557.
- Manzoni M., et al. “Enhancement of Saccharomyces cerevisiae glutathione and micronutrients content for nutraceuticals applications. Graduate School in Biochemical. Nutritional and Metabolic Sciences PhD Course in Experimental and Clinical Nutrition XXIV Cycle University À DeguI Student di Milano. Facoltà di Agrari (2011).
- Mierziak J., et al. “Flavonoids as important molecules of plant interactions with the environment. Review”. Molecules19(2014): 16240-16265.
- Moore J., et al. “Effects of solid-state yeast treatment on the antioxidant properties and protein and fiber compositions of common hard wheat bran”. Journal Agricultural Food Chemistry55(2011): 10173-10182.
- N’guessan K.F., et al. “Identification of yeasts during alcoholic fermentation of tchapalo, a traditional sorghum beer from Côte d’Ivoire, Antonie van Leeuwenhoek 99.4(2011): 855-864.
- Pandey RP., et al. “Microbial production of natural and non-natural flavonoids: Pathway engineering, directed evolution, and systems/synthetic biology”. Research Biotechnology Advances 34.5(2016): 634-662.
- Percival M. “Antioxidants. Clinical Nutritional Insights Nut, 031, 1/96 Rev. 10/98 Copyright 1996. Advanced Nutrition Publications, Inc., Revised (1998):1-4.
- Pham T., et al. “Evaluation of its PCR, RFLP for differentiation, identification of brewing yeast and brewery ‘Wild’ Yeast Contaminants”. Journal of Instusterial Brewery 117(4): 556-568.
- Pe´rez-Gregorio RM., et al. “Identification and quantification of flavonoids in traditional cultivars of red and white onions at harvest. Journal of Food Composition and Analysis, 23(2014): 592–598.
- Rashidi AN., et al. “Nutritional and antioxidant values of oyster mushroom P. sajor-caju cultivated on rubber sawdust”. International Journal on Advanced Science Engineering Information Technology 6 (2016): 161-164.
- Restuccia C., et al. “An alkaline β-glucosidase isolated from an olive brine strain of Wickerhamomyces anomalous”. FEMS Yeast Research (2011): 487-493.
- Sarker SD., et al. “In natural products isolation, 3rd edition, Follow-up of natural products isolation, Humana Press/Springer-Verlag, New Jersey, (2012): 473-514.
- Stankovic M.S., et al. “Total phenolic content, flavonoids concentration and antioxidant activity of Marrubium peregrinum L. extracts”. Kragujevac Journal Science33(2011): 63-72.
- Stankovic MS., et al. “Total phenols, flavonoid content and antioxidant power of leaf, flower, and fruits from cornelian cherry Cornus mas L”. Bulgarian Journal Agricultural Science 20.2 (2014): 358-363.
- Turner WB. “Fungal metabolites. Academic Press, London (1971).
- Wickerham LJ. “Taxonomy of yeasts. Technical Bulletin of the U.S. Department of Agriculture”. 1029(1951): 1-55.
- Vimalkumar CS., et al. “Comparative preliminary phytochemical analysis of ethanolic extracts of leaves of Olea dioica Roxb., infected with the rust fungus Zaghouania oleae (EJ Butler) Cummins and non-infected plants”. Journal of Pharmacognosy and Photochemistry 3.4 (2014): 69-72.
- Zheng W., et al. “Antioxidant activity and phenolic compounds in selected herbs”. Journal of Agricultural & Food Chemistry 49.11(2001): 5165-5170.
Citation:
Eman M Mohamed., et al. “Screening of the Yeast Phenols and Flavonoids by Many Analytical Methods”. Clinical Biotechnology
and Microbiology 2.6 (2018): 521-534.
Copyright: © 2018 Eman M Mohamed., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.






























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.