Research Article
Volume 1 Issue 2 - 2018
Hormone Receptor Status, Tumor Characteristics and Clinicopathological Features in Breast Carcinomas - 3 year Retrospective Study.
Professor, Department of Pathology, Jawaharlal Nehru Medical College, Aligarh Muslim University, Aligarh. (U.P)-India
*Corresponding Author: Dr. Kafil Akhtar, Professor, Department of Pathology, Jawaharlal Nehru Medical College, Aligarh Muslim University, Aligarh. (U.P)-India.
Received: February 22, 2018; Published: March 03, 2018
Abstract
Introduction: Breast cancer is a heterogeneous disease with a wide spectrum of clinical, pathologic, and molecular features. The molecular classification is becoming the gold standard for complete characterization of breast cancer and the underlying technology has already generated gene profiling models to predict outcomes. Despite these remarkable achievements, in general, clinicians still rely on traditional clinic-pathologic features and readily available tumor markers such as estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (Her2/neu).
Material and Methods: A total of 125 cases of invasive breast cancers were classified into molecular subtypes by the immune-histochemical marker status by staining with immunostains for ER, PR and Her 2/Neu with primary antibody (Novocastra ready to use mouse monoclonal antibodies). Clinico-pathological parameters like age, tumor grade, tumor type and stage were also analyzed.
Results: Out of the 125 cases of invasive breast cancers, infiltrating ductal carcinoma (IDC) comprised 107 (85.0%) cases followed by mixed cancers, 4 (3.2%) cases. There were 45 ER positive cases, 34 PR positive and 70 Her 2/neu positive cases and 33 triple negative, with 38 (84.4%) ER positive cases of IDC. Morphoimmunohistologic findings in breast cancer molecular classes were 19 cases (15.2%) of luminal A subtype, 11 (8.8%) luminal B, 33 cases (26.4%) basal like and 57 (45.6%) cases of Her2/neu over-expression. Her2/neu positivity was more frequent in postmenopausal women and in stage IIa, 10 cases (27.0%).
Conclusions: ER, PR and Her2/neu are reliable, inexpensive immune-markers and useful for therapeutic decision making, and the results of these tests are recorded in cancer registries allowing for population based research which make them a reasonable substitute for the more expensive molecular subtyping.
Keywords: Breast Carcinoma; Histopathology; Immunohistochemistry
Introduction
Over the last few decades there have been outstanding advances in breast cancer management leading to earlier detection of disease and the development of more effective treatments resulting in significant declines in breast cancer deaths and improved outcomes for women living with the disease [1,2]. Breast cancer is a heterogeneous and multifaceted disease comprising of distinct biological subtypes with diverse natural history. It presents a varied spectrum of clinical, pathologic and molecular features with different prognostic and therapeutic implications [3].
Recent attention has been directed singularly at molecular classifications of breast cancer [3,4]. While molecular and genetic testing is very elegant, prognostic and predictive, it is expensive and not yet widely available. The molecular classification is becoming the gold standard for complete characterization of breast cancer and the underlying technology has already generated gene-profiling models to predict outcomes [3,4]. The immunohistochemistry (IHC) classification provides both therapeutic and prognostic information [5]. In general, clinicians still rely on traditional clinicopathologic features and readily available tumor markers such as estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2).
ER, PR, and HER2 are reliable and inexpensive immune-markers, and useful for therapeutic decision making, and the results of these tests are recorded in cancer registries allowing for population based research which make them a reasonable substitute for the more expensive molecular subtyping [5,6].
Material and Methods
The present study was conducted on 125 patients of invasive breast cancers in the Department of Pathology, Jawaharlal Nehru Medical College, Aligarh Muslim University, and Aligarh.
The archival details of the patients with complete history, including age of presentation, duration of disease, age at menarche, age at menopause, duration of breast feeding, any first degree relative having positive history for breast cancer, intake of oral contraceptive pills/hormone replacement therapy; complete physical examination and preliminary investigations were recorded in each case.
Mastectomy and lumpectomy specimen blocks of invasive breast cancers cases were cut into 3-5μ thick sections and immunostains for ER, PR and Her2/Neu were employed with primary antibody (Novocastra ready to use mouse monoclonal antibodies). Haematoxylin & Eosin stained histological sections were assigned diagnosis and typed according to WHO classification of tumours of the breast (2012) and grading of malignant cases was done as per Modified Scarff-Bloom-Richardson grading scheme.
ER/PR scoring: 0: No nuclear staining, 1 + : < 10% nuclear staining in tumor cells, 2+: 10-75% nuclear staining in tumor cells, 3 + : > 75% nuclear staining in tumor cells.
Her2/Neu scoring: 0: No staining, 1+: faintly perceptible membrane staining in < 10% tumor cells, 2+: weak to moderate complete membrane staining in > 10% tumor cells, 3+: strong complete membrane staining in > 30% tumor cells.
Results
Out of the 125 cases of breast cancers, invasive ductal type was the most common subtype, 107 (85.0 %) cases with clusters of pleomorphic ductal cells in the stroma (Figure 1), followed by mixed cancers, 4 (3.2%) cases. Invasive ductal cancers were maximally seen in the fourth decade 43(40.2%) cases, closely followed by 28 (26.1%) cases in the third decade of life (Table 1).
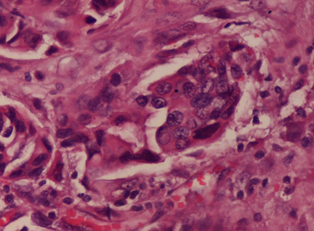
Figure 1: Infiltrating Ductal Carcinoma: Microscopic section shows clusters
of pleomorphic ductal cells in the stroma. Hematoxylin & Eosin x 40X.
| Age (YRS) | IDC | ILC | Mucinous | Medullary | Tubular | Metaplastic | Mixed (Lobular/Ductal) |
Papillary | Poorly Differentitated |
| 21-30 | 07 | - | - | - | - | - | - | - | 01 |
| 31-40 | 28 | 01 | - | 02 | 01 | 01 | 01 | 01 | - |
| 41-50 | 43 | 01 | - | 01 | - | 01 | 02 | - | - |
| 51-60 | 19 | 01 | - | - | - | - | 01 | - | - |
| > 60 | 11 | - | 02 | - | - | 01 | - | - | - |
Table 1: Age Wise Distribution of Breast Cancer Subtypes.
Hormone reactivity in different subtypes of breast cancers showed 45 ER reactive cases with strong nuclear positivity (Figure 2), of which 38(84.5%) were of invasive ductal type and 3 (6.7%) were mixed cancers. PR positivity was seen in 34 cases, majority of which were invasive ductal carcinomas, 29 (85.3%) cases. Her2/neu positivity was seen in 70 cases, with 63 (90.0%) cases being invasive ductal type. Majority of the total 33 triple negative cases were invasive ductal type, 29 (87.9%) (Table 2).
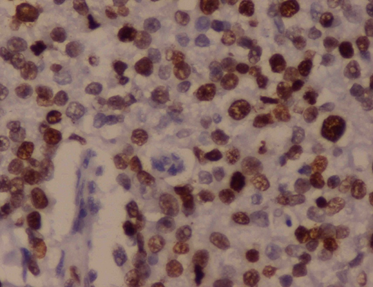
Figure 2: Infiltrating Ductal Carcinoma: Section shows strong
nuclear positivity of tumor cells. IHC ER x 40X.
| Type of tumor | ER Positive | PR Positive | Her2positive | Triple Negative |
| IDC | 38 | 29 | 63 | 29 |
| ILC | - | 01 | 02 | - |
| Mucinous | 01 | - | - | - |
| Medullary | - | 02 | - | |
| Tubular | 01 | 01 | - | - |
| Metaplastic | 01 | 01 | 01 | 01 |
| Mixed | 03 | 01 | 02 | - |
| Invasive Papillary | 01 | 01 | - | - |
| Poorly Differentiated | - | - | - | 01 |
| Total | 45 | 34 | 70 | 33 |
Table 2: Breast Cancer Subtypes and Hormone Receptor Status.
On categorization in accordance with molecular classification of breast cancers, most of our cases were Her2/neu positive, 57 (45.6%), followed by triple negative, 33 (26.4%) cases, luminal A type, 19(15.2%) cases, luminal B, 11(8.8%) cases and unclassified, 5 cases (0.25%) (Table 3).
| Type of Breast Cancer | No of Cases | Percentage |
| Luminal A (ER +, PR +, HER2 -) | 19 | 15.2 |
| Luminal B (ER +, PR +, HER2 +) | 11 | 8.8 |
| Triple Negative (ER -, PR -, HER2 -) | 33 | 26.4 |
| Her2neu (ER -, PR -, HER2 +) | 57 | 45.6 |
| Unclassified | 05 | 4.0 |
Table 3: Molecular Classification of Breast Cancer.
Correlation between stage of tumor with hormone status showed that most of ER positive cases belonged to Stage IIb, 7 (22.5 %), closely followed by Stage IIIa 6 (19.4 %). Majority of the PR positive cases were in Stage IIIa, 5 (22.7 %) and most of the Her2/neu positive cases were in Stage IIa, 10 (27.0%) (Table 4).
| Stage | ER Positive | PR Positive | Her2/Neu Positive |
| I A | 02 | 01 | 02 |
| I B | 01 | - | - |
| II A | 05 | 04 | 10 |
| II B | 07 | 04 | 08 |
| III A | 06 | 05 | 08 |
| III B | 03 | 03 | 09 |
| III C | - | - | - |
| IVA | - | - | - |
| Total | 31 | 22 | 37 |
Table 4: Breast Cancer Staging and Hormone Receptor Status.
Hormone reactivity with respect to age in our study showed that ER and Her2neu positivity was mostly seen in the fourth decade, 19(39.5%) and 25 cases (35.2 %) respectively. A high in PR positivity was seen in the third decade, 12 cases (34.2%) (Table 5).
| AGE (Years) | ER Positive | PR Positive | Her2 Positive | Triple Negative |
| 21-30 | 03 | 03 | 06 | 01 |
| 31-40 | 15 | 12 | 21 | 09 |
| 41-50 | 19 | 11 | 25 | 15 |
| 51-60 | 05 | 05 | 14 | 05 |
| > 60 | 06 | 04 | 05 | 03 |
| Total | 48 | 75 | 71 | 33 |
Table 5: Hormone Receptor Status in Relation to Age.
Discussion
With rising incidence and awareness, breast cancer has become the commonest cancer in urban Indian females and the second commonest in the rural Indian women [1].
The most common histological type of breast cancer encountered in our study was invasive ductal type, which correlates to WHO classification of invasive breast carcinomas [6]. We found ER and PR positive expression to be greater in ductal than in lobular tumors, unlike previous studies by Jalava., et al. [7]. Raina., et al. have reported that invasive ductal carcinoma not otherwise specified (IDC NOS) was the most common type (88.0%) followed by infiltrating lobular carcinoma (3.7%), colloid carcinoma (1.1%), ductal carcinoma in situ (DCIS) (1.1%), and met aplastic types (0.9%) [8].
Estrogen receptors (ER) and progesterone receptors (PR) was found to be positive in 50-60.0% patients in our study, though previous studies showed positivity in only 20-45.0% of Indian patients with ER-positive rates to be lower in Indian patients than in western countries [9,10]. Subset of patients in India undergoing hormonal receptor testing is quite less, as only 35.5% of patients had receptor testing [10]. The ER and PR hormone expression in our study was 38.4% and 28.0% respectively, whereas western studies have reported a higher incidence of receptor expressions [11]. The cause for the low receptor expression in Indian patients can be attributed to geographic differences, younger age and higher grade of breast cancers [11,12]. Discordant findings in our study may also be due to racial differences, as earlier reports were based on western population [13].
In quite contrast to the well-known age group affected by the disease, our results showed 72.0% of the affected patients to be less than 50 years. Majority of our patients (34.0%) were in the early fifth decade of life. Primary breast carcinoma arising before 40 years age are far more aggressive and more likely to metastasize and reduce patients survival than those arising in older patients, regardless of hormone receptor status [13]. Hormone receptor positivity has been associated with decreased breast cancer mortality, independent of various demographic factors and clinical tumor characteristics [14].
ER positivity in premenopausal and postmenopausal women was found to be 41.1% and 31.4% respectively in our study. Very few studies done in India look at the differences in ER status with respect to menopausal status. Kaul et al and Gautam et al have demonstrated ER positivity to be 23.1% and 56.1% in premenopausal women and 36.6% and 47.7% in postmenopausal women respectively [13,14].
The proportions of PR positive tumors in the premenopausal and postmenopausal age groups were found to be 28.8% and 25.7% respectively by us. Kaul., et al. in their study conducted in the Himalayan Region of Northern India found PR positivity to be 23.1% and 39.0% respectively in premenopausal and postmenopausal women [13]. Another study by Gautam., et al. recorded it to be 47.7% and 34.7% respectively [14]. The proportion of breast carcinoma cases with HER2/neu overexpression was 56.0% compared to 10-34.0 % HER2/neu positive breast carcinomas as reported earlier [15]. With respect to the association of HER-2neu expression with age, our study showed 75.0% cases were less than 50 years old, a finding concordant to reports in earlier literatures that HER-2/neu over expression declines with age [16,17].
The younger age group of patients with higher Her2/neu reactivity in our study can be attributed to geographical variations. Overexpression of HER2 in tumor cells is associated with poor prognosis [18]. The poor prognosis of HER2 positivity is related to high risk of early relapse [18].
Triple- negative breast carcinoma are associated with poor prognosis [17]. The proportion of the triple-negative breast carcinoma in our study was 26.0%, quite concordant with other Indian studies [11,12]. The term "basal" type of carcinoma is often used interchangeably with triple-negative carcinoma, having adverse clinico-pathological features and poor prognosis [18].
Considering the intrinsic or molecular subtypes, our study showed 45.6% cases to be Her2/neu type, 26.0% were basal like and 24.0% were luminal type, a finding discordant with previous studies, which showed higher proportion of luminal subtypes [12]. Basically, the luminal subtypes share expression of estrogen receptor (ER)–related genes and have better overall survival than the HER2-related and the basal-like subtypes, which are typically (but not uniformly) ER negative [17,18].
Conclusions
The different subset of patients with quite distinct hormonal profile pose a diagnostic and therapeutic challenge, and necessitate further research. Emerging gene expression analysis has unraveled distinct molecular classes of breast cancer that can provide more accurate prognostic and therapeutic prediction than existing histological classification. But still hormone receptor status is widely used by clinicians in guiding treatment, due to cost effectiveness, easy availability and time constraints.
References
- National Cancer Registry Program. Ten year consolidated report of the Hospital Based Cancer Registries, 1984–1993, an assessment of the burden and care of cancer patients. New Delhi: Indian Council of Medical Research; 2001.
- Verma S., et al. “Immunohistochemical characterization of molecular subtypes of invasive breast cancer: a study from North India”. Acta Pathologica, Microbiologica, et Immunologica Scandinavica 120.12 (2012): 1008-1019.
- Shawarby MA., et al. “Molecular classification of breast cancer: An overview with emphasis on ethnic variations and future perspectives”. Saudi Journal of Medicine and Medical Sciences 1.1 (2013): 14-19.
- Dunnwald LK., et al. “Hormone receptor status, tumor characteristics, and prognosis: a prospective cohort of breast cancer patients”. Breast Cancer Research 9.1 (2007): 12-14.
- Suvarchala SB and Nageswararao R. Carcinoma Breast histopathological and hormone receptors correlation. Journal of Bioscience and Technology 2.4 (2011): 340-48.
- Ellis IO., et al. “World Health Organization classification of Tumours, Pathology and Genetics of Tumours of the Breast and Female Genital Organs”. International Agency for Research on Cancer (2003): 20-24.
- Jalava P., et al. “Immunohistochemical staining of estrogen and progesterone receptors: Aspects for evaluating positivity and defining the cut points”. Anticancer Research 25.3 (2005): 253525-42.
- Raina V., et al. “Clinical features and prognostic factors of early breast cancer at a major cancer center in North India”. Indian Journal of Cancer 42.1 (2005): 40-45.
- Azizun-nisa., et al.“Comparison of ER, PR, and Her-2/neu (C-erb B2) reactivity pattern with histologic grade, tumor size and lymph node status in breast cancer”. Asian Pacific Journal of Cancer Prevention 9.4 (2008): 553-56.
- Ratnatunga N and Liyanapathirana LV. “Hormone receptor expression and Her/2neu amplification in breast carcinoma in a cohort of SriLankans”. Ceylon Medical Journal 52.4 (2007): 133-36.
- Cummings MC., et al. “Molecular classification of breast cancer: is it time to pack up our microscopes?” The Journal of Pathology 43.1 (2011): 1-8.
- Saleh F and Abdeen S. “Patho-biological features of breast tumours in the state of Kuwait: A comprehensive analysis”. Journal of Carcinogenesis 6 (2007): 12-15.
- Kaul R., et al. “Hormone receptor status of breast cancer in the Himalayan Region of Northern India”. Indian Journal of Surgery 73.1 (2011): 9-12.
- Gautham R., et al. “Estrogen and Progesterone receptor status in breast cancer: a cross sectional study of 450 women in Kerala”. World Journal of Surgical Oncology 12 (2014): 120-23.
- Shahi KS., et al. “Steroid receptor status and its clinicopathological correlation in postmenopausal breast cancer patients of Kumaon region of Uttarakhand”. Journal of Cancer Research and Therapeutics 7.1 (2011): 19-22.
- Ambroise M., et al. “Immunohistochemical profile of breast cancer patients at tertiary care hospital in South India”. Asian Pacific Journal of Cancer Prevention 12.3 (2011): 625-629.
- Ghosh J., et al. “Estrogen, progesterone and HER2 receptor expression in breast tumors of patients, and their usage of Her2-targeted therapy, in a tertiary care centre in India”. Indian Journal of Cancer 48 (2011): 391-396.
- Thike AA., et al. “Triple-negative breast cancer: clinicopathological characteristics and relationship with basal -like breast cancer”. Modern Pathology 23.2 (2010):123-133.
Citation:
Kafil Akhtar., et al. “Hormone Receptor Status, Tumor Characteristics and Clinicopathological Features in Breast Carcinomas
- 3 year Retrospective Study.”. Chronicle of Medicine and Surgery 1.2 (2018): 72-78.
Copyright: © 2018 Kafil Akhtar., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.