Review Article
DOI: 10.5281/zenodo.2247890
Volume 3 Issue 1 - 2018
Alzheimer’s FDA Approved Drugs & Alternatives: What are They Really Treating?
New York Institute of Medical Research, USA
*Corresponding Author: Dr Lawrence Broxmeyer MD, New York Institute of Medical Research, USA. Email: drlawrencebroxmeyermd@alumni.usc.edu
Received: November 28, 2018; Published: December 10, 2018
Abstract
A 2018 study by Tricco., et al. ranking the safety and effectiveness of the four leading drugs now taken to enhance concentration, memory, alertness and moods in, found that donepezil (Aricept®) was most likely to effectively improve cognition in patients with Alzheimer’s dementia. However, patients who took donepezil were more likely to experience side effects including nausea, vomiting and diarrhea than those who received a placebo, according to the study, published online in the Journal of the American Geriatrics Society [1]. And an earlier study showed that almost 19% of patients in an Aricept 23 mg daily group discontinued treatment due to side effects [2]. The fact is that as for all conventional drugs used in Alzheimer’s treatment –only a small percentage of AD victims are helped by them.
Yet, in both the case of FDA approved and alternative treatments presently available, much can be gleaned by scrutinizing which pathogen or pathogens the bulk of their activity is directed towards. It seems that several compounds said to have effects upon the neuronal systems in Alzheimer’s may also exert influence through other mechanisms, such as antimicrobial actions.
Introduction
Razadyne® (galantamine), Exelon® (rivastigmine), and Aricept® (donepezil) are all classified as cholinesterase inhibitors. According to the National Institute on Aging (https://www.nia.nih.gov/health/how-alzheimers-disease-treated), scientists do not yet fully understand how cholinesterase inhibitors work to treat Alzheimer’s disease. To fill in this void researchers have drawn upon the fact that such inhibitors prevent the breakdown of acetylcholine, a brain chemical believed to be important for memory and thinking. The 4th medication in Tricco’s 2018 Alzheimer’s study however, Namenda® (memantine), is an N-methyl D-aspartate (NMDA) antagonist, which works differently. It blocks the actions of the neurotransmitter, glutamate. Glutamate is needed for memory, but too much of it is toxic to nerve cells, and it appears that in people with Alzheimer’s, there is too much of it (again, for unknown reasons).
The Experimental Agent That Invariably Causes Alzheimer’s
Alzheimer’s is spoken of as a ‘disease’. So what is that disease? Recently there has been a resurgence in thought that a chronic, in most cases sub-clinical, infectious agent is behind it. Yet the most widely used method to create the symptoms and signs of Alzheimer’s disease in animals is Complete Freund’s Adjuvant (CFA or FCA). It is Complete Freund’s Adjuvant (CFA) which creates an experimental allergic encephalitis (EAE) in laboratory animals that demonstrates a cognitive impairment in EAE rats 90 days post-immunization (dpi). It is CFA which is associated with a significant decrease in the enzyme choline acetyltransferase (Chat), which synthesizes acetylcholine in the brain for memory acquisition and maintenance. And it is the EAE as a result of Complete Freund’s Adjuvant that is used to show that acetyl cholinesterase selective inhibitors such as rivastigmine and donepezil almost completely restore behavioral/cognitive performance and Chat activity levels [3]. Moreover, with such experimental EAE, there is also excess glutamate levels, and multiple abnormalities in glutamate degrading enzymes, and glutamate receptors [4]. So all of the theoretical underpinning upon which today’s leading Alzheimer’s drugs were FDA approved are based and can be found in that experimental method devised by Jules T. Freund. Freund might have been looking for improved vaccine techniques, but what he developed would be used subsequently experimentally, at least in animals, to induce aspects of a variety of diseases, including neurodegenerative disease similar to Alzheimer’s. At least initially, Julius Freund’s adjuvant included marinating apparently “killed” species of one of the most deadly infectious pathogens in the world, the tubercle bacilli, in oily vehicles. He knew, through the work of previous researchers, that such oily vehicles potentiated the activity of TB. Freund’s initial concoction eventually became known as “Complete Freund’s Adjuvant” (CFA), with its “killed” TB. If the complete form of Freund’s Complete Adjuvant (FCA or CFA) is composed of inactivated and dried mycobacteria (usually M. tuberculosis), then its incomplete form (FIA or IFA), which Freund later devised, lacked the mycobacterial components (hence just the water in oil emulsion).
Alzheimer’s is spoken of as a ‘disease’. So what is that disease? Recently there has been a resurgence in thought that a chronic, in most cases sub-clinical, infectious agent is behind it. Yet the most widely used method to create the symptoms and signs of Alzheimer’s disease in animals is Complete Freund’s Adjuvant (CFA or FCA). It is Complete Freund’s Adjuvant (CFA) which creates an experimental allergic encephalitis (EAE) in laboratory animals that demonstrates a cognitive impairment in EAE rats 90 days post-immunization (dpi). It is CFA which is associated with a significant decrease in the enzyme choline acetyltransferase (Chat), which synthesizes acetylcholine in the brain for memory acquisition and maintenance. And it is the EAE as a result of Complete Freund’s Adjuvant that is used to show that acetyl cholinesterase selective inhibitors such as rivastigmine and donepezil almost completely restore behavioral/cognitive performance and Chat activity levels [3]. Moreover, with such experimental EAE, there is also excess glutamate levels, and multiple abnormalities in glutamate degrading enzymes, and glutamate receptors [4]. So all of the theoretical underpinning upon which today’s leading Alzheimer’s drugs were FDA approved are based and can be found in that experimental method devised by Jules T. Freund. Freund might have been looking for improved vaccine techniques, but what he developed would be used subsequently experimentally, at least in animals, to induce aspects of a variety of diseases, including neurodegenerative disease similar to Alzheimer’s. At least initially, Julius Freund’s adjuvant included marinating apparently “killed” species of one of the most deadly infectious pathogens in the world, the tubercle bacilli, in oily vehicles. He knew, through the work of previous researchers, that such oily vehicles potentiated the activity of TB. Freund’s initial concoction eventually became known as “Complete Freund’s Adjuvant” (CFA), with its “killed” TB. If the complete form of Freund’s Complete Adjuvant (FCA or CFA) is composed of inactivated and dried mycobacteria (usually M. tuberculosis), then its incomplete form (FIA or IFA), which Freund later devised, lacked the mycobacterial components (hence just the water in oil emulsion).
Billiau and Matthys, after a thorough review of the literature, stated, “Clearly, the host response to an injection of Complete Freund’s Adjuvant (CFA) should be seen as resembling one coping with a slowly fading primary infection with living mycobacteria” [5] —specifically Mycobacteria tuberculosis. Billiau and Matthys’s caution is well taken. “Heat-killed” tuberculosis and its related mycobacteria, either in vaccination or even as a tuberculin skin test, have dormant, practically indestructible cell-wall-deficient forms that can revert back to virulent TB bacilli—“killed” by neither heat nor sterilization [6-8]. Yet it was just this introduction of tubercular CFA in the late 1940s that quickly became and remains the gold standard for invoking experimental “autoimmune” disease in animals— including the induction of a brain-inflaming, plaque-producing, Experimental Autoimmune Encephalopathy [EAE]. All it took was a single injection of what Freund eventually settled on as brain material mixed with killed tuberculosis in an oily vehicle.
Freund would also go on to develop an “Incomplete” adjuvant without TB in it, subsequently called “Incomplete Freund’s Adjuvant”
(IFA). But this paled when compared to the “immune response” created by Freund’s with TB. The way Freund’s Complete Adjuvant
works still is not clearly understood; yet immunologists continue to use it experimentally to create disease in animals, complete with
its tubercular components. This lack of understanding is likewise true for Freund’s Incomplete Adjuvant without the TB, used again
without really knowing or caring how it supposedly “bolsters the immune system” or the exact profile of its side effects. For reasons
such as this, Janeway and others have called adjuvants such as Freund’s “the immunologist’s dirty little secret” [9]. As testimony to this,
we read paper after paper on Medline involving the obligatory use of tubercular elements to create serious experimental “autoimmune”
disease such as EAE –papers in which the use of “dead” tubercular elements are hidden in the Materials and Methods section, only to be
seemingly selectively forgotten about in fashioning a concluding discussion.
In the meantime, what currently remains the theory behind how Freund’s Complete Adjuvant is thought to work, and how it really
works, do not always match. For example, many assume that when the “active” ingredient, such as injected neural material, is
introduced into Complete Freund’s Adjuvant with TB (CFA) that such CFA adjuvant works merely by increasing such neural or brain
material’s effective antigenic delivery. This supposedly sets off a complex set of signals to the immune system that alter white-bloodcell
proliferation and differentiation. Yet in guinea pigs and mice, the tubercular elements in CFA are mandatory for EAE, and just brain
or nervous tissue alone will usually not work [10]. Thus, it is Freund’s Complete Adjuvant, complete with elements of TB, which is
needed in experimental models of “autoimmune” diseases, including EAE, an animal model of brain inflammation with a propensity
for instigating plaque in the brain, including Alzheimer’s neuritic amyloid plaque and tangles. When injected into mice, CFA showed
widespread ß-amyloid plaque in the cerebral cortex and hippocampus—the part of the brain that is involved in memory—and within
just four months of their being injected [11].
Although Freund’s Adjuvant and the EAE it causes could go a long way in defining which drugs would (at least temporarily) help
with cognition, this method alone could not explain why Tricco., et al. found donepezil (Aricept®) was most likely to effectively improve
cognition in patients with Alzheimer’s dementia among the other drugs of its class. However recent studies point in an interesting direction
which might explain its efficacy. Oskar Fischer, the co-discover of Alzheimer’s disease, mentioned on many occasions that a germ
called Streptothrix, frequently and consistently mistaken for filamentous, cell-wall-deficient forms of the tubercular bacilli, was quite
possibly the cause of Alzheimer’s [12]. Bacteriologist Victor Babes backed this finding up when he injected subdural TB into animals
and the result was a central nervous system disease which appeared just like Fischer’s Streptothrix [13].
Unexplained Avenues
The precise mechanism of action of donepezil in patients with Alzheimer’s disease is not fully understood. One unexplored avenue is that certain piperidine analogues of drugs similar to Aricept® have been shown recently to be highly active against Mycobacteria such as the tubercular bacillus [14-17]. Also, in 2018, an investigation of the drug fasudil for Alzheimer’s was done. Importantly, the investigators indicated that fasudil, which is safe in humans and readily enters the brain, was a very promising candidate treatment for Alzheimer’s disease [18]. But fasudil has already been approved in some other countries for the treatment of tuberculosis [19].
The precise mechanism of action of donepezil in patients with Alzheimer’s disease is not fully understood. One unexplored avenue is that certain piperidine analogues of drugs similar to Aricept® have been shown recently to be highly active against Mycobacteria such as the tubercular bacillus [14-17]. Also, in 2018, an investigation of the drug fasudil for Alzheimer’s was done. Importantly, the investigators indicated that fasudil, which is safe in humans and readily enters the brain, was a very promising candidate treatment for Alzheimer’s disease [18]. But fasudil has already been approved in some other countries for the treatment of tuberculosis [19].
The notion that first line anti-tubercular drugs such as rifampicin might have a role in the treatment or prevention of Alzheimer’s
never went away, and recent studies show it to be more conceivable than ever [20,21]. By 1984, de Beer [22], studying the relationship
between a major rises in serum amyloid and having tuberculosis, also saw a rapid descent in amyloid in patients treated with antitubercular
drugs. As an offshoot of de Beer’s work, Tomiyama [23] dissolved ß-amyloid plaque with rifampicin, a first-line drug in the
treatment of TB, and one of the few agents, to this day, which is able to dissolve amyloid plaque. And so by 2004, Anthony L. Fink., et al.
working out of the University of California, Santa Cruz concluded that rifampicin also inhibits α-Synuclein fibrillation and disaggregates
fibrils [24]. Government trials followed which used inadequate amounts of rifampicin for an inadequate time. Yet such trials could not
rule out a therapeutic role for its use in mild to moderate AD and authors encouraged more research in this area. In the meanwhile, a
2012 Journal of Alzheimer’s disease study pointed also to the enhanced brain Amyloid-β clearance by rifampicin and caffeine as a possible
protective mechanism against Alzheimer’s disease [25]. The possible role of mycobacterial disease, a prominent genus of Actinobacteria,
has been described for Alzheimer’s [26-28].
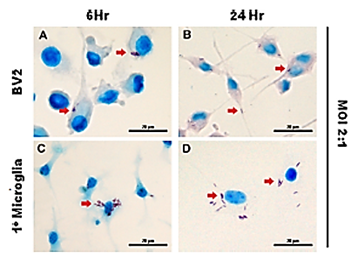
Figure 1: Neurons in the brain hosting tuberculosis. (Red arrows) M. tuberculosis infecting microglia. The microglial cell line BV2 (A and B) and primary microglia (C and D), isolated from the cortices
of 1-day-old C57BL/6 neonates, were infected with M. tuberculosis at an MOI of 2:1 for 6 and
24h. The arrows indicate the uptake of acid-fast bacilli by the microglial cells. Scale bars, 20 μm.
With permission. [Randall PJ, Hsu N-J, Lang D., et al. Neurons Are Host Cells for Mycobacterium
tuberculosis. Appleton JA, Ed. Infection and Immunity. 2014; 82(5): 1880-1890.]
The findings in mouse models [29] indicated that rifampicin could serve as a promising available medicine for the prevention of AD.
It therefore, once again, became of interest whether rifampicin had such preventive effects in humans. Thus by June of 2017, Iizuka., et
al. set out to determine this in a study [20] using 40 patients with mycobacterial disease in which dementia had not yet set in, and yet
who showed AD-like hypometabolism patterns under stereotaxic statistical analysis. As an important offshoot of this study, besides addressing
whether rifampicin had preventative effects with Alzheimer’s, Iizuka’s raw data showed that in his two groups [Group A and
Group B] of elderly patients with mycobacterial disease –approximately 46% of those in Group A showed Alzheimer’s type hypometabolism
while in Group B approximately 55% showed AD-like hypometabolism. This averages out to slightly over half of the patients
with mycobacterial disease showing AD-like hypometabolism on FDG-PET.
To examine whether rifampicin has such preventive effects against dementia in humans, Iizuka retrospectively reviewed FDG-PET
scan findings of elderly patients with mycobacterial infection treated with rifampicin. For over 20 years, FDG-PET [also known as 18FFDG-
PET] has been used to measure cerebral metabolic rates of glucose (CMRglc) as a proxy for neuronal activity in AD. Many studies
have shown that CMRglc reductions occur early in AD, both correlating with disease progression, and predicting histopathological
diagnosis [30].
In their introduction Iizuka., et al. mention that statistically Alzheimer disease (AD) is the most common cause of neurodegenerative
dementia and is becoming increasingly common as the global population ages [31]. And that therefore, development of preventive
therapy for the disease has been urgently needed. As both amyloid-β and tau were believed to play central roles in AD pathogenesis,
they have been targets of disease-modifying therapy. However clinical studies of amyloid-β-targeting therapies in AD have revealed that
the treatments after disease onset have little effect on the cognition of patients [32-34]. One presumable reason why rifampicin was not
performing up to expectations in previous Alzheimer’s studies might be that the treatment of AD should have been started prior to the
onset of clinical symptoms [35]. Also, rifampicin is an antibiotic which is easy for mycobacterium to gain resistance to. To prevent the
manifestation of resistance to rifampicin in mycobacterium, it should be administered with other antibiotics as a combination therapy.
Iizuka’s results showed, through multiple regression analysis, the dose of rifampicin and treatment duration significantly
influenced FDG uptake in the PCC. Iizuka concluded that the preventive effect of rifampicin depended on the dose and the treatment
duration, and that the desired effect required at least 450 mg daily for 1 year.
By March 2018, still another rifampicin/Alzheimer’s study appeared on Medline, this one a multi-center probe, concluding, once
again, that rifampicin exerts significant brain protective functions in multiple experimental Alzheimer’s models [21]. In this capacity
rifampicin was found to have a neuroprotective and pro-cognitive effect that was mediated by its anti-inflammatory, anti-tau, and antiamyloid
effects.
Alternative Treatments Desperately Sought
Because of the generally dismal outlook for people taking presently FDA approved Alzheimer’s drugs, there have been attempts to treat Alzheimer’s disease through alternative methods. No alternative treatment should be taken that is not under the direct supervision of a physician. However, let’s take a look at some of these.
Because of the generally dismal outlook for people taking presently FDA approved Alzheimer’s drugs, there have been attempts to treat Alzheimer’s disease through alternative methods. No alternative treatment should be taken that is not under the direct supervision of a physician. However, let’s take a look at some of these.
Curcumin
Though research has shown that curcumin can prevent the formation, and even break up the amyloid-beta plaques associated with Alzheimer’s disease, similar results have not been found in clinical studies, and the exact way that curcumin works within the brain is unclear, although under active investigation [36]. Potter’s evidence suggesting that populations that eat food with a substantial amount of curcumin were at lower risk of Alzheimer’s disease (AD) led to the idea that this compound might have a neuroprotective effect. After a review of the literature, Potter stressed that there is evidence to suggest that the addition of curcumin to cultured neuronal cells protects against β-amyloid-induced neurotoxicity. Moreover curcumin decreases β-amyloid formation from amyloid precursor protein, and also inhibits aggregation of β-amyloid into pleated sheets. Studies in transgenic mice with overproduction of β-amyloid demonstrate a neuroprotective effect of curcumin as well [37].
Though research has shown that curcumin can prevent the formation, and even break up the amyloid-beta plaques associated with Alzheimer’s disease, similar results have not been found in clinical studies, and the exact way that curcumin works within the brain is unclear, although under active investigation [36]. Potter’s evidence suggesting that populations that eat food with a substantial amount of curcumin were at lower risk of Alzheimer’s disease (AD) led to the idea that this compound might have a neuroprotective effect. After a review of the literature, Potter stressed that there is evidence to suggest that the addition of curcumin to cultured neuronal cells protects against β-amyloid-induced neurotoxicity. Moreover curcumin decreases β-amyloid formation from amyloid precursor protein, and also inhibits aggregation of β-amyloid into pleated sheets. Studies in transgenic mice with overproduction of β-amyloid demonstrate a neuroprotective effect of curcumin as well [37].
The most studied activity of the curcumin, a derivative of Turmeric, in the past 12 years has been its anticancer effects [38,39].
However, The first paper describing the biological action of curcumin, including its antibacterial activity was submitted by Schraufstätter.,
et al. to the journal Nature in 1949, demonstrated conclusively that curcumin has anti-bacterial properties and was active against
strains of S. aureus, Trichophyton gypseum, Salmonella paratyphi, and Mycobacterium tuberculosis [40]. Schraufstätter witnessed “a
remarkable inhibition of growth” of Mycobacterium tuberculosis by curcumin in vitro, even at high dilution. And a 2016 study went a
long way further to explain just why. Curcumin is also a potent inducer of apoptosis –an effector mechanism used by macrophages to
kill intracellular Mycobacterium tuberculosis (MTB). As such it may even help fight drug-resistant tuberculosis. Investigators found that
by stimulating human immune cells called macrophages, curcumin was able to successfully remove Mycobacterium tuberculosis, the
causative bacterium of tuberculosis, from experimentally infected cells in culture [41]. This was reminiscent to what Masoumi., et al.
reported from UCLA in a 2009 Journal of Alzheimer’s Disease when they found that vitamin D3 plus curcumin may help stimulate the
immune system to clear the brain of amyloid beta, which forms the plaques considered the hallmark of AD [42].
Coconut Oil
There was just enough previous evidence with coconut oil in Alzheimer’s to support an NIH-sponsored study –a randomized, double-blind, placebo-controlled trial, based at the University of South Florida and designed to determine the effect of taking coconut oil on the cognition, functioning, and behavior of older adults with mild to moderate Alzheimer’s. [Clinical Trials.gov ID# NCT01883648. Accessed Jan. 28, 2018] Nafar and Mearow had published a short communication in the Journal of Alzheimer’s Disease indicating that Coconut oil attenuates the effects of amyloid-β on cortical neurons in vitro and that neuron survival in cultures cotreated with coconut oil and Aβ were rescued compared to cultures exposed only to Aβ [43]. And over at the British Journal of Nutrition Fernando., et al. concluded that phenolic compounds and hormones (cytokinins) found in coconut may assist in preventing the aggregation of amyloid-β peptide, potentially inhibiting a key step in the pathogenesis of AD. However as with the Nafer study the exact mechanism of action of coconut oil for the prevention and treatment of Alzheimer’s disease was unknown [44]. Almost concurrent with the British study Hu Yang., et al. concluded that there was a positive influence of coconut oil at the cognitive level of patients with Alzheimer’s, but that this improvement was dependent on sex, presence or absence of diabetes and degree of dementia [45].
There was just enough previous evidence with coconut oil in Alzheimer’s to support an NIH-sponsored study –a randomized, double-blind, placebo-controlled trial, based at the University of South Florida and designed to determine the effect of taking coconut oil on the cognition, functioning, and behavior of older adults with mild to moderate Alzheimer’s. [Clinical Trials.gov ID# NCT01883648. Accessed Jan. 28, 2018] Nafar and Mearow had published a short communication in the Journal of Alzheimer’s Disease indicating that Coconut oil attenuates the effects of amyloid-β on cortical neurons in vitro and that neuron survival in cultures cotreated with coconut oil and Aβ were rescued compared to cultures exposed only to Aβ [43]. And over at the British Journal of Nutrition Fernando., et al. concluded that phenolic compounds and hormones (cytokinins) found in coconut may assist in preventing the aggregation of amyloid-β peptide, potentially inhibiting a key step in the pathogenesis of AD. However as with the Nafer study the exact mechanism of action of coconut oil for the prevention and treatment of Alzheimer’s disease was unknown [44]. Almost concurrent with the British study Hu Yang., et al. concluded that there was a positive influence of coconut oil at the cognitive level of patients with Alzheimer’s, but that this improvement was dependent on sex, presence or absence of diabetes and degree of dementia [45].
But Sadly, back in the US, the NIH-sponsored study based at the Byrd Alzheimer’s Institute of the University of South Florida to
determine the effect of taking coconut oil on the cognition, functioning, and behavior of older adults with mild to moderate Alzheimer’s
had to be discontinued as the number of people enrolled in the trial and its funding was not enough.
Virgin coconut oil has both anti-bacterial and anti-mycobacterial properties. Yet Carbardo found that when ten selected strains of
Mycobacterium tuberculosis (MTB) were exposed for 24 hours to the known minimum inhibitory concentration of GML (Glycerol Monolaurate,
the active ingredient in coconut oil), its activity alone was comparable to any of the first-line antitubercular drugs (isoniazid,
rifampicin and ethambutol) in use today [46]. Carbado’s impressive results gave clear evidence that the GML in coconut oil could kill
highly pathogenic Mycobacterium tuberculosis, both drug susceptible and resistant strains. Glycerol monolaurate’s effect on the pathogen
included partial or complete loss of the tubercle bacilli’s cell components due to burst cells. Furthermore GML stopped cell division
altogether in the pathogen. A SEM (Scanning Electron Microscope) image of a single tubercular bacilli (Mycobacterium tuberculosis)
showing its destruction by the Glycerol Monolaurate in Virgin Coconut Oil was shown in his paper. (Figure 2)
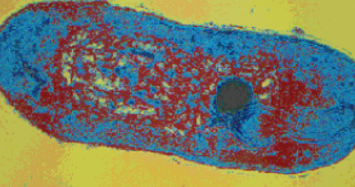
Figure 2: SEM (Scanning Electron Microscope) image of a single
tubercular bacilli (Mycobacterium tuberculosis) showing damage
caused by Glycerol Monolaurate in Virgin Coconut Oil.
Suggesting the Off-Label use of Anti-Herpetics for Alzheimer’s
In the face of a 2006 study by Helmer [47] which found that anti-herpetic medicines such as acyclovir were “not significant” for lessening dementia such as found in Alzheimer’s; Tzeng’s., et al. in a 2018 retrospective Taiwanese study not only suggested that patients with HSV infection “may have” a 2.56-fold increased risk of developing dementia, but that the usage of anti-herpetic medications was associated with a decreased risk of dementia [48].
In the face of a 2006 study by Helmer [47] which found that anti-herpetic medicines such as acyclovir were “not significant” for lessening dementia such as found in Alzheimer’s; Tzeng’s., et al. in a 2018 retrospective Taiwanese study not only suggested that patients with HSV infection “may have” a 2.56-fold increased risk of developing dementia, but that the usage of anti-herpetic medications was associated with a decreased risk of dementia [48].
The history of the Herpes Simplex Type 1 itself harbors inconsistencies. The notion that Herpes simplex might have been caused
by a living infectious agent resulted from the 1912 work of Grüter [49], but its certainty as to whether it was a virus was not at all clear.
Also in Germany, in 1919, Löwenstein, in his Aetiological Studies on Febrile Herpes, produced a typical herpes keratitis, transmissible in
series. Later on, Löwenstein confirmed that herpes was an infectious disease, extending these findings to herpes of the skin and mucous
membranes. Löwenstein showed that not only corneal herpes, but also simple herpes and the symptomatic herpes accompanying
diseases such as tuberculosis produced a specific keratitis when inoculated in the cornea of a rabbit [50].
But when Kooy [51] revisited and confirmed Löwenstein’s results, inoculating the cornea of rabbits with herpetic dendritic
keratitis, and herpes labialis in febrile disease; what Kooy isolated was not a virus at all. Rather from twenty-five inoculations
upon the cornea he succeeded twenty-two times in culturing a polymorphic, cell-wall-deficient, viral-like microorganism belonging
to the Mycobacteriaceae. Mycobacteria are a genus of the Actinobacteria, given their own family, the Mycobacteriaceae. In direct
culture from herpes labialis (cold sores of the lip), Kooy again succeeded three times in culturing these same Mycobacteria.
When she worked on the original anti-herpetic for HSV, Gertrude Elion determined exactly how and why it worked. To Elion,
acyclovir, marketed as Zovirax®, interfered with the replication process of the herpesvirus –and only the herpesvirus –proving that
drugs can be selective. That is until a Polish study proved this wrong. And so while Western literature remained silent about acyclovir
as having anything but anti-viral activity, this Polish study found that acyclovir actually had activity against Pseudomonas aeruginosa
and E. coli –two out of only 4 bacteria used in this study that acyclovir was tested against [52]. The mycobacteria were excluded from
Kruszewska’s study, but a year later, Pochet [53] showed that drugs similar to acyclovir act therapeutically on the thymidylate kinases of Mycobacterium tuberculosis. Pyrimidine and Purine nucleoside analogs such as acyclovir have been extensively studied as anticancer
and antiviral agents. In addition to this, they have recently shown great potential as agents against Mycobacterium tuberculosis [54].
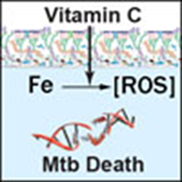
Vitamin C
Harrison, in the Journal of Alzheimer’s Disease concluded that overall, there is a large body of evidence that maintaining healthy vitamin C levels can have a protective function against age-related cognitive decline and Alzheimer’s disease [55]. Vitamin C has a wide spectrum of activity which includes bacteria, mycobacteria and viruses –yet non-mycobacterial microbes are not nearly as sensitive to the compound. In appropriate doses vitamin C is known to drive the Fenton reaction, in all cases sterilizing cultures of drug-susceptible and drug-resistant Mycobacterium tuberculosis, the causative agent of tuberculosis [56]. The exact molecular mechanism is that vitamin C causes iron to react with other molecules to create reactive oxygen species (ROS) that in turn kill the TB (Mtb) mycobacteria.
Harrison, in the Journal of Alzheimer’s Disease concluded that overall, there is a large body of evidence that maintaining healthy vitamin C levels can have a protective function against age-related cognitive decline and Alzheimer’s disease [55]. Vitamin C has a wide spectrum of activity which includes bacteria, mycobacteria and viruses –yet non-mycobacterial microbes are not nearly as sensitive to the compound. In appropriate doses vitamin C is known to drive the Fenton reaction, in all cases sterilizing cultures of drug-susceptible and drug-resistant Mycobacterium tuberculosis, the causative agent of tuberculosis [56]. The exact molecular mechanism is that vitamin C causes iron to react with other molecules to create reactive oxygen species (ROS) that in turn kill the TB (Mtb) mycobacteria.
What Irwin Stone, who introduced Linus Pauling to vitamin C through his book The Healing Factor – Vitamin C Against Disease [57],
had so long ago predicted ― that TB was “particularly sensitive” to vitamin C ― was now scientific reality. Furthermore, vitamin C’s
activity against M. tuberculosis was rather specific since the MICs (Minimum Inhibitory Concentrations) for non-mycobacteria (other
Gram-positive and Gram-negative bacteria) are at least 16 to 32 times higher than against M. tuberculosis. M. tuberculosis was not only
the most susceptible strain to vitamin C among the mycobacterial strains tested, but far more susceptible then other bacteria. Therefore,
whereas the effect of vitamin C on various diseases has been widely described and remains controversial [58]; the sterilizing
effect of vitamin C on M. tuberculosis cultures can no longer be considered controversial.
Conclusion
The cause of Alzheimer’s disease is unknown, and even to so much as speculate otherwise must be done with caution. Chlamydia,
the Herpes Virus type 1, spirochetes, and even fungus have been hypothesized. Yet there is indirect evidence which points in still another
direction. Presently, the only regularly used infectious agent that causes Alzheimer’s-like signs and symptoms in labs worldwide
is Freund’s Complete Adjuvant (FCA), a mycobacterial based formula. And FCA does so with great regularity, causing that experimental
autoimmune encephalomyelitis (EAE) with the plaques and tangles [11] all too familiar in Alzheimer’s. FCA and its resultant EAE are
also the agents of departure for animal experimentation to determine the efficacy of the present FDA approved drugs [3]. Moreover
Freund’s Complete Adjuvant does not have to be injected into the brain directly, as its introduction to create an extra-cerebral focus of
infection, and thereby an immune reaction that involves the brain, can happen from most any part of the body. This in itself indicates
that a mycobacterial agent that might set off this (delayed) immune reaction in the brain can be hiding elsewhere in the body.
Another avenue, often overlooked, is to not only determine which agents, both FDA approved and alternative, work best with Alzheimer’s,
but whether they have a common therapeutic path in doing so. Is there a common thread in their activity through which we
can better predict why they assuage Alzheimer’s signs and symptoms? For example Tricco., et al. determined that of the four leading
drugs presently used to treat Alzheimer’s, it was donepezil (Aricept®), that was most likely to effectively improve cognition in patients with the disease. Is this due to the unexplored avenue that certain piperidine analogues of drugs similar to Aricept® have been shown
recently to be highly active against Mycobacteria such as the tuberculosis bacillus? [14-17] In 2018, fasudil was said to be a very promising
treatment for Alzheimer’s [18]. But fasudil has already been approved in some countries for the treatment of tuberculosis [19].
Just as recently, studies have shown that it is more feasible than ever, at the proper dose, for first line anti-tubercular drugs such as
rifampicin might have a role in the treatment or prevention of AD – all of it based on an earlier study by de Beer [22], who found a relationship
between a major rise in serum amyloid and having tuberculosis, as well as a rapid descent in amyloid in patients treated with
anti-tubercular drugs. Jordal [59] reconfirmed that mycobacteria like TB produce a level of amyloid not previously described for any
other bacteria. Even the “fact” that certain anti-virals only have activity against viruses remains under scrutiny [60]. And in the alternative
arena –curcumin, coconut oil and vitamin C all have extremely high activity against the mycobacteria.
Statistically, it is estimated that there are approximately 13 million people in the U.S. with “latent” TB. https://www.ncbi.nlm.nih.gov/pubmed/26866439. To many, this NIH assessment is a gross underestimation as Fox [61] projected that half the world has TB, and
immigration from countries where the disease is endemic has stepped up considerably in the US, exposing the population at large. During
Alzheimer’s lifetime, tuberculosis’s annual death rate of seven million persons per year was more than twice the approximately two
million to three million people that some estimate it still kills annually [62]. Moreover most mycobacteriologists are familiar with the
concept that “latent” disease can easily reactivate into “active” disease, especially during periods of immunosuppression which increase
naturally with age. At that point a subclinical ‘latent’ infection becomes more active. Early investigators estimated the average survival
with tubercular plaque-ridden brain disease was approximately seven years [63]. Most people who have Alzheimer’s disease die within
eight years of their diagnosis—typically from infection or pulmonary involvement, not cognitive failure.
In general, mycobacteria are extremely difficult to isolate, their preferred form being their multi-shaped cell-wall-deficient forms,
which through evolution the disease has learned to successfully adapt to inclement conditions in our body as a survival mechanism
[64]. Yet most labs in the United States are ill-equipped to detect such forms, even using sophisticated diagnostics such as next-generation
PCRs. But just as it is the nature of certain diseases of man, especially insidious ones like Alzheimer’s, to not always offer a crystal
clear path leading to their causal discovery –by the same token such pathogens cannot help but give off helpful biomedical clues along
their destructive path.
References
- Tricco AC., et al. “Comparative Effectiveness and Safety of Cognitive Enhancers for Treating Alzheimer's Disease: Systematic Review and Network Meta-analysis”. Journal of the American Geriatrics Society66.1 (2018):170-178.
- PL Detail-Document, Pharmacotherapy and Advancing Alzheimer’s disease. Pharmacist’s Letter/Prescriber’s Letter (2012).
- D’Intino G., et al. “Cognitive deficit associated with cholinergic and nerve growth factor down-regulation in experimental allergic encephalomyelitis in rats”. PNAS Proceedings of the National Academy of Sciences 102.8 (2005): 3070-3075
- Levite M. “Glutamate, T cells and multiple sclerosis”. Journal of Neural Transmission124.7 (2017): 775-798.
- A Billiau and P Matthys. “Modes of Action of Freund’s Adjuvants in Experimental Models of Autoimmune Diseases”. Journal of Leukocyte Biology70.6 (2001): 849-860.
- P Zwadyk., et al. “Rendering of Mycobacteria Safe for Molecular Diagnostic Studies and Development of a Lysis Method for Strand Displacement amplification and PCR”. Journal of Clinical Microbiology32.9 (1994): 2140–2146.
- P Bemer-Melchior and HB Drugeon. “Inactivation of Mycobacterium Tuberculosis for DNA Typing Analysis”. Journal of Clinical Microbiology37.7 (1999): 2350–2351.
- AP Lysenko., et al. “The Tuberculin Skin Test: How Safe Is Safe?—The Tuberculins Contain Unknown Forms Capable of Reverting to Cell-Wall-Deficient Mycobacteria”. Clinical and Experimental Medical Sciences2.2 (2014): 55–73.
- CA Janeway Jr. “Approaching the Asymptote? Evolution and Revolution in Immunology”. Cold Spring HarborSymposia on Quantitative Biology54 (1989): 1–13.
- RG White. “Role of Adjuvants in the Production of Delayed Hypersensitivity”. British Medical Bulletin23.1 (1967): 39–45.
- A Schneider “Hyperphosphorylation and Aggregation of Tau in Experimental Autoimmune Encephalomyelitis”. The Journal of Biological Chemistry 279.53 (2004): 55833–55839.
- Fischer. “Miliare Nekrosen mit drusigen Wucherungen der Neurofibrillen, eine regelmässige Veränderung der Hirnrinde bei seniler Demenz”. European Neurology 22.4 (1907): 361-372.
- V Babes and C et Levaditi. “On the Actinomycotic Shape of the Tuberculosis Bacilli” (“Sur la Forme Actinomycosique du Bacilli de la Tuberculosis”). In Arch. of Med. Exp. et D’anat, part 2, 9.6 (1897): 1041–1048.
- Guy CS., et al. “Identification of the anti-mycobacterial functional properties of piperidinol derivatives”. British Journal of Pharmacology 174.14 (2017): 2183-2193.
- Badawneh M and Aljamal J. “Synthesis And Antitubercular Activity Of Piperidine And Morpholine 1, 8 Naphthyridine Analogues”. International Journal of Pharmacy and Pharmaceutical Sciences 8.12 (2016): 252-257.
- Weis R., et al. “Antimycobacterial and H1-antihistaminic activity of 2-substituted piperidine derivatives”. Bioorganic & Medicinal Chemistry 16.24 (2008):10326-10331.
- Naik M., et al. “4-Aminoquinolone Piperidine Amides: Noncovalent Inhibitors of DprE1 with Long Residence Time and Potent Antimycobacterial Activity”. Journal of Medicinal Chemistry 57.12 (2014): 5419–5434
- Sellers KJ., et al. “Amyloid β synaptotoxicity is Wnt-PCP dependent and blocked by fasudil”. Alzheimer’s & Dementia 14.3 (2018): 306-317.
- Mahon RN and Hafner R. “Immune Cell Regulatory Pathways Unexplored as Host-Directed Therapeutic Targets for Mycobacterium tuberculosis: An Opportunity to Apply Precision Medicine Innovations to Infectious Diseases”. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America61(Suppl 3) (2015): S200-S216.
- Iizuka T., et al. “Preventive Effect of Rifampicin on Alzheimer Disease Needs at Least 450 mg Daily for 1 Year: An FDG-PET Follow-Up Study”. Dementia and Geriatric Cognitive Disorders EXTRA 7.2 (2017): 204-214.
- Yulug B., et al. “Therapeutic role of rifampicin in Alzheimer's disease”. Psychiatry and Clinical Neurosciences 72.3 (2018): 152-159.
- De Beer FC., et al. “Serum Amyloid A-Protein and C-reactive protein Levels in Pulmonary Tuberculosis: Relationship to Amyloidosis”. Thorax 30.3 (1984):196–200.
- Tomiyama T., et al. “Rifampicin prevents the aggregation and neurotoxicity of amyloid beta protein in vitro”. Biochemical and Biophysical Research Communications 204.1 (1994): 76-83.
- Li J., et al. “Rifampicin inhibits alpha-synuclein fibrillation and disaggregates fibrils”. Chemistry & Biology 11.11 (2004): 1513-1521.
- Qosa H., et al. “Enhanced Brain Amyloid-β Clearance by Rifampicin and Caffeine as a Possible Protective Mechanism against Alzheimer’s Disease”. Journal of Alzheimer’s disease 31.1 (2012): 151-165.
- Broxmeyer L. “Are the Infectious Roots of Alzheimer’s Buried Deep in the Past?” Journal of MPE Molecular Pathological Epidemiology3.S2.2 (2017): 1-19.
- Broxmeyer L. “Dr. Oskar Fischer’s Curious Little Alzheimer’s Germ”. Current Opinions in Neurological Science 1.3 (2017): 160-178.
- Broxmeyer L. “Alzheimer's disease –How Its Bacterial Cause Was Found and Then Discarded”. Create Space Independent Publishing Platform (2016): 190
- Umeda T., et al. “Rifampicin is a candidate preventive medicine against amyloid-β and tau oligomers”. Brain 139 (2016):1568–1586.
- Mosconi L., et al. “Pre-Clinical Detection of Alzheimer’s Disease Using FDG-PET, with or without Amyloid Imaging”. Journal of Alzheimer’s disease : JAD 20.3 (2010): 843-854.
- Alzheimer’s Association. “2016 Alzheimer’s disease facts and figures”. Alzheimer's & Dementia12.4 (2016): 459–509.
- Iizuka T and Kameyama M. “Cholinergic enhancement increases regional cerebral blood flow to the posterior Cingulate cortex in mild Alzheimer’s disease”. Geriatrics & Gerontology International17.6 (2017): 951-958.
- Iizuka T and Kameyama M: “Cingulate island sign on FDG-PET is associated with medial temporal lobe atrophy in Dementia with Lewy bodies”. Annals of Nuclear Medicine30.6 (2016): 421–429.
- Molloy DW., et al. “A multicenter, blinded, randomized, factorial controlled trial of doxycycline and rifampin for treatment of Alzheimer’s disease: the DARAD trial”. International Journal of Geriatric Psychiatry 28.5 (2013): 463–470.
- Sperling RA., et al. “Testing the right target and right drug at the right stage”. Science Translational Medicine 3.111 (2011): 111cm33.
- Okuda M., et al. “Design and synthesis of curcumin derivatives as tau and amyloid ß dual aggregation inhibitors”. Bioorganic & Medicinal Chemistry Letters26.20 (2016): 5024-5028.
- Potter PE. “Curcumin: a natural substance with potential efficacy in Alzheimer’s disease”. Journal of Experimental Pharmacology 5 (2013): 23-31.
- D Ghosh., et al. “Clinical Aspects of Functional Foods and Nutraceuticals”. CRC Press/Taylor and Francis Group (2014).
- Parasuraman Aiya Subramani., et al. “Curcumin Nanotechnologies and Its Anticancer Activity”. Nutrition and Cancer 69.3 (2017): 381-393.
- Schraufstätter E and Brent H. “Antibacterial action of curcumin and related compounds”. Nature 164 (1949): 456–457.
- Xiyuan Bai., et al. “Curcumin enhances human macrophage control of Mycobacterium tuberculosis infection”. Respirology21.5 (2016): 951-957.
- Masoumi A., et al. “1alpha, 25-dihydroxyvitamin D3 interacts with curcuminoids to stimulate amyloid-beta clearance by macrophages of Alzheimer's disease patients”. Journal of Alzheimer's disease 17.3 (2009): 703-717.
- Nafar F and Mearow KM. “Coconut oil attenuates the effects of amyloid-β on cortical neurons in vitro”. Journal of Alzheimer's disease 39.2 (2014): 233-237.
- Fernando WM., et al. “The role of dietary coconut for the prevention and treatment of Alzheimer's disease: potential mechanisms of action”. British Journal of Nutrition114.1 (2015): 1-14.
- Hu Yang I., et al. “Coconut Oil: Non-Alternative Drug Treatment against Alzheimer´s Disease”. Nutrición Hospitalaria 32.6 (2015): 2822-2827.
- Cabardo J. “Glycerol Monolaurate (GML) in Virgin Coconut Oil destroys tuberculosis bacteria”. Ad Veritatem: Research Journal of the UST Graduate School 8.1 (2008).
- Helmer Catherine., et al. “Herpes simplex virus, anti-herpetic medication, and dementia: Results from the three-city population-based cohort”. Alzheimer’s & Dementia 11.7 (2015): 153.
- Tzeng NS., et al. “Anti-herpetic Medications and Reduced Risk of Dementia in Patients with Herpes Simplex Virus Infections- a Nationwide, Population-Based Cohort Study in Taiwan”. Neurotherapeutics15.2 (2018): 417-429
- Grüter W. Experimentelle und klinische Untersuchungen über den sogenannten Herpes corneae. Berichte über die Versammlung des Ophthalmologischen Gesellschaft. (Heidelberg) 42 (1912): 162.
- Löwenstein A. “Aetiologische Untersuchungen über den fieberhaften Herpes”. Münchener medizinische Wochenschrift 66 (1919): 769-770.
- Kooy JM. “Virus of febrile herpes”. American Journal of Ophthalmology 66 (1921): 75.
- Kruszewska H., et al. “Search of antimicrobial activity of selected non-antibiotic drugs”. Acta Poloniae Pharmaceutica 59.6 (2002):436-439.
- Pochet S., et al. “Comparative study of purine and pyrimidine nucleoside analogues acting on the thymidylate kinases of Mycobacterium tuberculosis and of humans”. ChemBioChem 4.8 (2003): 742-747.
- Ferrari V and Serpi M. “Nucleoside analogs and tuberculosis. New weapons against an old enemy”. FutureMedicinal Chemistry7.3 (2015): 291-314
- Harrison FE. “A critical review of vitamin C for the prevention of age-related cognitive decline and Alzheimer's disease”. Journal of Alzheimer's disease29.4 (2012): 711-726.
- Vilchèze C., et al. “Mycobacterium tuberculosis is extraordinarily sensitive to killing by a vitamin C-induced Fenton reaction”. Nature communications 4 (2013): 1881
- Stone, Irwin. The Healing Factor- "Vitamin C" Against Disease. Grosset & Dunlap Publishers. New York (1972).
- Naidu KA. “Vitamin C in human health and disease is still a mystery? An overview”. Nutrition Journal 2 (2003): 7.
- Jordal PB., et al. “Widespread Abundance of Functional Bacterial Amyloid in Mycolata and Other Gram-Positive Bacteria”. Applied and Environmental Microbiology 75.12 (2009): 4101-4110.
- Broxmeyer L and Perry G. “Alzheimer’s: Do Anti-Herpetics Reduce the Risk of Dementia and if so, Why?” Current Opinions in Neurological Science 2.6 (2018): 594-598.
- JL Fox. “TB: A Grim Disease of Numbers”. ASM News 56 (1990): 363–364.
- CJ Alteri., et al. “Mycobacterium Tuberculosis Produces pili during Human Infection”. Proceedings of the National Academy of Sciences 104.12 (2007): 5145–5150.
- Chantemesse A. Ėtude Sur la Méningite Tuberculeuse de L’adulte. Les Formes Anormales en Particulier These de Medecine de Paris 124 (1884).
- Mattman LH. “Cell wall-deficient forms of mycobacteria”. Annals of the New York Academy of Sciences174.2 (1970): 852-861.
Citation:
Lawrence Broxmeyer. “Alzheimer’s FDA Approved Drugs & Alternatives: What are They Really Treating?” Current Opinions in
Neurological Science 3.1 (2018): 613-623. DOI: 10.5281/zenodo.2247890.
Copyright: © 2018 Lawrence Broxmeyer. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.






























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.