Review Article
Volume 4 Issue 1 - 2019
Dementia with Lewy Bodies: A Review
Professor, International Institute of Medicine & Science, California, U.S.A
*Corresponding Author: Alain L Fymat, Professor, International Institute of Medicine & Science, California, U.S.A.
Received: July 12, 2019; Published: July 24, 2019
Abstract
Dementia with Lewy bodies (DLB) is a progressive, neurodegenerative disorder in which abnormal deposits of a protein called alpha-synuclein build up in multiple areas of the brain. The abnormal protein deposits were discovered in 1912 by Frederic Lewy after whom the disease is named. The disease is accompanied by changes in cognition, behavior, and movement. Memory loss is not always an early symptom, in distinction with Alzheimer's disease (AD). A core feature is REM sleep behavior disorder (RSD) in which individuals lose normal muscle paralysis during REM sleep and act out their dreams. RSD appears years or decades before other symptoms. Other frequent symptoms include visual hallucinations; marked fluctuations in attention or alertness; and slowness of movement, trouble walking or rigidity. The autonomic nervous system is also usually affected. These symptoms are joined in the later course of the disease by Parkinsonism with slowness, stiffness and other symptoms similar to PD. The exact cause of the disease is unknown; it is not usually inherited, but there is a genetic association in a small number of families. In this article, I review the classification of the disease; its signs and symptoms; its risk factors and potential causes; and its pathophysiology. I also cover at some length the approach to diagnosis, including the updated criteria, clinical features and their supportive factors; the biomarkers utilized and the associated screening and imaging tests. The pharmacological and non-pharmacological management of these symptoms and risk factors are addressed with particularity on cognitive, motor and neuropsychiatric symptoms. Current research in the field is also succinctly summarized.
Keywords: Atypical parkinsonian syndrome; Blood brain barrier; Brain Inflammation; Lewy bodies dementia; Dementia with Lewy bodies; Alzheimer's disease dementia; Corticobasal palsy; Corticobasal syndrome; Multiple system atrophy; Neuroleptic malignant syndrome; Parkinson's disease; Parkinson's disease dementia; Progressive supranuclear palsy; REM; REM sleep behavior disorder; Restless legs syndrome; Vascular dementia
Abbreviations: AchEI: Acetylcholinesterase inhibitors; AD: Alzheimer's Disease; ANS: Autonomous Nervous System; ApoE: Apolipoprotein E; APS: Atypical Parkinsonian Syndrome; BBB: Blood brain barrier; CBP: Corticobasal Palsy; CBS: Corticobasal Syndrome; CNS: Central Nervous System; CSF: Cerebrospinal Fluid; CT: Computed Tomography; DCFS: Dementia Cognitive Fluctuation Scale; DLB: Dementia with Lewy Bodies; EEG: Electroencephalogram; ERS: Endoplasmic Reticulum Stress; FDA: (U.S.) Food & Drug Administration; IRSBDI: Innsbruck REM Sleep Behavior Disorder Inventory; LBD: Lewy Body Dementias; MCA: Montreal Cognitive Assessment; MIBG: Metaiodobenzylguanidine; MMSE: Mini-Mental State Examination; MRI: Magnetic Resonance Imaging; MSA: Multiple System Atrophy; NINDS: (U.S.) National Institute for Neurological Disorders and Stroke; NMS: Neuroleptic Malignant Syndrome; PD: Parkinson's disease; PDCRS: Parkinson's Disease Cognitive Rating Scale; PDD: Parkinson's Disease Dementia; PET: Positron Emission Tomography; PNS: Peripheral Nervous System; PSP: Progressive Supranuclear Palsy; REM: Rapid Eye Movement; RLS: Restless Legs Syndrome; RSBDSQ: REM Sleep Behavior Disorder Screening Questionnaire; RSD: REM Sleep behavior Disorder; RSDSQC: RSD Single-Question Screen; SOPD-C: Scales for Outcomes in Parkinson's Disease – Cognition; SPECT: Single Photon Emission Computed Tomography; VD: Vascular Dementia.
Disorders Mentioned: Alzheimer disease; Apathy; Atypical parkinsonian syndrome; Autosomal-lysomal autophagy system dysfunction; Capgras delusion; Corticobasal palsy; Corticobasal syndrome; Delirium; Delusions; Dementia with Lewy bodies; Depression; Endoplasmic reticulum stress; Exhaustion; Hallucinations; Hyper/hypo/pansomnia; Lewy body dementias; Inflammation; Mitochondrial dysfunction; Multiple system atrophy; Neuroleptic malignant syndrome; Parkinson's disease; Parkinson's disease dementia; Parkinsonism; Prion-like infection; Progressive Supranuclear Palsy; Psychosis; REM sleep behavior disorder; Restless legs syndrome; Synucleinopathies; Sleep apnea; Stroke; Vascular dementia.
Drugs Listed: Agitation; Anesthetics; Antidepressants (SSRIs, SNRIs, tricylics, MAOIs); Antipsychotics; Armodafinil (an anti-daytime sleepiness); Aromaxetine (for autonomic symptoms; orthostatic hypotension); Benzodiazepine; Beta blockers; Clonazepam (anti-sleep disorder); Clozapine (anti-hallucinations); Dextroamphetamine (a stimulant); Diphenhydramine (Benadryl); Donepezil; Dopamine agonists; Droxidona; Fludrocortisone; Levodopa; MAOI; Melatonin (for sleep disorders); Memantine; Methylphenidate (a stimulant); Midrodrine (for autonomic symptoms; orthostatic hypotension); Mirtazapine (for hypersomnia); Modafinil (for hypersomnia); Neuroleptics; Quetiapine; Rivastigmine (Excelon); SNRIs; SSRIs; Stools softeners; Tramadol; Zolpidem.
Introduction
Dementia with Lewy bodies (DLB) is a progressive, neurodegenerative disorder in which abnormal deposits of a protein called alpha-synuclein build up in multiple areas of the brain. It typically begins after the age of 50, and about 0.4% of people over 65 are affected. The abnormal protein deposits were discovered in 1912 by Frederic Lewy after whom the disease is named. DLB is accompanied by changes in cognition, behavior, and movement. Memory loss is not always an early symptom, in distinction with Alzheimer's disease (AD). A core feature is REM sleep behavior disorder (RSD) in which individuals lose normal muscle paralysis during REM sleep and act out their dreams RSD appears years or decades before other symptoms. Other frequent symptoms include visual hallucinations; marked fluctuations in attention or alertness; and slowness of movement, trouble walking or rigidity. The autonomic nervous system is usually affected, resulting in changes in blood pressure, heart and gastrointestinal function with constipation as an ordinary symptom. Mood changes such as depression and apathy are also common. These symptoms are joined in the later course of the disease by Parkinsonism with slowness, stiffness and other symptoms similar to PD. The exact cause of the disease is unknown, but the mechanism involves widespread deposits of abnormal clumps of Lewy bodies in neurons, as well as Lewy neurites. DLB is not usually inherited, but there is a genetic association in a small number of families.
In this article, following a brief historical and contextual background, I will review the classification of the disease; its signs and symptoms; its risk factors and potential causes; and its pathophysiology. I will cover at some length the approach to diagnosis including the updated criteria, clinical features and their supportive factors; the biomarkers utilized and the associated screening and imaging tests. Since the cause of the disease remains unknown, I will limit my considerations to pharmacological and non-pharmacological management of the extensively known symptoms with particularity on cognitive, motor and neuropsychiatric symptoms. I will subsequently address the disease from the patient's point of view, including prognosis after diagnosis, caregiving, and will lastly provide a vista on current research [1-10].
A Brief History of the Disease
Frederic Henry Lewey (born Friedrich Heinrich Lewy, 1885-1950), a prominent German-born American neurologist, is best known for the discovery of the Lewy “bodies” (or “body inclusions”) - a characteristic indicator of Parkinson's disease (PD) and dementia with Lewy bodies (DLB). In 1912, studying PD (paralysis agitans), he described findings of these inclusions in the vagus nerve, the nucleus basalis of Meynert and other brain regions. In 1923, he published a book, The Study on Muscle Tone and Movement Including Systematic Investigations on the Clinic, Physiology, Pathology, and Pathogenesis of Paralysis agitans and, except for one brief paper a year later, he never mentioned his findings again.
Frederic Henry Lewey (born Friedrich Heinrich Lewy, 1885-1950), a prominent German-born American neurologist, is best known for the discovery of the Lewy “bodies” (or “body inclusions”) - a characteristic indicator of Parkinson's disease (PD) and dementia with Lewy bodies (DLB). In 1912, studying PD (paralysis agitans), he described findings of these inclusions in the vagus nerve, the nucleus basalis of Meynert and other brain regions. In 1923, he published a book, The Study on Muscle Tone and Movement Including Systematic Investigations on the Clinic, Physiology, Pathology, and Pathogenesis of Paralysis agitans and, except for one brief paper a year later, he never mentioned his findings again.
Half-a-century later, in 1961, Okazaki., et al. published an account of “diffuse” Lewy-type inclusions associated with dementia in two autopsied cases. Later, in 1976, DLB was fully recognized and described in an autopsied case by Japanese psychiatrist and neuropathologist Kenji Kosaka. Four years later, or some thirty years after Lewy's death, Kosaka first proposed the term Lewy body disease (LBD) based on 20 autopsied cases. LBD was then thought to be rare until it became easier to diagnose in the 1980s following the discovery of the alpha-synuclein immunostaining procedure that highlighted the Lewy bodies in post mortem brains. Kosaka., et al. described thirty-four more cases in 1984, bringing attention of the Japanese work to the Western world. These were followed by four U.K. cases by Gibb., et al. in 1987 in the journal Brain, A year later, Burkhardt., et al. published the first general description of diffuse DLB.
In the 1980s, the Japanese, U.K., and U.S. research findings showed that DLB was a common dementia. There were nonetheless no generally accepted diagnostic guidelines - each group using a different terminology. The different research groups began to realize that a collaborative approach was needed if research was to advance. The Lewy Body Dementia Consortium was then formed. In 1996, the term dementia with Lewy bodies was agreed upon and the first criteria for diagnosing it were set forth.
Two 1997 discoveries highlighted the importance of Lewy body inclusions in neurodegenerative processes: (1) a mutation in the SNCA gene that encodes the alpha-synuclein protein was found in kindreds with Parkinson's disease, and (2) Lewy bodies and neurites were found to be immunoreactive for alpha-synuclein. Thus, alpha-synuclein aggregation as the primary building block of the synucleinopathies was established.
Between 1995 and 2005, the LBD Consortium issued three Consensus Reports on the disease. Further, DLB was included in the fourth text revision of the Diagnostic & Statistical Manual (DSM-IV-TR, published in 2000) and also in DSM-V under the heading "Dementia due to other general medical conditions”. In the 2010s, the possibility of a genetic basis began to emerge. The Fourth Consensus Report was issued in 2017, giving increased diagnostic weighting to RSD and Iodine-123 MIBG myocardial scintigraphy.
Affected Areas of the Brain
Areas of the brain that are affected by the disease and the associated functions are the:
Areas of the brain that are affected by the disease and the associated functions are the:
- Cerebral cortex: Thought, perception and language;
- Cortex: Emotions and behavior;
- Hippocampus: Memory;
- Midbrain and substantia nigra: Movement;
- Brainstem: Sleep, alertness, and autonomic dysfunction;
- Hypothalamus: Autonomic dysfunction;
- Olfactory cortex: Smell.
Also affected are the:
- Spinal cord and the peripheral nervous system (PNS): Autonomic dysfunction.
Disease Classification
Together with PD, multiple system atrophy (MSA), and other rarer brain conditions, DLB is classified as an atypical parkinsonian syndrome (APS). It is one of the synucleopathies – those neurodegenerative diseases that are due to an abnormal accumulation of the alpha-synuclein protein in the brain. This is illustrated in the flowchart of Figure 1:
Signs and Symptoms
The alpha-synuclein protein deposits that cause DLB can damage different regions of the nervous system, including multiple brain regions in the central nervous system (CNS), as well as the autonomic nervous system. (ANS). (At the end of this article, I provide a Sidebar for those readers who may be interested in better understanding how the various divisions of the vertebrate nervous system are integrated with each other and work synergistically.) This widespread damage can affect several domains of functioning, resulting in a disease that is more complex than many other dementias with widely varying symptoms. The symptoms of DLB are divided into a prodromal phase and essential, core, and supportive features [11-18].
- Prodromal phase: It can be identified with earliest signs being constipation and dizziness from autonomic dysfunction, reduced ability to smell, visual hallucinations, and REM sleep behavior disorder (RSD). It is distinguishable from Alzheimer's prodromal phase.
- Essential features: According to the 2017 Fourth Consensus Report of the LBD Consortium, dementia is diagnosed when a "progressive cognitive decline of sufficient magnitude to interfere with normal social or occupational functions, or with usual daily activities exists”. While dementia is an essential feature, it does not always appear early on with DLB, and is more likely to present as the condition progresses - typically after age 55. In contrast with AD, memory loss is not always noticeable in the early stages. There, the hippocampus is among the first brain structures affected and “episodic” memory loss that is related to the encoding of memories is typically the earliest symptom. Memory impairment occurs later in DLB; further, it has a different progression from AD because frontal structures are involved earlier, with later involvement of temporo-parietal brain structures. Deterioration in memory function occurs because new memories may be encoded, but not retrieved. “Verbal” memory is not as severely affected as in AD.
- Core features: While specific symptoms may vary, core features are based on "diagnostic specificity and the volume of good-quality evidence available" as designated by the 2017 LBD Consortium. They are:
- Fluctuating cognition, alertness or attention;
- REM sleep behavior disorder (RSD);
- One or more of the cardinal features of parkinsonism, not due to medication or stroke; and
- Repeated visual hallucinations.
Let me elaborate on these several symptoms. Impaired attention, executive function, and visuospatial function are common symptoms that are present early in the course of the disease. Individuals with DLB may be easily distracted, have a hard time focusing on tasks, or appear to be "delirium-like", "zoning out", or in states of altered consciousness. They may have disorganized speech and their ability to organize their thoughts may change during the day. Problems with executive function surface in activities requiring planning and organizing. Deficits can present with impaired job performance, difficulty multitasking, inability to follow conversations, or problems driving a car—such as misjudging distances or becoming lost. A person with DLB may experience problems of wakefulness or sleep disorders including daytime sleepiness, drowsiness or napping more than two hours a day, insomnia, and restless legs syndrome (RLS).
REM sleep behavior disorder (RSD) is a parasomnia in which individuals lose the paralysis of muscles (atonia) that is normal during rapid eye movement sleep, and act out their dreams or have other abnormal movements or vocalizations. About 80% of people with DLB have RSD. Abnormal sleep behaviors may begin before cognitive decline is observed, and may appear decades before any other symptoms, often as the first clinical indication of DLB and an early sign of a synucleinopathy. On autopsy, 94%-98% of individuals with polysomnography-confirmed RSD are found to have a synucleinopathy—most commonly DLB or PD. Other symptoms of the specific synucleinopathy usually manifest within 15 years of the diagnosis of RSD, but may emerge up to 50 years after RSD diagnosis. A confounding factor is that RSD may subside over time.
Individuals with RSD may not be aware that they act out their dreams; a sleeping partner may be the only person who knows of these behaviors. Such behaviors may include yelling, screaming, laughing, crying, unintelligible talking, nonviolent flailing, or more violent punching, kicking, choking, or scratching. The reported dream enactment behaviors are frequently violent and involve a theme of being chased or attacked. People with RSD may fall out of bed or injure themselves or their bed partners, with possible resulting bruises, fractures, or subdural hematomae. However, because people are more likely to remember or report violent dreams and behaviors—and to be referred to a specialist when injury occurs – recall or selection bias may explain the prevalence of violence reported in RSD.
Parkinsonism occurs in more than 85% of people with DLB, who may have one or more of the cardinal features that occur in PD, slowness of movement, rigidity and, less commonly, tremor at rest. Motor symptoms may include shuffling gait, problems with balance, falls, blank expression, reduced range of facial expression, and low speech volume or a weak voice. Presentation of motor symptoms is variable, but they are usually symmetric, presenting on both sides of the body. For a DLB diagnosis, only one of the cardinal symptoms of Parkinsonism need be present, and the symptoms may be less severe than in persons with PD.
Visual hallucinations are defined as "well-formed images perceived without the presence of real stimuli". Up to 80% of people with DLB have visual hallucinations, typically early in the course of the disease. They are recurrent and frequent; may be scenic, elaborate and detailed; and usually involve animated perceptions of animals or people, including children and family members. These hallucinations can sometimes provoke fear, although their content is more typically neutral. In some cases, the person with DBL may have insight that the hallucinations are not real. Individuals with visual hallucinations generally have more severe cognitive impairment. Among those with more disrupted cognition, the hallucinations can become more complex, and they may be less aware that their hallucinations are not real. Different from visual hallucinations, but also common in DLB, are visual misperceptions or illusions. Persons with DLB may have problems with vision (for example, at night) and misinterpret what they see (for example, mistaking a pile of socks for snakes or a clothes closet for the bathroom).
Supportive features: According to the LBD Consortium, although "carrying less diagnostic weight, supportive items are often valuable in clinical decision-making, acting as signposts to or adding evidence for a DBL diagnosis". They may persist over time, be present early in the progression of the disease, and are common but not specific to the diagnosis. The supportive features are:
- Marked sensitivity to antipsychotics (neuroleptics);
- Marked dysautonomia (autonomic dysfunction) in which the autonomic nervous system does not work properly;
- Hallucinations in senses other than vision (hearing, touch, taste, and smell);
- Hypersomnia (excessive sleepiness);
- Hyposomnia (reduced ability to sleep);
- False beliefs and delusions organized around a common theme;
- Postural instability, loss of consciousness, and frequent falls;
- Apathy, anxiety and depression.
Again, I will elaborate further on these several supportive factors. Marked sensitivity to antipsychotics can occur. Antipsychotic treatment of people with DLB results in adverse reactions about half of the time, and can be fatal after a single dose. The Fourth Consensus Report continues to caution against the use of antipsychotics (neuroleptics) for people with DLB. Partly because of dopamine cell loss, people with DLB may have neuroleptic malignant syndrome (NMS), impairments in cognition or alertness, or irreversible exacerbation of Parkinsonism including severe rigidity and dysautonomia from the use of antipsychotics. Antipsychotic medications with D2 dopamine receptor-blocking properties are used only with great caution.
Dysautonomia (autonomic dysfunction) occurs when Lewy pathology affects the peripheral nervous system (PNS) or the nerves that serve organs such as the intestines, heart, and urinary tract. The first signs of this dysfunction are often subtle. Symptoms include blood pressure problems, such as dizziness after quickly standing up and supine hypertension, constipation, urinary problems, and sexual dysfunction, loss of or reduced ability to smell; and excessive sweating, drooling, or salivation. Alpha-synuclein deposits can affect cardiac muscle and blood vessels. Almost all people with synucleinopathies have cardiovascular dysfunction, although most are asymptomatic; 50–60% of individuals with DLB have orthostatic hypotension due to reduced blood flow, which results in symptoms of lightheadedness, feeling faint, and blurred vision.
From chewing to defecation, alpha-synuclein deposits affect every level of gastrointestinal function. Almost all persons with DLB have upper gastrointestinal tract dysfunction (such as delayed gastric emptying) or lower gastrointestinal dysfunction (such as constipation and prolonged stool transit time). Persons with DLB almost universally experience nausea, gastric retention, or abdominal distention from delayed gastric emptying. Constipation can present a decade before diagnosis. Difficulty swallowing is milder than in other synucleinopathies, and presents later in the course of the disease. Urinary difficulties (urinary retention, waking at night to urinate, increased urinary frequency and urgency, and over- or under-active bladder) typically appear later and may be mild to moderate. Sexual dysfunction usually appears early in synucleinopathies, and may include erectile dysfunction and difficulties achieving orgasm or ejaculation.
Among the other supportive features, psychiatric symptoms are often present when the individual first comes to clinical attention. Compared to AD, they are more likely to cause more impairment. Anxiety and depression are more common in DLB than in the general population, and may have been present for decades. Anxiety leads to increased risk of falls, and apathy may lead to less social interaction. Agitation, behavioral disturbances, and delusions typically appear later in the course of the disease. Delusions may have a paranoid quality, revolving around themes like a house being broken into, infidelity, or abandonment. Individuals with DLB who misplace items may have delusions centered around theft. Capgras delusion may occur, where the person with DLB loses knowledge of the spouse, caregiver, or partner's face, and is convinced that an imposter has replaced the caregiver. Hallucinations in other modalities are sometimes present, but are less frequent. Sleep disorders are common in DLB and may lead to hypersomnia; these include disrupted sleep cycles, sleep apnea, and arousal from periodic limb movement disorder. Loss of sense of smell may occur several years before other symptoms.
Medications (including tricyclic antidepressants and treatments for urinary incontinence) with anticholinergic properties that cross the blood brain barrier (BBB) can cause memory loss. Dopamine agonists may lead to psychosis. The antihistamine medication diphenhydramine (Benadryl), sleep medications like Zolpidem and benzodiazepines may worsen confusion or neuropsychiatric symptoms. Some general anesthetics may cause confusion or delirium upon waking and may result in permanent decline.
Risk Factors and Potential Causes of the Disease
As already stated, the exact cause of DLB -, indeed the cause of synucleinopathies—is not known, but many factors probably are present, including both genetic and environmental influences. Less than 10% of cases can be accounted for by inheritance of a single gene, while in most cases, genetic and environmental factors mediate other disease processes to cause synucleinopathies. These other processes include mitochonrial or autosomal-lysomal autophagy system dysfunction, inflammation, endoplasmic reticulum stress, or even prion-like infection [11-18].
As already stated, the exact cause of DLB -, indeed the cause of synucleinopathies—is not known, but many factors probably are present, including both genetic and environmental influences. Less than 10% of cases can be accounted for by inheritance of a single gene, while in most cases, genetic and environmental factors mediate other disease processes to cause synucleinopathies. These other processes include mitochonrial or autosomal-lysomal autophagy system dysfunction, inflammation, endoplasmic reticulum stress, or even prion-like infection [11-18].
The genetics are not well studied because the large homogeneous DLB populations needed for genetic studies have not been identified. DLB is not usually inherited, but there is a genetic association in a small number of families. The risk is heightened with inheritance of the ε4 allele of the apolipoprotein (ApoE) gene. Mutations in the PARK11 and GBA genes are associated with the disease. Further, genetic overlap exists among LBD, PD, PD dementia, and AD. Also, RSD is found more often among individuals who have GBA and SNCA mutations.
Having REM sleep behavior disorder, having another family member with DLB, and being 50 years of age and over are risk factors for developing DLB. Additional risk factors for rapid conversion of RSD to a synucleinopathy are impairments in color vision or the ability to smell, mild cognitive impairment, and abnormal dopaminergic imaging.
Pathophysiology
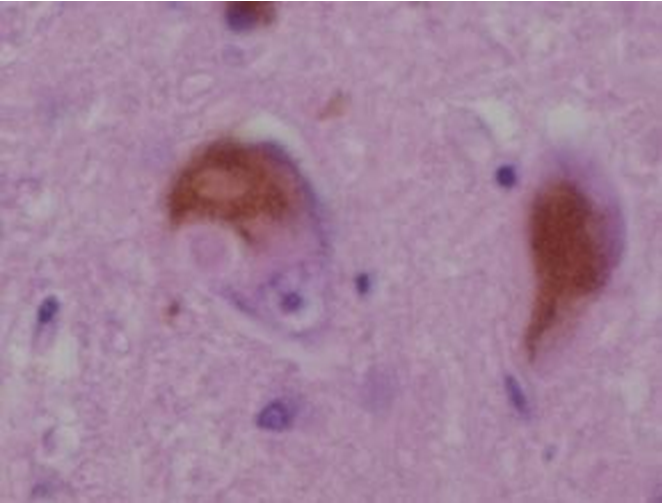
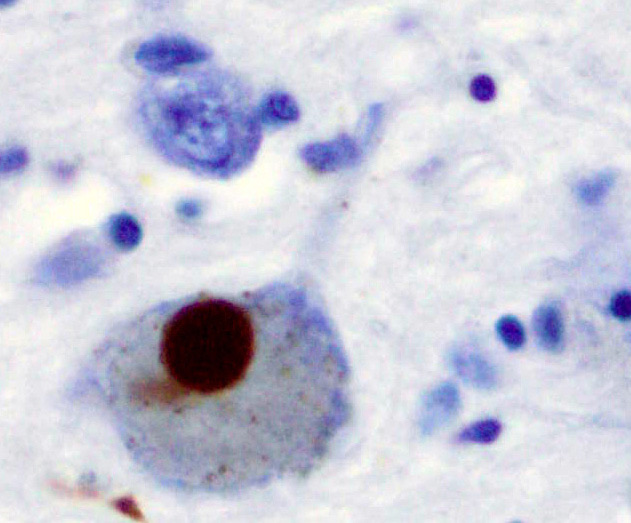
As already stated earlier, DLB is characterized by the development of abnormal collections of alpha-synuclein protein within neurons, known as Lewy bodies and Lewy neurites in diseased neurons. When these clumps of protein form in neurons, those neurons function less optimally and die. Damage in the brain is widespread, and affects many domains of functioning. Loss of acetylcholine-producing neurons is thought to account for degeneration in memory and learning, while the death of dopamine-producing neurons appears to be responsible for degeneration of behavior, cognition, movement, motivation, sleep, and mood. Figures 2 and 3 are micrographs of sections through the neocortex of deceased persons with Lewy bodies. They show brown immunostained alpha-synuclein in Lewy bodies (large clumps) and Lewy neurites (thread-like structures).
As already stated earlier, DLB is characterized by the development of abnormal collections of alpha-synuclein protein within neurons, known as Lewy bodies and Lewy neurites in diseased neurons. When these clumps of protein form in neurons, those neurons function less optimally and die. Damage in the brain is widespread, and affects many domains of functioning. Loss of acetylcholine-producing neurons is thought to account for degeneration in memory and learning, while the death of dopamine-producing neurons appears to be responsible for degeneration of behavior, cognition, movement, motivation, sleep, and mood. Figures 2 and 3 are micrographs of sections through the neocortex of deceased persons with Lewy bodies. They show brown immunostained alpha-synuclein in Lewy bodies (large clumps) and Lewy neurites (thread-like structures).
The amount of Lewy body pathology correlates with the extent of dementia in the Lewy body disorders, but amyloid imaging and autopsy studies indicate that tau protein pathology and amyloid plaques, which are hallmarks of Alzheimer's disease, are also common. Amyloid-beta (Aβ) deposits are found in tauopathies - neurodegenerative diseases characterized by neurofibrillary tangles or hyperphosphorylated tau proteins - but the mechanism underlying dementia is often mixed, and Aβ is also a factor in DLB.
The precise mechanisms contributing to DLB are not well understood, and a matter of some dispute. This is reminiscent of all neurodegenerative diseases. Nonetheless, in the case of Alzheimer's, this author has proposed that AD is a runaway autoimmune disease, a theory that remains the most attractive of all 20 theories advanced so far for that disease.
Further, that explanation accounts for the deep biology of the disease, provides a new vista on the disease and offers a path to a cure. The role played by the alpha-synuclein deposits is unclear because individuals with no signs of DLB have been found on autopsy to have advanced alpha-synuclein pathology. How Lewy pathology relates to dopaminergic degeneration and later to more widespread cell death has remained contentious. It is not known if the pathology spreads between cells or follows another pattern. The mechanisms that contribute to cell death, how the disease advances through the brain, and the timing of cognitive decline are all poorly understood; nor is there a model to account for the specific neurons and brain regions that are affected [19, 20].
A proposed pathophysiology for RSD implicates cells in the reticular formation that regulates REM sleep. These cells are affected before other brain regions in DLB; this could be a potential explanation for the appearance of RSD often decades earlier than other symptoms.
Diagnosis Criteria, Biomarkers, and Screen Tests
DLB can only be definitively diagnosed after death with an autopsy of the brain. For the living, the diagnosis is referred to as probable or possible. The disease is often misdiagnosed or confused in its early stages with AD. Another complicating factor is that DLB can occur along with AD. Autopsy may reveal previously undiagnosed Lewy bodies in as many as half of people with AD. Despite the difficulty in diagnosis, a prompt diagnosis is important because of the serious risks of sensitivity to antipsychotic medications and the need to inform both the person with DLB and the person's caregivers about potentially irreversible side effects of those medications. In comparison to many other neurodegenerative diseases, the management of DLB is difficult, so an accurate diagnosis is important [11-18].
DLB can only be definitively diagnosed after death with an autopsy of the brain. For the living, the diagnosis is referred to as probable or possible. The disease is often misdiagnosed or confused in its early stages with AD. Another complicating factor is that DLB can occur along with AD. Autopsy may reveal previously undiagnosed Lewy bodies in as many as half of people with AD. Despite the difficulty in diagnosis, a prompt diagnosis is important because of the serious risks of sensitivity to antipsychotic medications and the need to inform both the person with DLB and the person's caregivers about potentially irreversible side effects of those medications. In comparison to many other neurodegenerative diseases, the management of DLB is difficult, so an accurate diagnosis is important [11-18].
Criteria
The 2017 Fourth Consensus Report established diagnostic criteria for “probable” and “possible” DLB, in recognition of advances in detection and improvements from the earlier Third Consensus (2005 version). The 2017 criteria are based on essential, core, and supportive clinical features and diagnostic biomarkers [21-27].
The 2017 Fourth Consensus Report established diagnostic criteria for “probable” and “possible” DLB, in recognition of advances in detection and improvements from the earlier Third Consensus (2005 version). The 2017 criteria are based on essential, core, and supportive clinical features and diagnostic biomarkers [21-27].
Clinical features
The four core clinical features (discussed in the signs and symptoms section above) are:
The four core clinical features (discussed in the signs and symptoms section above) are:
- Fluctuating cognition;
- Visual hallucinations;
- REM sleep behavior disorder; and
- Parkinsonism.
Supportive clinical features are:
- Marked sensitivity to antipsychotics;
- Marked autonomic dysfunction;
- Nonvisual hallucination;
- Hypersomnia;
- Reduced ability to smell;
- False beliefs and delusions organized around a common theme;
- Postural instability;
- Loss of consciousness;
- Frequent falls;
- Apathy; and
- Anxiety or depression.
Diagnostic biomarkers
The diagnostic biomarkers are:
The diagnostic biomarkers are:
- Indicative: Reduced dopamine transporter uptake in the basal ganglia shown on PET or SPECT imaging; low uptake of Iodine 123 metaiodobenzylguanidine (I-123 MIBG) shown on myocardial scintigraphy; loss of atonia during REM sleep evidenced on polysomnography.
- Supportive:From PET, SPECT, CT or MRI brain imaging studies, or EEG monitoring: lack of damage to medial temporal lobe; reduced occipital activity; and prominent slow-wave activity.
On the above basis, probable or possible DLB can be diagnosed, thus:
- Probable DLB: It can be diagnosed when dementia and at least two core features are present, or one core feature with at least one indicative biomarker present.
- Possible DLB:It can be diagnosed when dementia and only one core feature are present or, if no core features are present, at least one indicative biomarker present.
Further,
- DLB is distinguished from Parkinson's disease dementia (PDD): (See Figure 1.) The distinction is aided by the time frame in which dementia symptoms appear relative to Parkinson symptoms. DLB is diagnosed when cognitive symptoms begin before or at the same time as Parkinsonism. PDD would be the diagnosis when Parkinson's disease is well established before the dementia occurs, that is, the onset of dementia is more than a year after the onset of parkinsonian symptoms.
- DLB is listed in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5): It is listed as a major or mild neurocognitive disorder with Lewy bodies. The differences between the DSM and DLB Consortium criteria are: (1) the DSM does not include low dopamine transporter uptake as a supportive feature, and (2) an unclear diagnostic weight is assigned to biomarkers in the DSM.
Clinical history and testing
Diagnostic tests can be used to establish some features of the condition and distinguish them from symptoms of other conditions. Diagnosis may include:
Diagnostic tests can be used to establish some features of the condition and distinguish them from symptoms of other conditions. Diagnosis may include:
- The person's medical history;
- Physical examination;
- Assessment of neurological function;
- Testing to rule out conditions that may cause similar symptoms;
- Brain imaging;
- Neuropsychological testing to assess cognitive function;
- Sleep studies; or
- Myocardial scintigraphy.
Laboratory testing can rule out conditions such as:
- Depression;
- Abnormal thyroid function;
- Syphilis;
- HIV; or
- Vitamin deficiencies
That may cause symptoms similar to dementia.
Dementia screening tests
Dementia screening tests include the:
- Mini-Mental State Examination (MMSE);
- Montreal Cognitive Assessment (MCA);
- and others such as the:
- Parkinson's Disease Cognitive Rating Scale” (PD CRS);
- Parkinson's Neuropsychometric Instrument” (PNDI); and
- Scales for Outcomes in Parkinson's Disease – Cognition (SOPD-C).
Tests of attention for simple screening include the:
- Digital span;
- Serial Sevens;
- Spatial span;
- Revised Digit Symbol Subtest of the Wechsler Adult Intelligence Scale that may show defects in attention that are characteristic of DLB;
- Frontal Assessment Battery;
- Stroop test; and
- Wisconsin Card Sorting test for evaluation of executive function; and
- Many other available screening instruments.
Questionnaires
Questionnaires such as the:
Questionnaires such as the:
- REM Sleep Behavior Disorder Screening Questionnaire (RSBDSQ);
- Mayo Sleep Questionnaire (MSQ);
- Innsbruck REM Sleep Behavior Disorder Inventory (IRSBDI)
Are well-validated.
Imaging tests
- DLB: If DLB is suspected when Parkinsonism and dementia are the only presenting features, PET or SPECT imaging tests may show reduced dopamine transporter activity. A DLB diagnosis may be warranted if other conditions with reduced dopamine transporter uptake can be ruled out.
- RSD: RSD is diagnosed either by sleep study recording or, when sleep studies cannot be performed, by medical history and validated questionnaires. In individuals with dementia and a history of RSD, a probable DLB diagnosis can be justified (even with no other core feature or biomarker); this would be based on a sleep study showing REM sleep without atonia because it is so highly predictive.
- Conditions similar to RSD: Conditions like severe sleep apnea and periodic limb movement disorder, must be ruled out. Prompt evaluation and treatment of RSD is indicated when a prior history of violence or injury is present as it may increase the likelihood of future violent dream enactment behaviors. Individuals with RSD may not be able to provide a history of dream enactment behavior, so bed partners are also consulted. The REM Sleep Behavior Disorder Single-Question Screen (RSD SQS) offers diagnostic sensitivity and specificity in the absence of polysomnography with one question: "Have you ever been told, or suspected yourself, that you seem to 'act out your dreams' while asleep (for example, punching, flailing your arms in the air, making running movements, etc.)?"
- Autonomic dysfunction:Since 2001, Iodine-123 metaiodobenzylguanidine (I-123 MIBG) myocardial scintigraphy has been used diagnostically in East Asia and Japan. MIBG is taken up by sympathetic nerve endings, such as those that innervate the heart, and is labeled for scintigraphy with radioactive Iodine-123. Autonomic dysfunction resulting from damage to nerves in the heart in patients with DLB is associated with lower cardiac uptake of I-23I MIBG.
Genetic testing
There is no genetic test to determine if an individual will develop DLB and, according to the Lewy Body Dementia Association, genetic testing is not routinely recommended because there are only rare instances of hereditary DLB.
There is no genetic test to determine if an individual will develop DLB and, according to the Lewy Body Dementia Association, genetic testing is not routinely recommended because there are only rare instances of hereditary DLB.
Differential testing
Many neurodegenerative conditions share cognitive and motor symptoms with dementia with DLB. The differential diagnosis includes Alzheimer's disease (AD); such synucleinopathies as Parkinson's disease dementia (PDD); Parkinson's disease (PD); multiple system atrophy (MSA); vascular dementia (VD); progressive supranuclear palsy (PSP); corticobasal palsy (CBP); degeneration; and corticobasal syndrome (CBS).
Many neurodegenerative conditions share cognitive and motor symptoms with dementia with DLB. The differential diagnosis includes Alzheimer's disease (AD); such synucleinopathies as Parkinson's disease dementia (PDD); Parkinson's disease (PD); multiple system atrophy (MSA); vascular dementia (VD); progressive supranuclear palsy (PSP); corticobasal palsy (CBP); degeneration; and corticobasal syndrome (CBS).
Lewy bodies pathology affects the peripheral autonomic nervous system (PANS). Autonomic dysfunction is observed less often in Alzheimer's, frontotemporal, or vascular dementias, so its presence can help differentiate them. Let me briefly review these several confounding factors:
- Alzheimer's disease: Short-term memory impairment is seen early in AD. It is a prominent feature, while fluctuating attention is uncommon. On the other hand, impairment in DLB is more often seen first as fluctuating cognition. While 74% of people with autopsy-confirmed DLB had deficits in planning and organization, they show up in only 45% of people with AD. Further, in AD, it is unclear if executive function is impacted early. Visuospatial processing deficits are present in most individuals with DLB, where they show up earlier and are more pronounced than in AD. Hallucinations typically occur early in the course of DLB, are less common in early AD, but usually occur later in AD. AD pathology frequently co-occurs in DLB, so the cerebrospinal fluid (CSF) testing for amyloid-beta and tau protein that is often used to detect AD is not useful in differentiating AD and DLB.
- PET or SPECT imaging can be used to detect reduced dopamine transporter uptake and distinguish AD from DLB. Severe atrophy of the hippocampus is more typical of AD than DLB. In East Asia and Japan, Iodine-123 MIBG is used in the differential diagnosis of DLB and AD, because reduced labeling of cardiac nerves is seen only in Lewy body disorders. Other indicative and supportive biomarkers are useful in distinguishing DLB and AD (preservation of medial temporal lobe structures, reduced occipital activity, and slow-wave EEG activity).
- Synucleinopathies:DLB and PDD are clinically similar after dementia occurs in PD. Delusions in PDD are less common than in DLB, and persons with PD are typically less caught up in their visual hallucinations than those with DLB. There is a lower incidence of tremor at rest in DLB than in PD; and signs of Parkinsonism in DLB are more symmetrical. In multiple system atrophy (MSA), autonomic dysfunction appears earlier and is more severe, and is accompanied by uncoordinated movements, while visual hallucinations and fluctuating cognition are less common than in DLB. Urinary difficulty is one of the earliest symptoms with MSA, and is often severe.
- Frontotemporal dementias: Corticobasal syndrome, corticobasal degeneration and progressive supranuclear palsy are frontotemporal dementias with features of parkinsonism and impaired cognition. Similar to DLB, imaging may show reduced dopamine transporter uptake. Motor movements in corticobasal syndrome are asymmetrical. There are differences in posture, gaze and facial expressions in the most common variants of progressive supranuclear palsy, and falling backwards is more common relative to DLB. Visual hallucinations and fluctuating cognition are unusual in corticobasal degeneration and progressive supranuclear palsy.
- Vascular dementia: MRI scans almost always show abnormalities in the brains of people with vascular dementia (VD), which can begin suddenly. The Dementia Cognitive Fluctuation Scale (DCFS) is a screening questionnaire used to measure fluctuating attention and alertness that has shown promise in distinguishing between DLB and VD but, as of 2014, it had not been fully validated.
- Other symptoms:The symptoms of DLB are easily confused with delirium, or more rarely as psychosis. Mismanagement of delirium is a particular concern because of the risks to people with DLB associated with antipsychotics. Dopamine transporter imaging can help distinguish DLB from delirium.
Disease Management - Pharmacological
Only palliative care can be offered, as there are no medications which will modify the course of the disease. No medications for DLB have been approved by the U.S. Food & Drug Administration (FDA) as of 2017, although Donepezil is licensed in Japan for the treatment of DLB. Management can be challenging because of the need to balance treatment of cognitive dysfunction, neuropsychiatric features, impairments related to the motor system, and other nonmotor symptoms. A multidisciplinary approach, going beyond early and accurate diagnosis to include educating and supporting the caregivers, is favored by the 2017 DLB Consortium.
Only palliative care can be offered, as there are no medications which will modify the course of the disease. No medications for DLB have been approved by the U.S. Food & Drug Administration (FDA) as of 2017, although Donepezil is licensed in Japan for the treatment of DLB. Management can be challenging because of the need to balance treatment of cognitive dysfunction, neuropsychiatric features, impairments related to the motor system, and other nonmotor symptoms. A multidisciplinary approach, going beyond early and accurate diagnosis to include educating and supporting the caregivers, is favored by the 2017 DLB Consortium.
Pharmacological management is complex because of adverse effects to medications and the wide range of symptoms to be treated (cognitive, motor, neuropsychiatric, autonomic, and sleep). Anticholinergic and dopaminergic agents can have adverse effects or result in psychosis in individuals with DLB, and a medication that addresses one feature might worsen another. For example, acetylcholinesterase inhibitors (AchEI) for cognitive symptoms can lead to complications in dysautonomia features; treatment of movement symptoms with dopaminergic agents may worsen neuropsychiatric symptoms; and treatment of hallucinations and psychosis with antipsychotics may worsen other symptoms or lead to a potentially fatal reaction.
Extreme caution is required in the use of antipsychotic medication in people with DLB because of their sensitivity to these agents. People with DLB who take neuroleptics are at risk for neuroleptic malignant syndrome (NLS), a life-threatening illness. There is little evidence to support the use of antipsychotics to treat the Lewy body dementias, and they carry the additional risk of stroke when used in the elderly with dementia [26-30].
Cognitive symptoms.
There is strong evidence for the use of AChEIs to treat cognitive problems. Donepezil was licensed in 2014 in Japan for the treatment of DLB, and Rivastigmine (Exelon) has a strong evidence base for treating cognitive symptoms. Even if the AChEIs do not lead to improvement in cognitive symptoms, people taking them may have less deterioration, although there may be adverse gastrointestinal effects. The AChEIs are initiated carefully as they may aggravate autonomic dysfunction or sleep behaviors. DLB may be more responsive than AD to Donepezil. There is less evidence for the efficacy of Memantine in DLB, but it may be used alone or with an AChEI because of its low side effect profile. Anticholinergic drugs are avoided because they worsen cognitive symptoms. To improve daytime alertness, there is mixed evidence for the use of stimulants such as Methylphenidate and Dextroamphetamine. Although worsening of neuropsychiatric symptoms is not common, they can increase the risk of psychosis. In the U.S., Modafinil and Armodafinil are not always covered by insurance, but may be effective for daytime sleepiness.
There is strong evidence for the use of AChEIs to treat cognitive problems. Donepezil was licensed in 2014 in Japan for the treatment of DLB, and Rivastigmine (Exelon) has a strong evidence base for treating cognitive symptoms. Even if the AChEIs do not lead to improvement in cognitive symptoms, people taking them may have less deterioration, although there may be adverse gastrointestinal effects. The AChEIs are initiated carefully as they may aggravate autonomic dysfunction or sleep behaviors. DLB may be more responsive than AD to Donepezil. There is less evidence for the efficacy of Memantine in DLB, but it may be used alone or with an AChEI because of its low side effect profile. Anticholinergic drugs are avoided because they worsen cognitive symptoms. To improve daytime alertness, there is mixed evidence for the use of stimulants such as Methylphenidate and Dextroamphetamine. Although worsening of neuropsychiatric symptoms is not common, they can increase the risk of psychosis. In the U.S., Modafinil and Armodafinil are not always covered by insurance, but may be effective for daytime sleepiness.
Motor symptoms
Motor symptoms appear to respond in DLB somewhat less to the medications used to treat Parkinson's disease, like Levodopa, and neuropsychiatric symptoms can be increased. If such medications are needed for motor symptoms, cautious introduction with slow increases to the lowest possible dose may help avoid psychosis.
Motor symptoms appear to respond in DLB somewhat less to the medications used to treat Parkinson's disease, like Levodopa, and neuropsychiatric symptoms can be increased. If such medications are needed for motor symptoms, cautious introduction with slow increases to the lowest possible dose may help avoid psychosis.
Neuropsychiatric symptoms
The first line of defense in reducing visual hallucinations is to reduce the use of dopaminergic drugs, which can worsen hallucinations. Although it has been shown effective in Parkinson's disease, there is limited evidence for the use of Clozapine to treat visual hallucinations in DLB, and its use requires regular blood monitoring. Quetiapine is relatively safe and well-tolerated for psychosis and agitation in DLB, but there is little evidence for its efficacy.
The first line of defense in reducing visual hallucinations is to reduce the use of dopaminergic drugs, which can worsen hallucinations. Although it has been shown effective in Parkinson's disease, there is limited evidence for the use of Clozapine to treat visual hallucinations in DLB, and its use requires regular blood monitoring. Quetiapine is relatively safe and well-tolerated for psychosis and agitation in DLB, but there is little evidence for its efficacy.
Apathy may be treated with AChEIs, and they may also reduce hallucinations and delusions. Reduction in anxiety and agitation may be a secondary effect. Mirtazapine and SSRIs can be used to treat depression, depending on how well they are tolerated, and guided by general advice for the use of antidepressants in dementia. Antidepressants can worsen RSD, and antidepressants with anti-cholinergic properties may worsen hallucinations and delusions. People with Capgras syndrome may not tolerate AChEIs.
Sleep disorders.
Injurious dream enactment behaviors are a treatment priority. RSD may be treated with Melatonin or Clonazepam. Melatonin may be more helpful in preventing injuries while offering a safer alternative because Clonazepam can produce deteriorating cognition and worsening of sleep apnea. For some people, Memantine or AChEIs are useful. No trials support its use in DLB, but Modafinil may be used for hypersomnia. Mirtazapine can be used for hypersomnia, but it can exacerbate RSD. Anti-depressants (SSRIs, SNRIs, tricyclics, and MAOIs), AchEIs, beta blockers, caffeine, and Tramadol may worsen RSD.
Injurious dream enactment behaviors are a treatment priority. RSD may be treated with Melatonin or Clonazepam. Melatonin may be more helpful in preventing injuries while offering a safer alternative because Clonazepam can produce deteriorating cognition and worsening of sleep apnea. For some people, Memantine or AChEIs are useful. No trials support its use in DLB, but Modafinil may be used for hypersomnia. Mirtazapine can be used for hypersomnia, but it can exacerbate RSD. Anti-depressants (SSRIs, SNRIs, tricyclics, and MAOIs), AchEIs, beta blockers, caffeine, and Tramadol may worsen RSD.
Autonomic symptoms.
Decreasing the dosage of dopaminergic or atypical antipsychotic drugs may be needed with orthostatic hypotension. High blood pressure drugs can sometimes be stopped. When non-pharmacological treatments for orthostatic hypotension have been exhausted, Fludrocortisone, Droxidopa, Midodrine or Aromoxetine are options.
Decreasing the dosage of dopaminergic or atypical antipsychotic drugs may be needed with orthostatic hypotension. High blood pressure drugs can sometimes be stopped. When non-pharmacological treatments for orthostatic hypotension have been exhausted, Fludrocortisone, Droxidopa, Midodrine or Aromoxetine are options.
Disease Management - Non-Pharmacological
There is no high-quality evidence for non-pharmacological management of DLB, but some interventions have been shown effective for addressing similar symptoms that occur in other dementias. Organized activities, music therapy, physical activity and occupational therapy may help with psychosis or agitation, while exercise and gait training can help with motor symptoms. Cognitive behavioral therapy can be tried for depression or hallucinations, although there is no evidence for its use in DLB. The frequency and severity of RSD may be lessened by treating sleep apnea, if it is present. Cues can be used to help with memory retrieval.
There is no high-quality evidence for non-pharmacological management of DLB, but some interventions have been shown effective for addressing similar symptoms that occur in other dementias. Organized activities, music therapy, physical activity and occupational therapy may help with psychosis or agitation, while exercise and gait training can help with motor symptoms. Cognitive behavioral therapy can be tried for depression or hallucinations, although there is no evidence for its use in DLB. The frequency and severity of RSD may be lessened by treating sleep apnea, if it is present. Cues can be used to help with memory retrieval.
For autonomic dysfunction, several non-medication strategies may be helpful. Dietary changes include avoiding meals rich in carbohydrates and sugary foods, eating smaller and more frequent meals, increasing table salt to reduce orthostatic hypotension, and increasing fluids or dietary fiber to treat constipation. Stool softeners and exercise also help with constipation. Physical exercise in a sitting or recumbent position, and exercise in a pool, help maintain conditioning when orthostatic hypotension is a problem. Compression stockings and elevating the head of the bed may also help. To lessen the risk of fractures in individuals at risk for falls, bone mineral density screening and testing of vitamin D levels are used. Caregivers are educated on the importance of preventing falls. Physical therapy may be helpful for motor symptoms [31].
Caregiving
Because of the neuropsychiatric symptoms associated with DLB, the demands placed on caregivers are higher than in AD, but education for caregivers has not been studied as thoroughly as in AD. Contributing factors to the caregiver burden in DLB are psychosis, aggression, agitation, and night-time behaviors such as Pansomnias that lead to a loss of independence earlier than in AD. Caregivers may experience depression, exhaustion, and be in need of support. Other family members who are not present in the daily caregiving may not observe the fluctuating behaviors or recognize the stress on the caregiver, and conflict can result when family members are not supportive. Caregiver education reduces not only distress for the caregiver, but symptoms for the individual with dementia. Visual hallucinations associated with DLB create a particular burden on caregivers; educating caregivers on how to distract or change the subject when confronted with hallucinations is more effective than arguing over the reality of the hallucination. Coping strategies may help and are worth trying, even though there is no evidence for their efficacy. These strategies include having the person with DLB look away or look at something else, focus on or try to touch the hallucination, wait for it to go away on its own, and speak with others about the visualization. Delusions and hallucinations may be reduced by increasing lighting in the evening, and making sure there is no light at night when the individual with DLB is sleeping.
Because of the neuropsychiatric symptoms associated with DLB, the demands placed on caregivers are higher than in AD, but education for caregivers has not been studied as thoroughly as in AD. Contributing factors to the caregiver burden in DLB are psychosis, aggression, agitation, and night-time behaviors such as Pansomnias that lead to a loss of independence earlier than in AD. Caregivers may experience depression, exhaustion, and be in need of support. Other family members who are not present in the daily caregiving may not observe the fluctuating behaviors or recognize the stress on the caregiver, and conflict can result when family members are not supportive. Caregiver education reduces not only distress for the caregiver, but symptoms for the individual with dementia. Visual hallucinations associated with DLB create a particular burden on caregivers; educating caregivers on how to distract or change the subject when confronted with hallucinations is more effective than arguing over the reality of the hallucination. Coping strategies may help and are worth trying, even though there is no evidence for their efficacy. These strategies include having the person with DLB look away or look at something else, focus on or try to touch the hallucination, wait for it to go away on its own, and speak with others about the visualization. Delusions and hallucinations may be reduced by increasing lighting in the evening, and making sure there is no light at night when the individual with DLB is sleeping.
With the increased risk of antipsychotics for people with DLB, educated caregivers are able to act as advocates for the person with DLB. If evaluation or treatment in an emergency room is needed, the caregiver may be able to explain that. Caregiver training, watchful waiting, identifying sources of pain, and increasing social interaction can help minimize agitation. Individuals with dementia may not be able to communicate that they are in pain, and pain is a common trigger of agitation. Medical alert bracelets or notices about medication sensitivity are available and can save lives.
When RSD is present, individuals and their caregivers need counseling about the need to improve bedroom safety. Sleep-related injuries from falling or jumping out of bed can be avoided by placing a mattress next to the bed to soften the impact of a fall, and removing sharp objects from around the bed. Sharp surfaces near the bed can be padded, bed alarm systems may help with sleepwalking, and bed partners may find it safer to sleep in another room. Firearms should be locked away, out of the bedroom.
Driving ability may be impaired early in DLB because of visual hallucinations, movement issues related to Parkinsonism, and fluctuations in cognitive ability and, at some point, it becomes unsafe for the person to drive. Driving ability is assessed as part of management and family members generally determine when driving privileges are removed. A home safety assessment can be done when there is risk of falling; handrails and shower chairs can help avoid falls [32].
Prognosis
The prognosis for DLB has not been well studied; early studies had methodological limitations, such as small sample size and selection bias. DLB generally leads to higher disability, lower life expectancy and a reduced quality of life with increased costs of care. Depression, apathy, and visual hallucinations contribute to the reduced quality of life. Decline may be more rapid when severe visuospatial deficits show up early in the course of the Lewy body dementias, when the ApoE gene is present, or when Alzheimer's or its biomarkers are also present. The severity of orthostatic hypotension also predicts a worse prognosis.
The prognosis for DLB has not been well studied; early studies had methodological limitations, such as small sample size and selection bias. DLB generally leads to higher disability, lower life expectancy and a reduced quality of life with increased costs of care. Depression, apathy, and visual hallucinations contribute to the reduced quality of life. Decline may be more rapid when severe visuospatial deficits show up early in the course of the Lewy body dementias, when the ApoE gene is present, or when Alzheimer's or its biomarkers are also present. The severity of orthostatic hypotension also predicts a worse prognosis.
Compared to AD, which is better studied, memory is retained longer, while verbal fluency may be lost faster. There are more neuropsychiatric symptoms in DLB than in AD, and they may emerge earlier, so those with DLB might have a less favorable prognosis, with accelerated cognitive decline, shorter lifespan, and increased admission to residential care. The difference in admissions to hospital and time in nursing care may be explained by the presence of hallucinations. Increased mortality relative to AD may be attributed to a higher risk of injury from falls. Life expectancy is difficult to predict, and limited study data are available. Survival may be defined from the point of disease onset, or from the (possibly delayed) point of diagnosis. A 2017 review found survival from disease onset between 5.5 and 7.7 years, survival from diagnosis between 1.9 and 6.3 years, and a poorer survival rate than in Alzheimer's. The (U.S.) National Institute for Neurological Disorders and Stroke (NINDS) writes that people with DLB typically live 8 years following diagnosis, about the same as AD though some people with Lewy body dementias have lived for 20 years. Shorter life expectancy is more likely when visual hallucinations, abnormal gait, and variable cognition are present early on. In the late part of the disease, people may be unable to care for themselves. Aspiration pneumonia, a complication of difficulty in swallowing that results from dysautonomia, commonly causes death among people with synucleinopathies [33].
Epidemiology
The Lewy body dementias are as a group the second most common form of dementia after Alzheimer's disease (AD). DLB itself is one of the three most common types of dementia, along with AD and vascular dementia, with AD making up about half of all cases. The diagnostic criteria for DLB before 2017 were highly specific, but not very sensitive so that more than half of cases were missed. DLB was under-recognized as of 2015, and there was little data on its epidemiology. The incidence and prevalence of DLB is not known accurately, but estimates are increasing with better recognition of the condition since 2017.
The Lewy body dementias are as a group the second most common form of dementia after Alzheimer's disease (AD). DLB itself is one of the three most common types of dementia, along with AD and vascular dementia, with AD making up about half of all cases. The diagnostic criteria for DLB before 2017 were highly specific, but not very sensitive so that more than half of cases were missed. DLB was under-recognized as of 2015, and there was little data on its epidemiology. The incidence and prevalence of DLB is not known accurately, but estimates are increasing with better recognition of the condition since 2017.
About 0.4% of those over the age 65 are affected with DLB, and between 1 and 4 per 1,000 people develop the condition each year. Symptoms usually appear between the ages of 50 and 80, and it is not uncommon for it to be diagnosed before the age of 65. DLB was thought to be slightly more common in men than women but a 2014 review challenged that view, claiming that the gender distribution was unclear. An estimated 10 to 15% of diagnosed dementias are Lewy body type, but estimates range as high as 24%.
A French study found an incidence among persons 65 years and older almost four times higher than a U.S. study (32 vs 112 per 100,000 person-years), but the U.S. study may have excluded people with only mild or no parkinsonism, while the French study screened for parkinsonism. Neither of the studies assessed systematically for RSD so DLB may have been under-diagnosed in both studies. A door-to-door study in Japan found a prevalence of 0.53% for persons 65 and older, and a Spanish study found similar results.
Research Directions
Cognitive training, deep brain stimulation, and transcranial direct-current stimulation have been studied more in Parkinson's and Alzheimer's disease than they have in dementia with Lewy bodies, and all are potential therapies for DLB. Medications that alter the underlying process of alpha-synuclein deposits are yet to be identified.
Future strategies involve interventions to modify the course of the disease using immunotherapy, gene therapy, and stem cell therapy, and reducing alpha synuclein accumulations. Identification of biomarkers for DLB will enable treatments to begin sooner and improve the ability to select subjects and measure efficacy in clinical trials. As of 2018 there are no well-validated neuroimaging modalities to measure alpha-synuclein in the brains of living people, but this is an active area of research.
Conclusions
While the classification, signs, symptoms, risk factors, underlying biological mechanisms and pathophysiology are well understood, the cause of the disease remains unknown more than a century after it was evidenced by Frederic Lewy. The affected areas of the brain and the associated functions are also understood. The approach to diagnosis has been set forth with diagnostic criteria, biomarkers and screen tests set forth. However, many neurodegenerative conditions share cognitive and motor symptoms with dementia with Lewy bodies, concurring to compounding the difficulties of diagnosis and requiring a differential diagnosis. The differential diagnosis includes Alzheimer's disease; such synucleinopathies as Parkinson's disease dementia; Parkinson's disease ; multiple system atrophy; vascular dementia; progressive supranuclear palsy; corticobasal palsy (CBP); degeneration; and corticobasal syndrome . Nonetheless, the disease can be managed both pharmacologically and non-pharmacologically. Current research attempts to apply applicable techniques that have been well tested such as cognitive training, deep brain stimulation, and transcranial direct-current stimulation that have been studied in Parkinson's and Alzheimer's disease but much less in the present disease. Medications that alter the underlying process of alpha-synuclein deposits are yet to be identified. But, even if such medications were to become available, they may not provide the desired cure because the deposits are not the root cause of the disease. This is very reminiscent of the tau proteins and neurofibrillary tangles in Alzheimer's in that, even though they could be eliminated, the disease progressed unabated and even was made worse in certain cases. Like for Alzheimer's, I am advocating dedicating our research efforts identifying the root cause(s) of the disease for it will be then, and only then, that a cure could be found. Such future strategies could involve interventions to modify the course of the disease using immunotherapy, gene therapy, and stem cell therapy, and reducing accumulations of alpha synuclein proteins. In the meantime, identification of biomarkers will enable palliative treatments to begin sooner, and improve the ability to select subjects and measure efficacy in clinical trials. As of 2018, such efforts are not underway and there are no well-validated neuroimaging modalities to measure alpha-synuclein in the brains of living people, but this is an active area of research.
Sidebar
The vertebrate nervous system
For those readers interested in better understanding how the various divisions of the vertebrate nervous system are integrated with each other and work synergistically, I provide the flowchart of Figure 4.
For those readers interested in better understanding how the various divisions of the vertebrate nervous system are integrated with each other and work synergistically, I provide the flowchart of Figure 4.
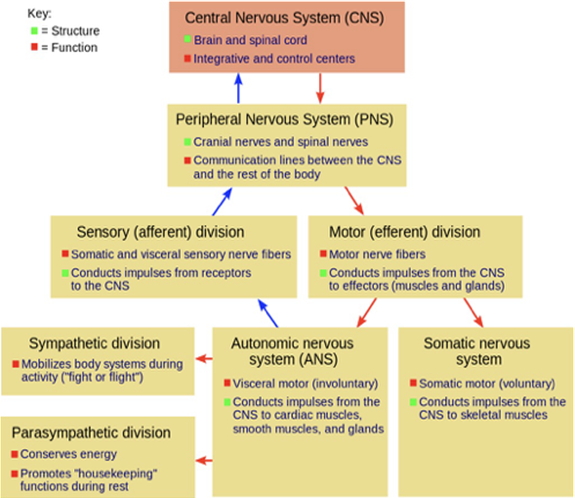
Figure 4: Major divisions of the vertebrate nervous system
and their structural and functional signal flows.
Source: http://commons.wikimedia.org/wiki/File:NSdiagram.png
Source: http://commons.wikimedia.org/wiki/File:NSdiagram.png
There, for greater clarity, structure and function are colored differently (respectively, green and red). Starting from the bottom of the chart, initial sensations (input signal) originate in the autonomic nervous system (with its sympathetic and parasympathetic divisions), feed into the sensory (afferent) division that, in turn, relays this information to the peripheral nervous system, itself interacting with the central nervous system. The resulting response (output signal) is communicated downwards from the central nervous system to the peripheral nervous system to the motor efferent division from which it feeds back to the autonomic nervous system and the somatic nervous system. At each step of this multi-path communication process, one can easily trace the flow of information from the sensed signal all the way up to the central nervous system and the return command (output signal) from this system to the autonomous and somatic nervous systems.
References
- Boot BP. “Comprehensive treatment of dementia with Lewy bodies”. Alzheimer's Res Ther (Review). 7.1 (2015): 45. Doi: 10.1186/s13195-015-0128.
- Boot BP., et al. “Treatment of dementia with Lewy bodies”. Curr Treat Options Neurol (Review) 15.6 (2013): 738–64. Doi: 10.1007/s11940-013-0261-6.
- Lewy F. “Paralysis agitans”. I. Pathologische Anatomie In Handbuch der Neurologie, Lewandowsky M, Abelsdorff G, eds. Springer Verlag, Berlin 3 (1912): 920–33.
- Neef D and Walling AD. “Dementia with Lewy bodies: An emerging disease”. Am Fam Physician (Review) 73.7 (2006): 1223–29.
- (U.S.) National Institute on Aging. “Diagnosing Lewy body dementia”. National Institute on Aging. US National Institutes of Health (2017).
- (U.S.) National Institute on Aging. “What is Lewy body dementia”. National Institute on Aging (2017).
- (U.S.) National Institute on Aging. “Diagnosing dementia”. National Institute on Aging (2017).
- (U.S.) National Institute of Aging. “Vascular contributions to cognitive impairment and dementia”. National Institute on Aging (2017).
- (U.S.) National Institute of Neurological Disorders and Stroke. “Dementia with Lewy bodies information page”. National Institute of Neurological Disorders and Stroke (2017).
- (U.S.) National Institute of Neurological Disorders and Stroke (2017). “Lewy body dementia: Hope through research”, National Institute of Neurological Disorders and Stroke.
- Fymat AL. “Parkinson's Disease and Other Movement Disorders: A Review”. Journal of Current Opinions on Neurological Science 2.1 (2017j): 316-43.
- Fymat AL. “Neurological Disorders and the Blood Brain Barrier: 2. Parkinson's Disease and Other Movement Disorders”. Journal of Current Opinions on Neurological Science 2.1 (2017): 362-83.
- Fymat AL. “Alzheimer's Disease: A Review”. Journal of Current Opinions on Neurological Science 2.2 (2018a): 415-36.
- Fymat AL. “Alzheimer's Disease: Prevention, Delay, Minimization and Reversal”. Journal of Clinical Research in Neurology 1.1 (2018b): 1-16.
- Fymat AL. “Is Alzheimer an Autoimmune Disease Gone Rogue”. Journal of Clinical Research in Neurology 2.1 (2018c): 1-4.
- Fymat AL. “Is Alzheimer a Runaway Autoimmune Disease? and How to Cure it?”. Newsletter European Union Academy of Sciences Annual Report (2018).
- Fymat AL. “Dementia Treatment: Where Do We Stand?” Journal of Current Opinions on Neurological Science 3.1 (2018e): 1-3.
- Fymat AL. “On Dementia and Other Cognitive Disorders”. Journal of Clinical Research in Neurology 1.2 (2018f): 1-14.
- Fymat AL.”Dementia: A Review”. Journal of Clinical Psychiatry and Neuroscience 1.3 (2019g): 27-34. Karantzoulis S and Galvin JE. “Distinguishing Alzheimer's disease from other major forms of dementia”. Expert Rev Neurother (Review) 11.11 (2011): 1579-91. Doi: 10.1586/ern.11.155.
- Kosaka K. “Lewy body disease and dementia with Lewy bodies”. Proc Jpn Acad Ser B Phys 90.8(2014): 301–306.
- Chung EJ and Kim SJ. “123I-Metaiodobenzylguanidine myocardial scintigraphy in Lewy body related disorders: A literature review”. J Mov Disord (Review) 8.2 (2015): 55-66. Doi: 10.14802/jmd.15015.
- Gomperts SN. “Lewy body dementias: Dementia with Lewy bodies and Parkinson's disease dementia”. Continuum (Minneap Minn (Review). 22 (2 Dementia) (2016): 435-63. Doi: 10.1212/CON.0000000000000309.
- Finger EC. “Frontotemporal dementias”. Continuum (Minneap Minn) (Review). 22(2 Dementia) (2016): 464–89. Doi: 10.12.12/CON.0000000000000300.
- McKeith IG. "Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop". J Alzheimers Dis 9 (3 Suppl) (2006): 417–23. Doi: 3233/JAD-2006-9S347.
- McKeith IG., et al. “Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium”. Neurology (Review) 89.1 (2017): 88-100. Doi: 10.1212/WNL.000000000004058.
- Siderowf A., et al. "Biomarkers for cognitive impairment in Lewy body disorders: Status and relevance for clinical trials". Mov Disord (Review) 33.4 (2018): 528–36. Doi: 10.1002/mds.27355.
- St Louis EK., et al. "REM sleep behavior disorder in Parkinson's disease and other synucleinopathies". Mov Disord (Review) 32.5 (2017): 645-58. Doi: 10.1002/mds.27018.
- Tousi B. "Diagnosis and management of cognitive and behavioral changes in dementia with Lewy bodies". Curr Treat Options Neurol (Review) 19.11 (2017): 42. Doi: 10.1007/s11940-017-0478.
- Velayudhan L., et al. "New therapeutic strategies for Lewy body dementias". Curr Neurol Neurosci Rep (Review) 17.9 (2017): 68. Doi: 10.1007/s11910-017- 0778-2.
- Walker Z., et al. “Lewy body dementias”. Lancet (Review) 386 (10004) (2015): 1683–97. Doi: 10.1016/S0140-6736(15)00462-6.
- Connors MH., et al.. “Non-pharmacological interventions for Lewy body dementia: A systematic review”. Psychol Med (Review) 48.11 (2017): 1749-58. Doi: 10.1017/S0033291717003257.
- Zweig YR and Galvin JE. “Lewy body dementia: The impact on patients and caregivers”. Alzheimers Res Ther (Review) 6.2 (2014): 21. Doi: 1186/alzrt351.
- Mueller C., et al. "The prognosis of dementia with Lewy bodies". Lancet Neurol (Review) 16.5 (2017): 390–8. Doi: 10.1016/S1474-4422(17)30074-1.
Citation:
Alain L Fymat. “Dementia with Lewy Bodies: A Review”. Current Opinions in Neurological Science 4.1 (2019): 15-32.
Copyright: © 2019 Alain L Fymat. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.






























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.