Research Article
Volume 1 Issue 4 - 2017
Hepatic Transaminase’s Level in Chronically Treated Diabetic’s Patients (Type 2) at Alhusain Teaching Hospital in Al- Nasiriyah City
1Department of Pharmacognasy, College of Pharmacy, Thi-Qar University, Iraq
2General Directorate for Education in Thi-Qar/Iraq
3Department of Pediatric Nursing, College of Nursing, Thi-Qar University, Iraq
2General Directorate for Education in Thi-Qar/Iraq
3Department of Pediatric Nursing, College of Nursing, Thi-Qar University, Iraq
*Corresponding Author:Najim Abass Jabir, College of Pharmacy/Thi-Qar University Iraq.
Received: January 14, 2017; Published: August 10, 2017
Abstract
Background: The study was planned to evaluate the liver function of type 2 diabetes mellitus (T2MD) by measuring serum glutamic oxaloacetic transaminase (SGOT), serum glutamic pyruvic transaminase (SGPT), plasma glucose, urea and creatinine.
The study investigates the level of SGPT and SGOT in patients who suffer from diabetes mellitus at Al-Hussein teaching hospital in Al-nasiriyah city.
Materials and methods: 80 patients who diagnosed as type 2 diabetes mellitus (T2DM) (40 males and 40 females) were included in the study. Forty matched normal individuals were taken as a controlled group. Their age ranged between 40 and 90 years.
Results: In the present study, mean values of SGPT, SGOT, plasma glucose, urea and creatinine were significantly higher in patients than in the controlled group (P < 0.01), and this significant defirences founf between the two groups of patients (P < 0.01).
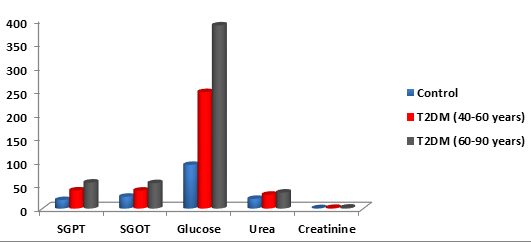
The mean (± S.D.) SGOT in the controlled group was 25.01 ± 0.77, whereas in T2DM (40-60 years) was 37.87 ± 3.88 and T2DM (60-90 years) was 53.73 ± 5.05. The mean (± S.D) blood sugar in controlled group was 92.24 ± 2.15, whereas in T2DM (40-60 years) was 246.12 ± 16.23 and 387.00 ± 18.55 in the T2DM (60-90 years) . Creatinine levels in controlled group were 0.82 ± 0.35 and in T2DM (40-60 years) was 1.50 ± 0.98 and T2DM (60-90 years) was 2.03 ± 1.10. There was a significant increase in the SGPT, SGOT, plasma glucose, urea and creatinine in T2DM (60-90 years) compared with T2DM (40-60 years).
Conclusion: The values of the liver function tests in patients with T2DM were significantly higher than that of controlled patients. Moreover, patients had higher glucose, urea and creatinine than in the controlled group.
Keywords:Diabetes mellitus; SGPT; SGOT; Glucose; Urea; Creatinine
Introduction
Type 2 diabetes mellitus is the most common type represents (90%) of DM patients worldwide (Rubino, 2002). Type 2DM can be characterized by insulin resistance or reduced insulin sensitivity (down regulation of Insulin receptors). Type-2 DM is also most commonly associated with obesity in middle-aged individuals. There is an association between diabetes and liver injury. In hyperglycemias states, there will be an intracellular glycogen accumulation in the hepatocytes due to increased glycogen synthesis, causing typical biochemical findings of mild to moderately elevated aminotransferases, normal liver synthetic function, with or without mild elevations of alkaline phosphatase (Chatila R., et al.1996). The prevalence of type 2 diabetes mellitus (T2DM) is higher in patients who have suffered from certain liver diseases. Therefore, it is speculated that there is a pathogenic link between liver disease progression and T2DM (Po-Shiuan and Yen-Ju, 2011).
AST (SGOT) and ALT (SGPT) are sensitive indicators of liver damage from different types of disease. But it must be emphasized that higher-than-normal levels of these liver enzymes should not be automatically equated with liver disease. They may mean liver problems or they may not. The interpretation of elevated AST and ALT levels depends upon the whole clinical picture and so it is best done by doctors experienced in evaluating liver disease (Elizabeth, 2005).
At the other side Idiosyncratic hepatotoxicity is a rare and unpredictable event of liver injury affecting generally less than 1 in 10,000 patients treated with certain drugs. However, it is a serious clinical problem as it accounts for 10% of all drug-induced liver failure cases (Kaplowitz, 2005). Since idiosyncratic drug reactions are not detected in preclinical testing and in most cases not even during clinical trials, the problem surfaces generally after the drug is approved and hundreds of thousands of patients are being treated. Idiosyncratic hepatotoxicities are currently the main cause for Food and Drug Administration-mandated warnings, restrictions of use or even withdrawals of drugs from the market (Kaplowitz, 2005). As such, this is a consider- able problem for the pharmaceutical industry and for regulatory agencies worldwide.
And certain medicinal agents, when taken in overdoses and sometimes even when introduced within therapeutic ranges, may injure the organ (Aashish., et al. 2012). The list of hepatotoxic drugs is huge and a complete coverage is difficult. To sum up thus there is a large category of drugs used for different therapeutic indications which are toxic to the liver and thus should be cautiously administered (Aashish., et al. 2012). One of the recent examples of drugs causing idiosyncratic hepatotoxicity and liver failure was the antidiabetic drugs.
Metformin is the first choice oral antidiabetic drug for type 2 diabetes and currently the most consumed, metformin-induced hepatotoxicity but rarely. Fewer than 10 cases have been reported (Cone., et al. 2010). However, given the increasing prevalence of type 2 diabetes and expanding indications for metformin, are found that the clinical and laboratory findings of metformin-induced hepatotoxicity and MALA (metformin- associated metabolic acidosis ) in the older diabetic patients (Ersin, 2007: Erbey., et al. 2000), it is important that clinicians be alert to the occurrence of rare but potentially serious side effects of this drug, such as idiosyncratic hepatotoxicity (Rhee., et al. 2010). Troglitazone was an effective antidiabetic drug with a fundamentally new mechanism of action. However, within a year after its widespread use, individual cases of liver injury and failure were reported (Watkins, 2005). If the injury does not progress, the model may explain the fact that about 2% of patients treated with troglitazone but only 0.6% of patients receiving placebo experienced a significant increase of plasma ALT activities (Graham., et al. 2003).
A recent analysis of genetic polymorphism in Japanese patients treated with troglitazone revealed that a combined glutathione-S- transferase GSTT1-GSTM1 null genotype correlated with in- crease ALT levels (Watanabe., et al. 2003).
So increased activities of liver enzymes such as aspartate aminotransferase (AST), alanine aminotransferase (ALT) and γ-glutamyltranspeptidase (GGT) are indicators of hepatocellular injury and associated with insulin resistance (Marchesini., et al. 2001). Liver pathology among diabetics is similar to that of alcoholic liver disease, including fatty liver (steatosis), steatohepatitis, fibrosis, and cirrhosis (Erbey., et al. 2000). The elevated serum activity of the two aminotransferases, aspartate aminotransferase (AST) and alanine aminotransferase (ALT), is the most frequently measured indicators of metabolic liver disease and occurs in chronically treated old diabetics patients more frequently than in the general population due to endogenous antioxidant deficiencies may uncover potential liabilities of drugs even at therapeutic levels, which could provide a strong indication for possible idiosyncratic reactions in humans. (Ong., et al. (2007).
Materials and Methods
Design of study: The study, conducted at Al-Hussein teaching hospital in Al-Nasiriyah city during 6 months (from 20th of October 2012 to 28th of March 2013). Our study samples were (80) patients selected between the ages of 18 to 90 years.
Collection of Blood Sample: 10 ml of venous blood sample was collected from each fasting type 2 DM and controlled group patients and allowed to clot and to get the serum by putting it in an empty disposable tube which is then separated in the centrifuge at 3000 (rpm) for 10 minutes, the serum samples were separated, stored at (-20˚C ) for later measurement biochemical parameters, unless used immediately. The clinical chart of this study describes experimental parameters in the following Table 1.
| Name:- |
| Sex:- |
| Age:- |
| Address:- |
| Occupation:- |
| Marital status:- |
| History of disease:- |
| Type of investigation:- |
| Type of treatment:- |
| Biochemical Parameters |
| Serum Glucose |
| Liver function test: Serum glutamic oxaloacetic transaminase (SGOT) Serum glutamic pyruvic transaminase (SGPT) |
| Renal function test: - Urea test creatinine test |
Table 1:
Biochemical Parameters: The used reagents were supplied by Biolabo (France), and Serum glucose was measured according to (Greiling., et al. 1995). (SGOT) and (SGPT) were measured according to (Reitman and Frankel, 1957). Determination of plasma creatinine was carried out using Jaffe´s method described by Bowers and Wong (1980). Urea was estimated using urease- Berthelot´s method described by Richterich and Kuffer (1973).
Statistical Analysis: Statistical analysis was done by using the software SPSS version 17.0; the results were expressed as mean ± standard deviations (Mean ± SD). One way ANOVA-test was used to compare parameters in different studied groups. P-values (P < 0.01) were considered statistically significant.
Results
| Parameters | Control (n = 40) | T2DM (40-60 years) | T2DM (60-90 years |
| SGPT (U/L) | 18.00 ± 1.12c | 38.20 ± 4.03b | 54.88 ± 5.88a |
| SGOT (U/L) | 25.01 ± 0.77c | 37.87 ± 3.88b | 53.73 ± 5.05a |
| Glucose (mg/dl) | 92.24 ± 2.15c | 246.12 ± 16.23b | 387.00 ± 18.55a |
| Urea (mg/dl) | 20.50 ± 0.99c | 29.21 ± 3.25b | 33.83± 3.89a |
| Creatinine(mg/dl) | 0.82 ± 0.35c | 1.50 ± 0.98b | 2.03 ± 1.10a |
* Note: Each value represents (mean ± SD) values with non identical superscript (a, b or c …exetra.) were considered significantly different (P < 0.01).
In the current study, SGPT, SGOT, plasma glucose, urea and creatinine were estimated in type 2 diabetic patients and controlled group (Table 2) and (Figure 1). The mean (± S.D.) SGPT in the controlled group was 18.00 ± 1.12, whereas in T2DM (40-60 years) was 38.20 ± 4.03 and T2DM in patients aged between (60-90 years) was 54.88 ± 5.88. The mean (± S.D.) SGOT in the controlled group was 25.01 ± 0.77, while in T2DM aged (40-60 years) was 37.87 ± 3.88 and T2DM (60-90 years) was 53.73 ± 5.05. The mean (± S.D) blood sugar in the controlled group was 92.24 ± 2.15, whereas in T2DM (40-60 years) was 246.12 ± 16.23. The mean (± S.D.) and T2DM (60-90 years) was 387.00 ± 18.55. Urea levels in controlled group were 20.50 ± 0.99 and in T2DM (40-60 years) were 29.21 ± 3.25 and T2DM (60-90 years) were 33.83 ± 3.89. Creatinine levels in the controlled patients were 0.82 ± 0.35 and in T2DM (60-90 years) were 1.50 ± 0.98 and T2DM (60-90 years) were 2.03 ± 1.10 (Table 2). There was a significant increase in the SGPT, SGOT, plasma glucose, urea and creatinine in T2DM (60-90 years) compared with T2DM (40-60 years) (P < 0.01).
Discussion
The findings in our study are similar to those of (Everhart, 1995) who reported that the elevated serum activity of the SGPT and SGOT, are the most frequently measured indicators of liver disease and occurs in diabetics more frequently than in the general population. Our study also supported the finding of important study conducted by (Sherif Gonem., et al. In 2007). He tried to assess the prevalence of abnormal liver function tests in patients with diabetes mellitus, and all these studies are coming with agreement with Ayman., et al. (2011) who pointed out that the mean values of SGPT and SGOT were significantly higher in diabetic patients than in the controlled patients (P < 0.01).
Our results also indicated another logic cause of transaminase enzymes elevation to improve the results of (Kaplowitz, 2005), which pointed out that a serious clinical hepatotoxicity problem as it accounts for 10% of all drug-induced liver failure cases which were characterized by elevation of SGOT and SGPT level. So, when certain drugs taken in overdoses or within therapeutic doses may induce hepatic injury (Aashish., et al. 2012). Liver pathology among diabetics is similar to that of alcoholic liver disease, including fatty liver (steatosis), steatohepatitis, fibrosis, and cirrhosis (Erbey., et al. 2000).
Actuel study improve also in additional to the dose - toxicity relation ship or effect there are age effect demonstrated in the table 2 of our study which showed theat the category of patients age (60-90 years) have significant a higest level of SGOT,SGPT than in the patient aged (40-60), this signifivant defences between the qges has been shown in the other parameters included in our study.
Plasma creatinine and urea are established markers of Glomerular Filtration Rate (GFR). Though plasma creatinine is more sensitive index of kidney function than the plasma urea level. This is because creatinine fulfills most of the requirements for a perfect filtration marker (Perrone., et al. 1992). This observation is similar to the reports of Aldler., et al. (2003), Judykay (2007) and Wagle (2010). Aldler., et al. (2003) in their report submitted that raised plasma creatinine and urea levels in diabetic patient may indicate a pre-renal problem such as volume depletion. Judykay (2007) in his submission suggested that high creatinine in diabetic patient may be due to nephrons impairment.
Conclusion
The values of liver function tests in patients with type 2 diabetes mellitus were significantly higher than that of controlled patients. Moreover, patients had higher glucose, urea and creatinine in comparison to the controlled group.Accordingly, assessment of liver function tests is highly recommended for diabetic patients. Additionally, further studies on the liver function of diabetic patients need to be done as well.
References
- Aashish P., et al. “Drug-Induced Hepatotoxicity”. Journal of Applied Pharmaceutical Science 2.5 (2012): 233-243.
- Aldler AI., et al. “Development and progression of nephropathy in type 2 diabetes: The United Kingdom Propective Diabetes Study (UKPDS 64)”. Kidney International 63.1 (2003): 225-232.
- Ayman S Idris., et al. “Liver function tests in type 2 Sudanese diabetic patients”. International Journal of Nutrition and Metabolism 3.2 (2011): 17-21.
- Bowers LD and ET Wong. “Kinetic serum creatinine assays critical evaluation and review”. Clinical Chemistry26.5 (1980): 555-561
- Chatila R and West AB. “Hep atomegaly and abnormal liver tests due to glycogenosis in adults with diabetes”. Medicine 75.6 (1996): 327-33.
- ConeCJ., et al. “Hepatotoxicity associated with metformin therapy in treatment of type 2 diabetes mellitus with nonalcoholic fatty liver disease”. Annals of Pharmacotherapy 44.10 (2010): 1655-1659.
- Elizabeth H and Haris. “ Elevated Liver Function Test In Type 2 Diabetes”. clinical Diabetes 23.3 (2005): 115-119.
- Erbey JR., et al. “Prevalence of abnormal serum alanine aminotransferase levels in obese patients and patients with type 2 diabetes”. American Journal of Medicine. 109.7 (2000): 588-590.
- Everhart JE. “Diabetes in America. 2nd ed. National Institute of Health. National Institute of Diabetes and Digestive and Kidney Diseases”. Washington, DC: GPO: 457-483.
- Gonem S et al . “Prevalence of abnormal liver function tests in patients with diabetes mellitus”. Endocrine Abstracts 13 (2007): P157.
- Graham DJ., et al. “Troglitazone- induced liver failure: A case study”. The American Journal of Medicine 114. (2003): 299-306.
- Greiling H., et al. “Chemie und Pathobiochemie, 3rd, Stuttgart/ New York ;Schattauer Verlag”.
- Judykay T. “Nutrition for reducing urea and creatinine in the blood”. Diabetes Cara 27 (2007): 2191-2192.
- Kaplowitz N. “Idiosyncratic drug hepatotoxicity”. Nature Reviews Drug Discovery 4.6 (2005): 489-499.
- Marchesini G., et al. “Nonalcoholic fatty liver disease. A feature of metabolic syndrome”. Diabetes50.8 (2001): 1844-1850.
- Ong MM., et al. “Troglitazone-induced hepatic necrosis in an animal model of silent genetic mitochondrial abnormalities”. Toxicological Sciences97.1 (2007): 205-213.
- Perrone RD., et al. “Serum creatinine as index of renal function”. Clinical Chemistry 38.10 (1992): 1933-1953.
- Po-Shiuan H and Yen-Ju H. “Impact of liver diseases on the development of type 2 diabetes mellitus”. World Journal of Gastroenterology 17.48 (2011): 5240-5245.
- Reitman S and Frankel S (1957). American Journal of Clinical Pathology. 28: 56.
- RheeMK., et al. “Many Americans have pre-diabetes and should be considered for metformin therapy”. Diabetes Care 33.1 (2010): 49-54.
- Richterich R and H Kuffer. “The determination of urea in plasma and serum by a urease/Berthelot method, adapted to the Greiner Electronic Selective Analyzer GSA”. Z Klin Chem Klin Biochem 11.12 (1973): 553-564
- Rubino F and Gagner M. “Potential of surgery for curing type 2 diabetes mellitus”. Annals of Surgery 236.5 (2002): 554-559.
- Wagle TJ. “Genderwise comparision of serum creatinine and blood sugar levels in type 2 diabetic pantients”. Bombay Hospital Journal 52.1 (2010): 64-68.
- Watanabe I., et al. “A study to survey susceptible genetic factors responsible for troglitazone-associated hepatotoxicity in Japanese patients with type 2 diabetes mellitus”. Clinical Pharmacology & Therapeutics 73.5 (2003): 435-455.
- Watkins PB. “Idiosyncratic liver injury: Challenges and approaches”. Toxicologic Pathology 33.1 (2005): 1-5.
Citation:
Najim A. Jabir AL-Awwadi., et al. “Hepatic Transaminase’s Level in Chronically Treated Diabetic’s Patients (Type 2) at Alhusain Teaching Hospital in Al- Nasiriyah City”. Chronicles of Pharmaceutical Science 1.4 (2017): 174-179.
Copyright: © 2017 Najim A. Jabir AL-Awwadi., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.