Case Report
Volume 1 Issue 2 - 2018
Thyroid Metastasis from Renal Cell Carcinoma
1Department of Endocrinology
2Department of Pathology, King Fahad Armed Forces Hospital, Jeddah, Kingdom of Saudi Arabia
3College of medicine, Um Al Qura University
2Department of Pathology, King Fahad Armed Forces Hospital, Jeddah, Kingdom of Saudi Arabia
3College of medicine, Um Al Qura University
*Corresponding Author: Khalid S Aljabri, Department of Endocrinology, King Fahad Armed Forces Hospital, Jeddah, Kingdom of Saudi
Arabia.
Received: April 05, 2018; Published: April 27, 2018
Abstract
Although secondary involvement of the thyroid gland by cell renal carcinoma (RCC) is rare, it is still one of the more common neoplasms to metastasize to this site. We report a case of 64 years old male with a clear RCC metastasize to the thyroid.
Keywords: Thyroid metastasis; Renal cell carcinoma
Introduction
The thyroid gland is a rare site of clinically detectable tumor metastasis [1-4]. Renal cell carcinoma (RCC) disseminates in an unpredictable manner and can show late recurrences [5-8]. Although secondary involvement of the thyroid gland by RCC is rare, it is still one of the more common neoplasms to metastasize to this site which accounts for approximately 2% of thyroid malig-nancies [4,7,9]. Autopsy studies demonstrate a higher prevalence with thyroid involvement in up to 24% of cases showing widespread malignant disease [1,3,4,6,7,9-14]. A thyroid mass can be the first clinical manifestation of RCC, even masquerading as a primary thyroid gland neoplasm [5-7]. We report a case of 64 years old male with a clear RCC metastasize to the thyroid.
Case Report
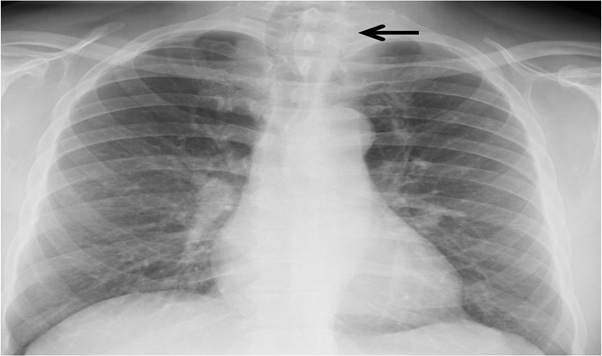
A 64-year-old Saudi man was referred to the Department of surgery for thyroid nodule that had been diagnosed June 2010 by thyroid ultrasound which revealed a left lobe is 6.8x3.9x5 cm showing coarse heterogenous echo pattern with well-defined outline large is echoic nodule 5x4cm with multiple hypo echoic areas (Figure 1). The patient was asymptomatic at that time, with a euthyroid status. His medical history included type 2 diabetes mellitus and a radical right nephrectomy with a diagnosis of RCC 18 years prior to presentation. Chest X-ray showed right tracheal deviation (Figure 2). October 2010, Fine needle aspiration (FNA) guided by ultrasound of the thyroid nodule was performed and the cytological examination was reported to be Hurthle cell neoplasm. December 2010, computed tomography (CT) scan of the neck with IV contrast showed the left thyroid lobe is markedly enlarged with a large nodule seen showing heterogenous contrast enhancement with predominantly central hypodensity. The lesion is seen extending mainly downward, however no evidence of retrosternal extension. The lesion displaced the major vascular structures at the ipsilateral side laterally and the trachea to the right side (Figure 3). February 2011, the patient underwent total thyroidectomy. Histological examination showed Left thyroid lobe mass (4.5x3x3 cm), consistent with metastatic clear RCC which was immunoreactive for the tumor cells are positive for EMA, vimentin, and CK7 and negative for Thyroglobulin, Galactin 3 and thyroid transcription factor-1 (TTF-1), confirming the renal origin of the tumor (Figure 4). The patient’s postoperative course and subsequent recovery were uneventful. During follow-up, a CT scan of the chest, abdomen and pelvis showed multiple lung nodules, mediastinal metastatic lymph nodes and lytic bony lesion noted in the left iliac bone. Unfortunately, patient lost follow up since May 2011.
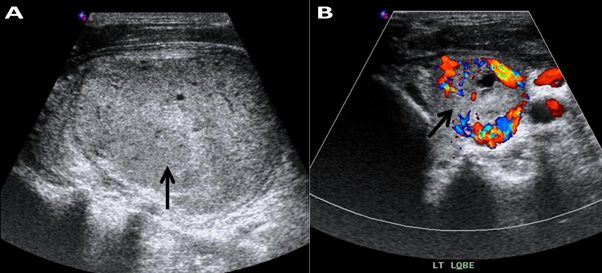
Figure 1: Ultrasonography of the left lobe.
A. Coarse heterogenous echo pattern with well-defined outline large isoechoic nodule 5 x 4 cm (black arrow) with multiple hypo echoic areas. Strong echoes with acoustic shadowing (calcifications) are not present.
B. Power Doppler ultrasonography showing marked chaotic vascularity (black arrow).
A. Coarse heterogenous echo pattern with well-defined outline large isoechoic nodule 5 x 4 cm (black arrow) with multiple hypo echoic areas. Strong echoes with acoustic shadowing (calcifications) are not present.
B. Power Doppler ultrasonography showing marked chaotic vascularity (black arrow).
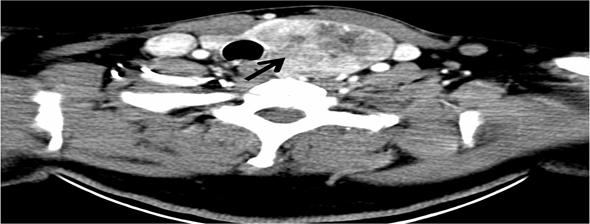
Figure 3: An intravenous contrast CT scan of the neck showed a left thyroid lobe is markedly
enlarged with a large nodule seen (black arrow) showing heterogenous contrast enhancement
with predominantly central hypodensity. The lesion is seen extending mainly downward, however
no evidence of retrosternal extension. The lesion displaced the major vascular structures at the
ipsilateral side laterally and the trachea to the right.
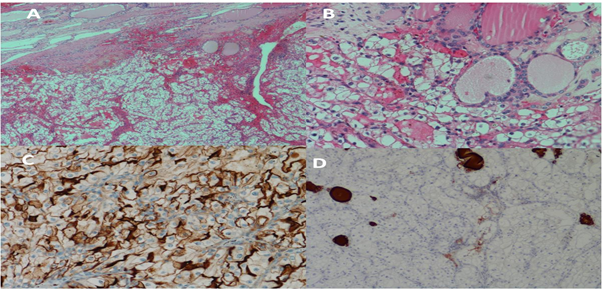
Figure 4: A: Metastatic renal cell carcinoma. Upper field shows residual thyroid follicles while the lower
field shows the malignant clear cells of RCC (Hematoxylin & eosin staining, 40X).
B: Unremarkable thyroid follicles and clear cells of metastatic renal cell carcinoma (H&E, 200X).
C: Epithelial membrane antigen (EMA) in the malignant cells.
D: Malignant cells are negative for Thyroglobulin while residual thyroid follicles are staining positively.
B: Unremarkable thyroid follicles and clear cells of metastatic renal cell carcinoma (H&E, 200X).
C: Epithelial membrane antigen (EMA) in the malignant cells.
D: Malignant cells are negative for Thyroglobulin while residual thyroid follicles are staining positively.
Discussion
Metastatic tumors represent 2 to 3% of all thyroid malignancies [4,7,9,15]. Several autopsy series reported in the previous literature have shown that the overall incidence of thyroid metastasis ranges from 1.9% to 24% suggests that metastatic thyroid lesions are often occult [1,12,16]. Although RCC is the most common malignancy to metastasize to the thyroid glands, its diagnosis is clinically challenging due to its indolent clinical course. Thyroid metastases from RCC account for 48.1% of Non-thyroid Malignancies metastases to the thyroid gland with a propensity to extend into the jugular veins. The low frequency of metastasis to the thyroid can be explained by high iodine and oxygen concentrations as well as a high intrathyroidal vascular flow that the thyroid gland would be more susceptible to metastatic growth when affected by goiter, neoplasms, or thyroiditis due to metabolic changes that consist of decrements in the oxygen and iodine content [11,17]. Because of these reasons, the thyroid is not a likely site for malignant metastatic cells. By contrast, other authors have reported that there is no difference in frequency of metastasis in altered thyroid glands versus normal thyroid glands [11].
Metastatic nodules are found long after surgical resection of the primary mass, with the mean interval ranging from 6 to 12.5 years. In total, 85% of general distant organ metastases in RCC are seen within 3 years following primary resection. However, some cases with metastases that occurred a few decades later have also been reported [3,18]. Thyroid metastases generally occur after 10 years and most patients present with no specific symptoms other than a palpable neck mass [12,13,16, 19-21]. This is consistent with the characteristic behavior of RCC, in which recurrence occurs after a long latency period. Our patient had a right radical nephrectomy 18 years before the metastasis to the thyroid gland was seen. Patient presented with no symptoms other than a palpable neck mass. It should be kept in mind that an early metastatic lesion can be seen in a patient with a mass in the neck and a history of RCC diagnosis.
The commonly known US features of thyroid metastasis are nonspecific findings including nonhomogeneous hypoechogenicity, noncircumscribed margins, no calcifications, and increased vascularity [13,16,19,20]. Only a few reports have described findings specific to RCC, which include solitary and solid nodules without calcification. According to a recently published study, metastatic RCC in the thyroid gland tends to form a nodular mass that mimics a primary thyroid tumor or even benign masses with few to no suspicious US features [20]. Nine metastatic RCC nodules showed uniform characteristic US features, appearing as hypo echoic solid nodules (100%) with well-defined smooth margins (87.5%) and increased vascularity (100%). All nodules were classified as high or intermediate suspicion according to the ATA guidelines and intermediate suspicion on K-TIRADS, which requires FNA of nodules ≥ 1 cm [22].
FNA is widely accepted as a diagnostic tool for thyroid malignancies or metastases, with a reported sensitivity of 73.7% 94% and specificity up to 100% [23,24]. However, the diagnostic yield of FNA specific to metastatic RCC needs further investigation, as false negative outcomes in FNA cytology have been increasingly recognized as a concern for these metastases. Chung., et al. searched PubMed for articles published between 2000 and 2010 on metastatic thyroid tumors and reported that preoperative FNA performed for thyroid metastases resulted in an incorrect diagnosis in 44 of 167 cases (26.3%) [13]. Of these, RCC had the highest rate of inaccurate FNA results (28.6%) The cause for this clinically significant rate of inaccurate or indeterminate FNA results in metastatic RCC compared to other metastatic tumors is not yet known. No study has demonstrated the limitations regarding the cytologic evaluation of metastatic RCC, and no study has addressed the possibility of renal clear cell cytology mimicking benign cytology. However, increased vascularity may be the most plausible reason for the low diagnostic yield of FNA. Hyper vascular nodules are more likely to yield no diagnostic results due to a higher likelihood of blood contamination, which makes cytologic examinations more difficult [25]. Our patient had thyroid gland enlargement with a large nodule. In such patients, attempts to establish diagnosis with Fine needle aspiration may yield false-negative results if a large nodule is sampled. However, if the pathologist is aware of the oncologic history of the patient, the tumor pathology may be revealed by immunohistochemical techniques.
Up-to-now, a thyroid gland monitoring is still not included in the follow-up program of RCC patients, even if metastases to this site can occur several years after the primary surgical treatment. In contrast, is known that a precocious surgical intervention on the thyroid may prevent local recurrence and extension into other regional structures like the recurrent laryngeal nerve and trachea, improving patients’ quality of life [13]. Notably, our review demonstrates that jugular vein infiltration and local widespread are found in 15.6% of cases of thyroid metastases from RCC. Finally, surgical treatment of patients with solitary thyroid gland metastases is recommended, since it demonstrated a favorable prognosis in patients treated with radical surgery (mean 5-year survival rate 30-60%) [26].
In conclusion, thyroid metastases should be kept in mind for patients who had a thyroid nodule and a history of RCC. The differentiation of primary and secondary tumors is possible only by the pathological examination of suspected thyroid nodules.
Competing interests
The authors declare that they have no competing interests.
The authors declare that they have no competing interests.
References
- Nakhjavani MK., et al. “Metastasis to the thyroid gland. A report of 43 cases.” Cancer 79.3 (1997): 574-578.
- Dionigi G., et al. “Solitary intrathyroidal metastasis of renal clear cellcarcinoma in a toxic substernal multinodular goiter.” Thyroid Research 1.6 (2008).
- Kihara M., et al. “Metastasis of renal cell carcinoma to thethyroid gland 19 years after nephrectomy: a case report.” Auris Nasus Larynx 31.1 (2004): 5-100.
- Medas F., et al. “Renal cell carcinoma metastasis to thyroid tumor: a case report and review of the literature”. Journal of Medical Case Reports 7. 265 (2013).
- Chin CJ., et al. “Metastasis from renal cell carcinoma to the thyroid 12 years after nephrectomy.” Canadian Medical Association Journal 183.12 (2011): 1398-1399.
- Lee JG., et al. “A case of metastatic renal cell carcinoma to thyroid gland”. Chonnam Medical Journal 47.2 (2011): 130-133.
- Sindoni A., et al. “Thyroid metastases from clear cell renal carcinoma 18 years after nephrectomy”. Annales d'Endocrinologie 71.2 (2010): 127-130.
- Tunio MA., et al. “Thyroid gland as an initial site of delayed metastasis from renal cell carcinoma: a case report.” Journal of Solid Tumors 2.4 (2012): 50-52.
- Rizzo M., et al. “Thyroid metastasis of clear cell renal carcinoma: report of a case.” Diagnostic Cytopathology 37.10 (2009): 759-762.
- P Madore and S Lan. “Solitary thyroid metastasis from clear-cell renal carcinoma.” Canadian Medical Association Journal 112.6 (1975): 719-721.
- Heffess CS., et al. “Metastatic renal cell carcinoma to the thyroid gland: a clinicopathologic study of 36 cases”. Cancer 95.9 (2002): 1869-1878.
- Kim TY., et al. “Metastasis to the thyroid diagnosed by fine-needle aspiration biopsy”. Clinical Endocrinology 62.2 (2005): 236-241.
- Chung AY., et al. “Metastases to the thyroid: a review of the literature from the last decade”. Thyroid 22.3 (2012): 258-268.
- Lehur PA., et al. “Thyroid metastasis of clear-cell renal carcinoma”. Canadian Medical Association Journal 28.2 (1983): 154-156.
- Bohn OL., et al. “Tumor-to-tumor metastasis: renal cell carcinoma metastatic to papillary carcinoma of thyroid. Report of a case and review of the literature”. Head and Neck Pathology 3.4 (2009): 327-330.
- Yoon JH., et al. “Sonographic features and ultrasonography-guided fine-needle aspiration of metastases to the thyroid gland”. Ultrasonography 33.1 (2014): 40-48.
- Ramirez-Plaza CP., et al. “Thyroid metastasis as initial presentation of clear cell renal carcinoma”. International Journal of Surgery 10 (2015): 101-103.
- Wada N., et al. “Solitary metachronous metastasis to the thyroid from renal clear cell carcinoma 19 years after nephrectomy: report of a case”. Surgery Today 35.6 (2005): 483-487.
- Surov A., et al. “Radiological features of metastases to the thyroid”. Acta Radiologica 57.4 (2016): 444-450.
- Macedo-Alves D., et al. “Thyroid metastasis from renal cell carcinoma-A case report after 9 years”. International Journal of Surgery Case Reports 16 (2015): 59-63.
- Iesalnieks I., et al. “Renal cell carcinoma metastases to the thyroid gland-8 cases reported”. Thyroid 17.1 (2007): 49-52.
- Song OK., et al. “Metastatic renal cell carcinoma in the thyroid gland: ultrasonographic features and the diagnostic role of core needle biopsy”. Ultrasonography 36.3 (2017): 252-259.
- Hegerova L., et al. “Metastasis to the thyroid gland: report of a large series from the Mayo Clinic”. American Journal of Clinical Oncology 38 (2015): 338-342.
- 24. Duggal NM and Horattas MC. “Metastatic renal cell carcinoma to the thyroid gland”. Endocrine Practice 14.8 (2008): 1040-1046
- Moon WJ., et al. “The value of gross visual assessment of specimen adequacy for liquid-based cytology during ultrasound-guided, fine-needle aspiration of thyroid nodules”. Endocrine Practice 21.11 (2015): 1219-1226.
- Riaz K., et al. “Renal cell carcinoma metastatic to thyroid gland, presenting like anaplastic carcinoma of thyroid”. Urology Case Reports (2013): 651081.
Citation:
Khalid S Aljabri., et al. “Thyroid Metastasis from Renal Cell Carcinoma”. Archives of Endocrinology and Diabetes Care 1.2
(2018): 65-70.
Copyright: © 2018 Khalid S Aljabri., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.