Research Article
Volume 1 Issue 2 - 2017
Effectiveness of Natural Herbal Oil Formulation against Infectious Myonecrosis Virus in Whiteleg Shrimp Penaeus vannamei
1PT Central Proteina Prima, Indonesia
2Panacea Natural Sciences, Indonesia
2Panacea Natural Sciences, Indonesia
*Corresponding Author: Rajeev Kumar Jha, PT Central Proteina Prima, Indonesia.
Received: February 17, 2017; Published: May 10, 2017
Abstract
Aim: Infectious Myonecrosis Virus (IMNV) of Penaeus vannamei shrimp is considered as a chronic virus. An effort was made to develop an anti-Infectious Myonecrosis Virus formulation by using the combination of oil blends.
Material and Methods: Altogether 10 plant oils extract Lavandulalatifolia, Pinussylvestris, Jasminumofficinale, Citrus limon, Prunumsavium, Viola odorata, Gardenia jasminoides, Cocos nucifera, Rosa damascene and Eucalyptus globulus,were selected as anti-IMNV candidates. The oil blends were mixed with the shrimp feed in required quantity. The experimental shrimp were fed on the blend oil enriched feed for 14 days prior to challenge. The per os challenge method was applied to infect the shrimp. Simultaneously, to support the feed, a liquid supplement named as “ Pondguard” (Reg no. KKP RI No. D16060285-HBC) was applied directly in the tank water.
Results: The cumulative mortality recorded in positive control was 58.3% whereas 8.3% in the exerimental group. The rate of mortality in experimental group were significantly lesser than positive control group. The typical gross sign appearance i.e. whitish muscle in 2nd and 3rd segment, in the control group were higher than the treatment.
Conclusion: The trial results showed that the developed blended oil extract formulation has significant effect against IMNV in controlled conditions. The developed product is user friendly as it is mixed and incorporated into the shrimp feed in specific amount to have anti-viral properties.
Introduction
Infectious myonecrosis is known to cause significant outbreak and mortalities in penaeid shrimp [1]. In Indonesia the first outbreak was in Situbondo in 2006, and it spread to all over the country: East Java, Bali, Lampung, Central Java, West Kalimantan and West Nusa Tenggara [3]. In Brazil IMNVcaused up to 70% mortality [2]. IMNV has been demonstrated to be horizontally transmitted by ingestion of infected moribund shrimp [4], and live feed [8].
The aquaculture experts have been continuously trying to control the spread of the virus by proper husbandary practices, uses of specific pathogen free (SPF) broodstock [3,8] and uses of rapid and sensitive detection kits [2]. The IMN Virus oppositive to the characteristics of white spot syndrome virus, causes chronic mortality in the ponds. It results in overall bigger losses due to high feed conversion ratio, lower survival rate and so the higher cost of production. The IMNV infected shrimp in stressed conditions show typical white or red muscle in the 5th to 6th segment. Its hard for the shrimp with white muscle to recovered in the cultured ponds. The herbs are recognized to have potential for preparing clinically useful drugs to effectively prevent and control many viruses of aquaculture origin [6]. Various plants and plant origin products have shown significant effect against pathogen specially in terms of reducing the mortality ofviral disease in shrimp, such as Phyllanthus niruriincreased the survival rate against IMNV from 66.67% to 86.67% [7].
A successful effort has been made todevelop a formulation by adding essential oil blend from ten plants and mixed it with the shrimp feed. The feed was supported by a liquid formulation, named as Pondguard, containing three essential oil extract. A trial was conducted to determine the efficacy of feed with Natural Herbal Oil formulation and Pondguard against IMNV in Penaeusvannamei.
Materials and Methods
Bioassay lab and Glass Aquaria
The trial was conducted at the Bioassay laboratory of Disease Research Centre, PT. CP PRIMA Indonesia. A bioassay trial was set-up using 10 aquaria for each group. The Specific Pathogen Free (SPF) juvenile shrimp 4 pieces with average weight 1.9g were reared in each aquarium. The glass aquaria were cleaned, sun dried and disinfected with 70% alcohol. Each aquarium was filled with 5 liters of seawater provided with required Dissolved Oxygen (DO) supply (> 5 ppm).
The trial was conducted at the Bioassay laboratory of Disease Research Centre, PT. CP PRIMA Indonesia. A bioassay trial was set-up using 10 aquaria for each group. The Specific Pathogen Free (SPF) juvenile shrimp 4 pieces with average weight 1.9g were reared in each aquarium. The glass aquaria were cleaned, sun dried and disinfected with 70% alcohol. Each aquarium was filled with 5 liters of seawater provided with required Dissolved Oxygen (DO) supply (> 5 ppm).
Water Quality parameters
The seawater utilized during the experiment were prepared by following the process of sedimentation, filtration and disinfection by (30 ppm active chlorine) and finally treated with Ultra Violet radiation (SS-L75W, Matala Water Technology, Taiwan). The water quality parameters maintained during the trial were as followed, dissolved oxygen (DO) (> 5 ppm), Temperature (27 ± 10C), Salinity (20 ppt), pH (7.5-8.15) and alkalinity (80-120 ppm).
The seawater utilized during the experiment were prepared by following the process of sedimentation, filtration and disinfection by (30 ppm active chlorine) and finally treated with Ultra Violet radiation (SS-L75W, Matala Water Technology, Taiwan). The water quality parameters maintained during the trial were as followed, dissolved oxygen (DO) (> 5 ppm), Temperature (27 ± 10C), Salinity (20 ppt), pH (7.5-8.15) and alkalinity (80-120 ppm).
Experiment start
The duration of trial was of 30 days i.e. 5 days acclimatization, 14 days anti-IMNV feed feeding and Pondguard application and observation, viral challenge on day 15 and then feeding and observation for 10 days. Shrimp of treatment group were fed on experimental feed whereas regular feed was fed to the control. There was no change in feed type and quality and the rate of feeding throughout the experiment. The liquid supplement, Pond guard was applied on daily basis in experimental tanks throughout the experiment. The shrimp were observed intensively for their feeding rate and cumulative mortality during the trial.
The duration of trial was of 30 days i.e. 5 days acclimatization, 14 days anti-IMNV feed feeding and Pondguard application and observation, viral challenge on day 15 and then feeding and observation for 10 days. Shrimp of treatment group were fed on experimental feed whereas regular feed was fed to the control. There was no change in feed type and quality and the rate of feeding throughout the experiment. The liquid supplement, Pond guard was applied on daily basis in experimental tanks throughout the experiment. The shrimp were observed intensively for their feeding rate and cumulative mortality during the trial.
Shrimp Food
The shrimp feed was produced in Feed Mill of PT. Central Proteina Prima, Indonesia. Treatment group of shrimp was fed on experimental feed i.e. CPPregular feed mixed with Natural Herbal Oil Formulation (NHOF)and control group on regular feed. The anti-IMNV NHOF were developed by combining the essential oil blend extracted from the following ten plants, Lavandulalatifolia, Pinussylvestris,Jasminumofficinale, Citrus limon, Prunusavium, Viola odorata, Gardenia jasminoides, Cocosnucifera, Rosa damascene,and Eucalyptus globulus. The expeller-pressing method (Anderson International Corp) was performed for the oil extractions from the selected plants. The essential oil blend were mixed with the feed in required amount. The basic formulation of both the feed were same except essential oil mixed in the experimental feed.
The shrimp feed was produced in Feed Mill of PT. Central Proteina Prima, Indonesia. Treatment group of shrimp was fed on experimental feed i.e. CPPregular feed mixed with Natural Herbal Oil Formulation (NHOF)and control group on regular feed. The anti-IMNV NHOF were developed by combining the essential oil blend extracted from the following ten plants, Lavandulalatifolia, Pinussylvestris,Jasminumofficinale, Citrus limon, Prunusavium, Viola odorata, Gardenia jasminoides, Cocosnucifera, Rosa damascene,and Eucalyptus globulus. The expeller-pressing method (Anderson International Corp) was performed for the oil extractions from the selected plants. The essential oil blend were mixed with the feed in required amount. The basic formulation of both the feed were same except essential oil mixed in the experimental feed.
Pondguard Application
A liquid product called Pondguard (KKP RI NO. D 16060285-HBC) was developed as a water supplement. The Pondgard is composed of three essential oils i.e. Eucalyptus globulus, Pinussylvestris and Lavandulalatifolia. It was applied in the treatment tank at the rate of 0.2 ppm on daily basis after tank cleaning and water exchange (approximately 20% per day).
A liquid product called Pondguard (KKP RI NO. D 16060285-HBC) was developed as a water supplement. The Pondgard is composed of three essential oils i.e. Eucalyptus globulus, Pinussylvestris and Lavandulalatifolia. It was applied in the treatment tank at the rate of 0.2 ppm on daily basis after tank cleaning and water exchange (approximately 20% per day).
IMNVDisease Challenge procedure
Per-oschallenge was appliedin the trial. The method in detail were as follows:
Per–os method of challenge :The muscle to use in per os challenge was prepared from the artificially IMNV challenged SPF shrimp. To prepare it, SPF shrimp were challenged by lethal dose (log 5) of IMNV by injection method. The typical gross sign of IMNV i.e. white muscle, started appearing after seven days of challenge. The moribund shrimp were collected, peeled and de-veined. The muscle were blend and homozenized. The viral load was checked by Real-Time PCR. The collected tissue containing viable IMNV were stored at -800C by adding sterile glyserol till further use.
Per-oschallenge was appliedin the trial. The method in detail were as follows:
Per–os method of challenge :The muscle to use in per os challenge was prepared from the artificially IMNV challenged SPF shrimp. To prepare it, SPF shrimp were challenged by lethal dose (log 5) of IMNV by injection method. The typical gross sign of IMNV i.e. white muscle, started appearing after seven days of challenge. The moribund shrimp were collected, peeled and de-veined. The muscle were blend and homozenized. The viral load was checked by Real-Time PCR. The collected tissue containing viable IMNV were stored at -800C by adding sterile glyserol till further use.
On the day of challenge, the shrimps of all the groups were not fed on artificial feed. The muscle containing IMNV were fed to shrimp at the rate of 5% MBW as morning meal to experimental and positive control whereas non-infected tissue was fed to the negative control shrimp.
Post challenge observation
The challenged shrimp were under intensive observation. The feeding rate and tank bottom-siphoning schedule were maintained the same for control and treatment group throughout the experiment. There was no water exchangein the tanks on the day of IMNV-challengefor both control and experimental group.
The challenged shrimp were under intensive observation. The feeding rate and tank bottom-siphoning schedule were maintained the same for control and treatment group throughout the experiment. There was no water exchangein the tanks on the day of IMNV-challengefor both control and experimental group.
Laboratory confirmation
The IMNVwas confirmed by nested PCR analysis and by histopathology test. The IQ 2000 PCR detection kit from Genreach, Taiwan utilized for the viral detection. The histopathology analysis was conducted at Central Laboratory of PT. CP PRIMA, Indonesia.
The IMNVwas confirmed by nested PCR analysis and by histopathology test. The IQ 2000 PCR detection kit from Genreach, Taiwan utilized for the viral detection. The histopathology analysis was conducted at Central Laboratory of PT. CP PRIMA, Indonesia.
Statistical analysis
Statistical analysis was performed by ANOVA with P < 0.05 confidence level using the programme SPSS.
Statistical analysis was performed by ANOVA with P < 0.05 confidence level using the programme SPSS.
Experimental design
| Group | Challenge | Rep. | MBW (g); Desnity/ tank | Feed | Pondguard | Water Exchange | |
| Type | Amount | ||||||
| 1 | Positive Control | 3 | 1.9 g; 4 pcs |
Regular | 5 % of biomass. 4 times/day | - | 20%/day (no water exchange on the day of challenge) |
| 2 | Treatmtment | NHOF enriched | 0.2 ppm.application in every 24 hours | ||||
| 3 | Negative Control | Regular | - | ||||
Table 1: Experimental design of anti-IMNV trial.
Result and Discussion
A successful effort was made to develop anti-IMNV product. The developed product is a combination of blend of essential oils mixed in the feed and also in liquid form (Pondguard). The artificial feed is served as one of the best carriers of anti-viral product. The Pondguard, which was directly poured in the tank water, supplemented the protection level. It was done with the to reduce the gross sign during the stressed condition specially when feed consumption dropped. The Pondguard was applied daily to maintain the optimum protection level. The experimental shrimp were applied NHOF in the form of feed and as liquid supplement for the first fourteen days to develop sufficient level of protection. The gross sign data in figure 1. showed after the challenge. The gross sign started to appear on Day Post Infection DPI 4 in positive control and reached 83.3% on DPI 14, while in the treatment group the gross sign occured on DPI 12 and the highest percentage of gross sign recorded was 16.7%. The histopathology analysis ( Figure 3 and 4) and PCR test (Table 3) results showed that the positive control shrimp were of grade severe to grade light positive to IMNV whereas the survived shrimp in the experimentalgroup were grade light positive and negative control group were detected negative to IMNV.It indicated that blend oil mixed feed was able to reduce IMN Virus multiplication and so the viral load in the shrimp body.
The per osviral challenge methodwas applied as its mimic of natural mode of infection in the environment. To avoid chances of contamination, the IMNV infected tissue was prepared by challenging SPF shrimp. The pure IMNVextract was used to infect the SPF shrimp and then the tissue of artificially diseased shrimp were utilized for per os challenge. Higher rate of cumulative mortalityrecorded in positive control than the experimental group.Thefirst mortality recorded on day 11 of challenge in the experimental feed groupswhich showed the anti-viral activities of the product compared to the positive control group where mortality started on day 4 of challenge. It shows that the viral multiplication slowed down in the presence of NHOF. It took longer period of time for the IMNV to reach at virulence level in treatment group as compared to control. The appearance of white muscle in IMNV infected shrimp is the typical sign. The white muscle appearance occur due to death of tissue in that region. The regeneration of tissue happens in the non-stressed condition and normally it takes 3-4 weeks. The chances of recovery would be less in stressed condition of the shrimp.
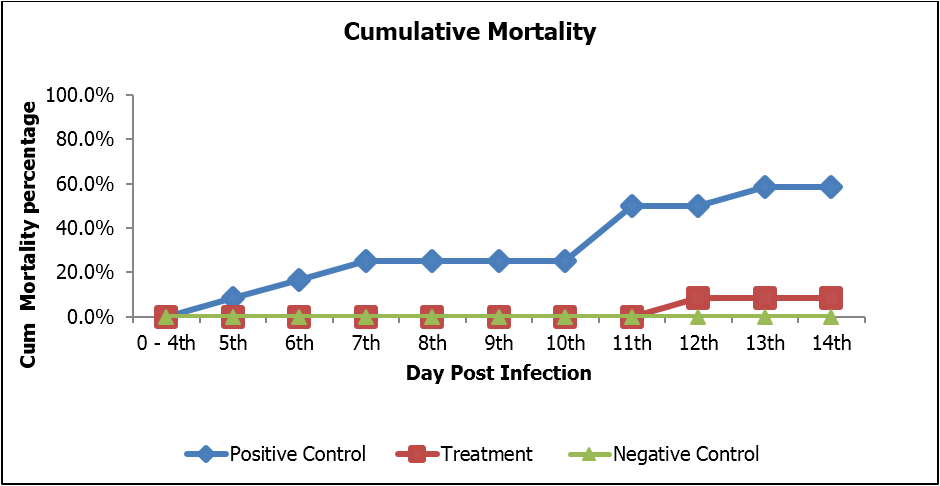
There was significant difference between experimental group and control group (Figure 2).
Figure 2: Cumulative Mortality percentage of experimental and control shrimp.
Figure 2: Cumulative Mortality percentage of experimental and control shrimp.
Gross Sign
Duncana
Duncana
| Treatment | N | Subset for Alpha = 0.05 | |
| 1 | 2 | ||
| Negative Control | 3 | 0 | |
| Treatment | 3 | 16.67 | |
| Positive Control | 3 | 83.33 | |
| Sig. | 0.315 | 1 | |
Means for groups in homogeneous subsets are displayed
a. Uses Harmonic Mean Sample Size = 3.000
Cumulative Mortality
Duncana
a. Uses Harmonic Mean Sample Size = 3.000
Cumulative Mortality
Duncana
| Treatment | N | Subset for Alpha = 0.05 | |
| 1 | 2 | ||
| Negative Control | 3 | 0 | |
| Treatment | 3 | 8.333 | |
| Positive Control | 3 | 58.333 | |
| Sig. | 0.42 | 1 | |
Means for groups in homogeneous subsets are displayed
a. Uses Harmonic Mean Sample Size = 3.000
Table 2: ANOVA test results of Gross sign appearance and Cumulative mortality.
a. Uses Harmonic Mean Sample Size = 3.000
Table 2: ANOVA test results of Gross sign appearance and Cumulative mortality.
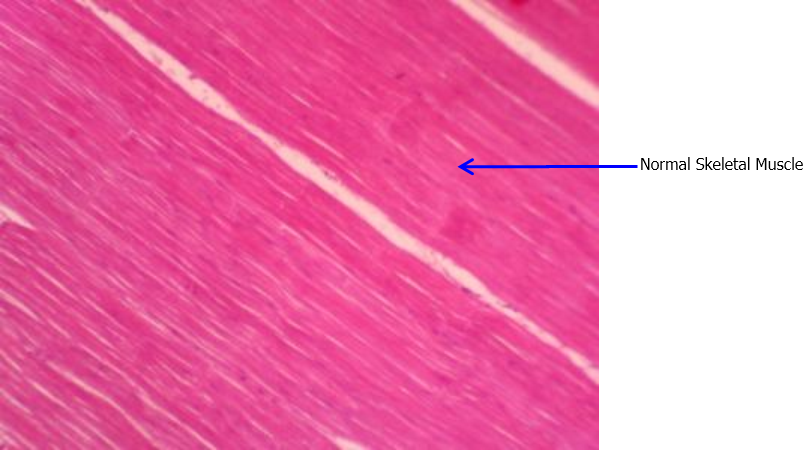
Figure 3: Histopathology observation: Normal Skeletal Muscle in treatment group. Slides
fixed in Longitudinal 4 μm paraffinsection, H & Estain, Davidson fixative, 100x.
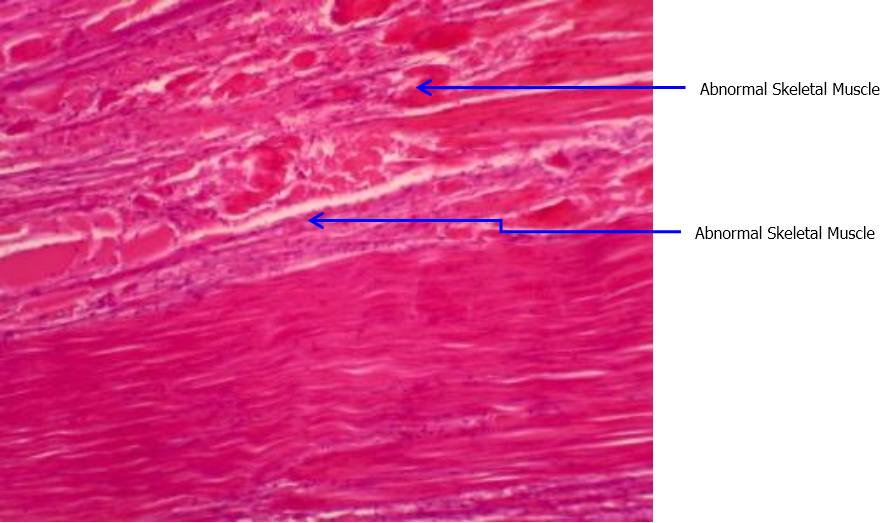
Figure 4: Histopathology observation: Abnormal Skeletal Muscle in positive
control group, showing sign of necrosis and hemocyte infiltration. Slides fixed in
Longitudinal 4 μm paraffinsection, H & Estain, Davidson fixative, 100x.
| Group | Replication | PCR Results | |
| IMNV | Severity | ||
| Positive Control | 1 | Positive | Medium |
| 2 | Positive | Light | |
| 3 | Positive | Severe | |
| Treatment | 1 | Positive | Light |
| 2 | Positive | Light | |
| 3 | Positive | Light | |
| Negative Control | 1 | Negative | |
| 2 | Negative | ||
| 3 | Negative | ||
Table 3: Nested PCR results of control and experimental groups.
The hispathological analysis (Figure 3 and 4) of survived shrimp from treatment and positive control showed clear difference. The slides of positive control group showed abnormal muscle in positive control group, showing sign of necrosis and hemocyte infiltration, which is the typical sign of IMNV infection. Likewise, The PCR analysis report (Table 3) showed that the positive control survived shrimp were positive to IMNV whereas the treatment and negative control shrimp were negative to IMNV. The nested two-step PCR test was done which is semi-quantitive test as it shows the severity level. The severity level is showed in the table as light (less no of virus), medium and severe (more numner of virus).
The current experiement showed the positive sign in terms of significantly less no of shrimp with white muscle, typical clinical sign of IMNV and less mortality in the NHF treated feed. More efforts would be required to achive the complete protection against IMNV.
Acknowledgements
We thank PT. Central Proteina Prima, Indonesia to provide funding for the research.
We thank PT. Central Proteina Prima, Indonesia to provide funding for the research.
Conflict of Interest
The authors declare that they have no conflicts of interest.
The authors declare that they have no conflicts of interest.
References
- Lightner DV., et al. “Infectious Myonecrosis: New Disease InPacific White Shrimp”. Global Aquaculture Advocate 7.85 (2004):
- Melo MVC., et al. “Molecular characterization of infectious myonecrosis virus (IMNV) isolatedfrom the shrimp Litopenaeusvannamei farmed in Ceará State, Brazil”. Latin American Journal of Aquatic Research 42.3 (2014): 649-652.
- Network of Aquaculture Centres in Asia-Pacific (NACA). Disease Advisory: InfectiousMyonecrosis (IMN): Status and Threat (2010).
- OIE. Manual of Diagnostic Tests for Aquatic Animals (2009).
- Poulos BT., et al. “Purification and Characterization Of Infectious Myonecrosis Virus OfPenaeid Shrimp”. The Journal of General Virology 87(Pt 4) (2006): 987–996.
- Sivasankar P., et al. “A Review on Plants and Herbal Extracts Against Viral Diseases in Aquaculture”.Journal of Medicinal Plants Studies 3.2 (2015): 75-79.
- Sukenda S., et al. “Administration of Phyllanthus niruri to control IMNV (myonecrosis infectious virus) infection white shrimp Litopenaeus vannamei”. Jurnal Akuakultur Indonesia 10.2 (2011): 192‒202.
- Taukhid and Nur’aini. “Infectious Myonecrosis Virus (IMNV) in Pacific White Shrimp (Litopenaeus vannamei) in Indonesia”.The Israeli Journal of Aquaculture - Bamidgeh 61.3 (2009): 255-262.
Citation:
Rajeev Kumar., et al. “Effectiveness of Natural Herbal Oil Formulation against Infectious Myonecrosis Virus in Whiteleg Shrimp
Penaeus vannamei”. Multidisciplinary Advances in Veterinary Science 1.2 (2017): 50-56.
Copyright: © 2017 Rajeev Kumar., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.