Research Article
Volume 1 Issue 5 - 2017
Epidemiology of Thermophilic Campylobacter Species in Rural Poultry in Kebbi State, Nigeria
1Department of Animal Health and Production Technology, Federal Polytechnic Mubi. Adamawa State. Nigeria
2Department of Veterinary Medicine, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria
3Department of Veterinary Medicine and Surgery, Faculty of Veterinary Medicine, Federal University of Agriculture, Abeokuta, Ogun State, Nigeria
4Department of Veterinary Microbiology, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Sokoto State, Nigeria
2Department of Veterinary Medicine, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria
3Department of Veterinary Medicine and Surgery, Faculty of Veterinary Medicine, Federal University of Agriculture, Abeokuta, Ogun State, Nigeria
4Department of Veterinary Microbiology, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Sokoto State, Nigeria
*Corresponding Author: Abubakar SM Abba Maiha, Department of Animal Health and Production Technology, Federal Polytechnic Mubi. Adamawa State. Nigeria.
Received: December 15, 2017; Published: December 27, 2017
Abstract
A study was conducted to determine the prevalence of thermophilic Campylobacter species in the four Emirate councils of Kebbi State. A total of 400 cloacal swabs from domestic birds were screened and analyzed using standard culture isolation technique and biochemical characterization. A total of 177 (44%) were positive for Campylobacter species. The prevalence of 89%, 51% and 37% were recorded for Campylobactercoli, C. lari and C. jejuni respectively.
The prevalence of 46% and 38% were recorded in chicken and guinea fowl, while 28%, 53% and 50% were found in pigeon, duck and turkey respectively. Sex specific prevalence was slightly higher in males than females. Using Chi square analysis, there was no statistical significant association between sex, species and infection (p-value > 0.05). The prevalence in the four Emirate councils revealed 58%, 53%, 36% and 30% in Gwandu, Argungu, Yauri and Zuru respectively. There was statistical association between the selected local governments in the Emirate councils and infection (p-value < 0.05). Poultry in the state have been shown to harbor Campylobacterspecies and may serve as reservoir of infection for others animals and humans. The transportation of poultry along with human passengers in the same vehicle while moving birds from different locations to live bird markets should be discouraged. Adequate environmental sanitation and strict hygiene measures should be implemented in the backyard poultry houses, slaughter slabs and processing units in the state.
Keywords: Epidemiology; Campylobacter; Poultry
Introduction
Campylobacter is a major cause of human enteritis, and food-borne Campylobacteriosis is considered a main problem of Public Health in many developed countries. Poultry products are suspected to be an important source of infection in many countries (Anon., 2000). The thermophilic or thermotolerant Campylobacter are those that require a slightly higher temperature for their growth than other Campylobacter species and they include; C. jejuni, C. coli and C. lari found in avians (Skirrow, 1994; Rosef., et al. 2001). Avian carriage of Campylobacter has been regarded as a potential hazard to animals and human health, either through consumption of raw or undercooked carcass or by contamination of water supplies (Varslot., et al. 1996). A wide variety of avian species, including domestic Chickens, turkeys, ducks, pigeons, quail, waterfowls, geese and ostriches, harbor Campylobacter species (Broman., et al. 2004).
However, they are unevenly distributed among species and the feeding behaviour of birds has been shown to influence the Campylobacter colonization rate (Alterkruse., et al. 1999; Waldenstrom., et al. 2003). The Nigerian domestic birds are raised on a small scale in most households in rural and semi-urban areas of Northwestern Nigeria. They are free range poultry (rural poultry) made up of mostly chickens, ducks and guinea fowls, which are domesticated for the purpose of eggs and meat production.
These birds are managed under the extensive systems (Emikpe., et al. 2005), and are usually not vaccinated (Adu., et al. 1986). It is widely believed that they act as reservoirs of most important poultry diseases, including Campylobacter infection (Bouzoubaa., et al. 1992). Chickens (Gallus gallus domesticus) are the most important of the free range poultry species in terms of number and development (Oluyemi and Roberts, 1979). They are kept by over 90% of rural households, especially women, as assets (Ajala., et al. 2007), providing an important source of high quality protein as well as a source of income for the families (Abubakar., et al. 2008).
Poultry being one of the sources of infection in animal and human, ecological approach is required to understand the epidemiology of the infection(Engberg., et al. 2001; Alfredson and Korolik, 2007; Han., et al. 2007). The aim of the study was to determine the prevalence of thermophilic Campylobacter species in rural poultry in Kebbi State, Northwestern Nigeria.
Materials and Methods
Study area
Kebbi State is geographically located to the North Western part of Nigeria at 11° 30’N 4° 00’E. Kebbi State falls within the Sudan Savanna with mean minimum temperature of 26°C (Kowal and Knabe, 1992). During the harmattan season (December to February), the temperature can go down to as low as 21°C and mean maximum temperature can go up to 40°C during the months of April to June (MANR, 1999). Annual rainfall is about 800mm and relative humidity is low (40%) for most of the year except during the wet season when it reaches an average of 80%. The wet season lasts from June to September, the hot season April to June while cool dry season lasts from December to February (Odjugo, 2010). Kebbi State was ranked among the five states with the highest number of livestock in Nigeria. Agriculture is the main occupation of the people especially in rural areas (animal rearing and fishing).
Kebbi State is geographically located to the North Western part of Nigeria at 11° 30’N 4° 00’E. Kebbi State falls within the Sudan Savanna with mean minimum temperature of 26°C (Kowal and Knabe, 1992). During the harmattan season (December to February), the temperature can go down to as low as 21°C and mean maximum temperature can go up to 40°C during the months of April to June (MANR, 1999). Annual rainfall is about 800mm and relative humidity is low (40%) for most of the year except during the wet season when it reaches an average of 80%. The wet season lasts from June to September, the hot season April to June while cool dry season lasts from December to February (Odjugo, 2010). Kebbi State was ranked among the five states with the highest number of livestock in Nigeria. Agriculture is the main occupation of the people especially in rural areas (animal rearing and fishing).
Research design
The study was a cross sectional study of Campylobacter infection in domestic birds. One hundred samples were collected from domestic birds at poultry markets from each of the selected four local government areas (Argungu, Birni Kebbi, Yauri and Zuru). Each was selected from one of the four local governments in the state. Random sampling techniques were used in sample collection.
The study was a cross sectional study of Campylobacter infection in domestic birds. One hundred samples were collected from domestic birds at poultry markets from each of the selected four local government areas (Argungu, Birni Kebbi, Yauri and Zuru). Each was selected from one of the four local governments in the state. Random sampling techniques were used in sample collection.
Sampling method
Domestic birds at live bird Markets were the target population while poultry at live Bird Markets were the sampling frame. Purposive sampling as described by Paul (2006) was used for selection of local governments’ areas while simple random sampling as described by Valerie and John (1997) was used for sampling domestic birds in selected areas.
Domestic birds at live bird Markets were the target population while poultry at live Bird Markets were the sampling frame. Purposive sampling as described by Paul (2006) was used for selection of local governments’ areas while simple random sampling as described by Valerie and John (1997) was used for sampling domestic birds in selected areas.
Sample size determination
The minimum sample size for this study was determined by the formula,
n = t2x pexp(1-pexp )/d2 (Thrusfield, 2005)
Where n = sample size, t2 = the score for a giving interval which is 1.96 (S.E) at 95%, confidence interval, pexp = Known or estimated prevalence, and d2 = precision at 0.05.
The samples were calculated at 38.8% prevalence, (Salihu., et al. 2009) at 95% confidence interval, with desired precision of 5%.
n = (1.96)2 x 0.39 x (1-0.39)/(0.05)2,
n = 0.9139/0.0025 = 356.5
n = 366
For more precision of the study, 400 samples were collected.
Thus, n = 400
The minimum sample size for this study was determined by the formula,
n = t2x pexp(1-pexp )/d2 (Thrusfield, 2005)
Where n = sample size, t2 = the score for a giving interval which is 1.96 (S.E) at 95%, confidence interval, pexp = Known or estimated prevalence, and d2 = precision at 0.05.
The samples were calculated at 38.8% prevalence, (Salihu., et al. 2009) at 95% confidence interval, with desired precision of 5%.
n = (1.96)2 x 0.39 x (1-0.39)/(0.05)2,
n = 0.9139/0.0025 = 356.5
n = 366
For more precision of the study, 400 samples were collected.
Thus, n = 400
Sample collection
Permission was obtained from the Ministry of Agriculture and Natural Resources and for each of the selected market 2 in every 5 bird (40%) counted were randomly sampled. A total of 400 domestic birds were sampled at poultry markets from four of the randomly selected local government, each from one of the four Emirate councils in the state. Cloacal swabs or freshly voided faeces were collected using sterile commercial swab sticks and were placed in Amies transport media, kept cold with the use of ice blocks (Butzler, 2004). Samples were transported within few hours after collection on the same day to the Veterinary Microbiology Laboratory, Faculty of Veterinary Medicine, Usmanu Danfodio University, and Sokoto for processing.
Permission was obtained from the Ministry of Agriculture and Natural Resources and for each of the selected market 2 in every 5 bird (40%) counted were randomly sampled. A total of 400 domestic birds were sampled at poultry markets from four of the randomly selected local government, each from one of the four Emirate councils in the state. Cloacal swabs or freshly voided faeces were collected using sterile commercial swab sticks and were placed in Amies transport media, kept cold with the use of ice blocks (Butzler, 2004). Samples were transported within few hours after collection on the same day to the Veterinary Microbiology Laboratory, Faculty of Veterinary Medicine, Usmanu Danfodio University, and Sokoto for processing.
Processing of samples
Samples were inoculated directly onto a selective medium, modified charcoal cefaperazone Deoxycholate Agar (mCCDA) and incubated at 42°C for 48hrs (Butzler and Megraud, 2002). Suspected Campylobacter colonies on the selective mCCDA medium were identified based on their characteristics features as creamy or white, moist, flat or slightly raised, extending along the streak line, or regular circular discrete colony based on the description of Atabay and Corry (1998).
Samples were inoculated directly onto a selective medium, modified charcoal cefaperazone Deoxycholate Agar (mCCDA) and incubated at 42°C for 48hrs (Butzler and Megraud, 2002). Suspected Campylobacter colonies on the selective mCCDA medium were identified based on their characteristics features as creamy or white, moist, flat or slightly raised, extending along the streak line, or regular circular discrete colony based on the description of Atabay and Corry (1998).
Suspected Campylobacter isolates were confirmed based on their biochemical reactions as follows: Oxidase test, Hippurate hydrolysis test, Catalase test, Hydrogen sulphide production test (Atabay and Corry, 1998) and sensitivity to Cephalothin, Nalidixic acid using agar disc diffusion method (CLSI, 2014.
Results
Out of the 400 samples analyzed, a total of 177 samples were positive for Campylobacterspp. The prevalence of 89%, 51% and 37% were recorded for C. coli, C. lariand C. jejuni, respectively (Table 1). The prevalence of 46% and 38% was recorded in both chicken and guinea fowl while 28%, 53% and 50% were recorded for pigeon, ducks and turkey respectively (Table 1).C. colirecorded high rates in guinea fowl and ducks with 60 and 64%, respectively (Table 1).
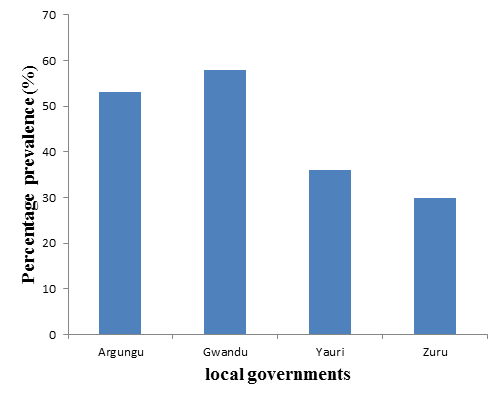
Sex-specific prevalence of 98 (47%) and 79 (40%) were recorded for males and females, respectively (Table 2). C. colihad a higher prevalence than other species of Campylobacterin both male and female (Table 2). Species to specific prevalence in the selected local governments showed that C. colihad the prevalence of 89(22.3%) which is higher than 51(12.8%) and 37(9.3%) recorded for C. lariand C. jejuni, respectively (Table 3). The prevalence per Emirate council in Kebbi State has recorded 58%, 53%, 36% and 30% in Gwandu, Argungu, Yauri and Zuru Local governments respectively (Table 4). Figure 1 represent percentage prevalence of Campylobacter per local governments. There was no association (P > 0.05) between prevalence rate, species and sex in poultry, but the association (P < 0.05) between prevalence and local governments were statistically significant.
| Species | Total sampled | Total positive (%) | C. jejuni (%) | C. coli (%) | C.lari (%) |
| Chicken | 278 | 129 (46) | 32 (24.8) | 61 (47.3) | 36 (27.9) |
| Guinea fowl | 52 | 20 (38.5) | 2 (10) | 12 (60) | 6 (30) |
| Pigeon | 36 | 10 (27.8) | 2 (20) | 4 (40) | 4 (40) |
| Duck | 32 | 17 (53) | 1 (5.9) | 11 (64) | 5 (29.4) |
| Turkey | 2 | 1 (50) | 0 (0) | 1 (50) | 0 (0) |
| Total | 400 | 177 (44) | 37 (9.3) | 89 (22.3) | 51 (12.8) |
ϰ2 = 6.237 p = 0.182 p > 0.05
Table 1: Prevalence of Campylobacter species in different species of domestic birds in Kebbi State.
Table 1: Prevalence of Campylobacter species in different species of domestic birds in Kebbi State.
| Sex |
Total Number sampled | Total Number positive | Species | ||
| C. jejuni (%) | C. coli (%) | C.lari (%) | |||
| Male | 207 | 98(47) | 21(21.4) | 51(52.0) | 26(26.5) |
| Female | 193 | 79(40.9) | 16(20.0) | 38(48.1) | 25(31.6) |
| Total | 400 | 177(44) | 37 | 89 | 51 |
ϰ2 = 1.664 p = 0.1971 p > 0.05
Table 2: Sex specific prevalence of Campylobacter infection in Kebbi state.
Table 2: Sex specific prevalence of Campylobacter infection in Kebbi state.
| LGA | Total Sampled | Total Positive (%) | C. jejuni | Species C. Coli | C. lari |
| Argungu | 100 | 53 | 10 (18.9) | 26 (49.1) | 17 (17.1) |
| Birni Kebbi | 100 | 58 | 13 (22.4) | 29 (50) | 16 (27.6) |
| Yauri | 100 | 36 | 8 (22.2) | 17 (47.2) | 11 (30.6) |
| Zuru | 100 | 30 | 6 (20) | 17 (23.3) | 7 (23.3) |
| 400 | 177 | 37 (9.3) | 89 (22.3) | 51 (12.8) |
Table 3: Species to Specific Prevalence of Campylobacter infection in domestic birds in the selected local government areas.
| Emirate Councils | LGA | Total Sampled | Total Positive (%) | Total Negative (%) |
| Argungu | Argungu | 100 | 53 | 47 |
| Gwandu | Birni Kebbi | 100 | 58 | 42 |
| Yauri | Yauri | 100 | 36 | 64 |
| Zuru | Zuru | 100 | 30 | 70 |
| Total | 400 | 177 | 223 |
ϰ2 = 21.758 p = 0.001 p < 0.05
Table 4: Prevalence of Campylobacter infection in domestic birds in the selected local government areas.
Table 4: Prevalence of Campylobacter infection in domestic birds in the selected local government areas.
Discussion
The prevalence of Campylobacter species in domestic birds has been established in the study area. The 44% prevalence in domestic birds was higher than 30% recorded in domestic birds by Nwankwo., et al. (2016) in Sokoto State and 33% recorded in poultry in Lagos State by Uaboi-Egbenni., et al. (2008). The increased rate could be due to lack of awareness and poor environmental sanitation at backyard poultry houses at different homes, live bird markets and poultry farms.
The prevalence rate in domestic birds was also in agreement with that of Salihu., et al. (2009) that recorded 38.8% prevalence in Sokoto State. However, it differs with the high prevalence rates of 94.2 and 89% recorded by Workman., et al. (2005) and Georgios., et al. (2004) in chicken meat and faeces respectively. The similarities and variations in the prevalence rates could be a reflection of environmental contamination, however, other factors such as stock density, season, feeding regimen and geographical location have been proposed to account for significant differences and similarities in the isolation rates (Mary., et al. 2004).
The prevalence among species of domestic birds was high in ducks, which is a water fowl. Ducks are known to tip up on the surface of shallow water or submerge completely and swim under the water in search of food. They get infected especially when the ground water is contaminated with Campylobacter species (Savill., et al. 2001). The low prevalence recorded in chickens might be linked to the free range system which is common in the study area as coprophagy which enhances bird to bird spread is limited. This can be supported by findings of Robino., et al. (2010) with a Campylobacterspecies prevalence of 78.4% in intensively reared poultry and 18.3% in small scale rural poultry farming in Italy. The prevalence in pigeons, turkeys and guinea fowls also revealed the possibilities of infection through feeds as they usually feed on insects, fruits, seeds and flowers which have been suggested as potential sources of infection in poultry (Waldenstrom., et al. 2003). The findings in this study showed no statistical association between Campylobacter infections in domestic birds at the study area to species of birds.
The higher prevalence of C. colithan other species in this study agreed with the findings of Nwankwo., et al. (2016) who reported the prevalence of 53%, 28% and 18% for C. coli, C. lariand C. jejuniin domestic poultry respectively and Wieczorek., et al. (2012) that revealed 58.9% as C. coliand 41.1% as C. jejuni. Other reports on the higher isolation rate of C. colicompared to C. jejunihave also been reported (Lynch., et al. 2011).
However, the findings disagreed with the higher isolation rate of C. jejunithan other species in the work of Salihu., et al. (2009) that reported 72.9% of the total isolate from chicken as C. jejuniand Cuiwei., et al. (2001) who recorded the prevalence of 53.6%, 41.3% and 5.1% for C. jejuni, C. coliand other species respectively. Such differences have been attributed to several factors, including isolation method, sample size, seasonal variation and geographical location (Stanley., et al. 1998; Allos, 2001). Campylobacter colialso had high rate of 50% while C. jejuni had the lowest rate in chicken which was in agreement with the record of 50%, 29% and 19% as C. coli, C. Lari and C. jejuni respectively by Nwankwo., et al. (2016) in Sokoto State.
The isolation rate for C. lari in poultry in this study was in agreement with that of 28% by Baserisalehi., et al. (2007) in Iran. Furthermore, the lower isolation rate of C. lari to C. coliin this study was in agreement to the work of Uaboi-Egbenni., et al. (2008) who reported a zero rate of C. lariand 14.2% for C. coli. The prevalence of Campylobacter species may be dependent on the sample size and weather conditions of different areas as some species grow optimally during the hot temperature and high humidity. Other species such as C. hyointestinalis, C. sputorumand C. fetusnot found in the study were likely due to the high temperature of birds that do not support their survival, agents in the selective medium such as cefoperazone that might have hindered their growth and unsuitable temperature at 42°C used in the isolation (Martin., et al. 2002).
The different prevalence rates as recorded in the Local governments can be used as a reflection of weather conditions and environmental contamination in the areas. High prevalence rates were recorded in Gwandu and Argungu while low prevalence rates were recorded in Yauri and Zuru Local governments respectively.
There was no statistical significant difference in prevalence rates in male and female birds which is in agreement with the findings of Nwankwo., et al. (2016) and Salihu., et al. (2009) that recorded similar rates suggesting no sex preference in Campylobacterinfection.
Conclusion
The study revealed a total of 177 (44%) samples, were positive for Campylobacter species. The prevalence of 89%, 51% and 37% were recorded for C. coli, C. lari and C. jejuni, respectively. Campylobacter coli were more prevalence than C. jejuni. Domestic birds were infected independent of species and sex while different prevalence rates were recorded in different Local governments.
Acknowledgement
We are grateful to Dr. Yusuf Yakubu of the Department of Public Health and Preventive Medicine for his help in data analysis and the staff of Central Research Laboratory Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Abdulmalik Shuaibu Bello and Nafiu, for their technical assistance during sampling processing.
We are grateful to Dr. Yusuf Yakubu of the Department of Public Health and Preventive Medicine for his help in data analysis and the staff of Central Research Laboratory Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Abdulmalik Shuaibu Bello and Nafiu, for their technical assistance during sampling processing.
References
- Abubakar MB., et al. “Seoprevalence of active and passive immunity against egg drop syndrome 1976 (EDS 76) in village poultry in Nigeria”. Asian Journal of Poultry Science2 (2008): 58-61.
- Adu FD., et al. “Newcastle disease: the immunological status of Nigerian local chickens”. Tropical Veterenarian4 (1988): 149-152.
- Ajala MK., et al. “Socio-economic of free-range poultry production among agropastoral women in Giwa Local Government Area of Kaduna State, Nigeria”. Nigerian Veterinary Journal 28.3 (2007): 11-18.
- Alfredson DA and Korolik V. “Antibiotic resistance and resistance mechanisms in Campylobacter jejuni and Campylobacter coli”. FEMS Microbiology Letter 277.2 (2007): 123-132.
- Allos BM. “Campylobacter jejuniinfections: update on emerging issues and trends”. Clinical Infectious Disease 32.8 (2001): 1201-1206.
- Altekruse SF., et al. “Campylobacter jejuni. An emerging food borne pathogen”. Emerging Infectious Diseases 5.1 (1999): 28-35.
- Anon. Annual Report on Zoonoses in Denmark, 1999. Danish Zoonosis Centre, Danish Veterinary Laboratory, Copenhagen, Denmark (2000).
- Atabay HI and Corry JE. “Isolation and prevalence of Campylobacters from the dairy using variety of methods”. Journal of Applied Microbiology84.5 (1998): 733-740.
- Baserisalehi M., et al. “Isolation and characterization of Campylobacter spp. from domestic animals and poultry in South of Iran”. Pakistan Journal of Biological Science 10.9 (2007): 1519-1524.
- Bouzoubaa K., et al. “Village chickens as reservoir of Salmonella pullorum and Salmonella gallinarum in Morocco”. Preventive Veterinary Medicine 12.1.2 (1992): 95-100.
- Broman T., et al. “Diversities and similarities of PFGE profiles of Campylobacter jejuni isolated from migrating birds and humans”. Journal of Applied Microbiology 96.4 (2004): 834-843.
- Butzler JP. “Campylobacter, from obscurity to celebrity”. Clinical Microbiology and Infection 10.10 (2004): 868-876.
- Butzler JP and Megraud F. Campylobacter and Helicobacter pylori, In: Zinner S.H., Young, and L.S, Acar, J.P., Neus, and H.C., eds. expanding indication for the new macrolides, azalides and streptogramins. New York: (2002): 237-249.
- Clinical and Laboratory Standard Institute (CLSI), “Performance standards for antimicrobial disc and dilution susceptibility tests for bacteria isolated from animals”. Approved Standard s" ed. M31-A (2014).
- Cuiwei ZB., et al. “Prevalence of Campylobacter spp., Escherichia coli, and Salmonella Serovars in Retail Chicken, Turkey, Pork, and Beef from the Greater Washington DC, Area”. Applied and Environmental Microbiology67.12 (2001): 5431-5436.
- Emikpe BO., et al. “Serological evidence of chicken anaemia virus in Nigerian indigenous chickens”. Journal of Veterinary Research72.1 (2005): 101-103.
- Engberg J., et al. “Quinolone and macrolide resistance in Campylobacter jejuni and C. coli: resistance mechanisms and trends in human isolates”. Emergence Infectious Disease 7.1 (2001): 24-34.
- Georgios K., et al. “Use of PCR analysis and DNA microarrays for detection of Campylobacter jejuniand Campylobacter colifrom chicken faeces”. Journal of Clinical Microbiology 42.9 (2004): 3985-3991.
- Han K., et al. “Prevalence, genetic diversity and antibiotic resistance patterns of Campylobacter jejuni from retail raw chickens in Korea”. Journal of Food Microbiology 114.1 (2007): 50-59.
- Kowal JM and Knabe DT. “An agrodimatological atlas of the northern States of Nigeria with explanatory notes”. Ahmadu Bello University Press Zaria, Nigeria (1992).
- Lynch OA., et al. “Occurrence of fastidious Campylobacter spp. in fresh meat and poultry using an adapted cultural protocol”. International journal of Food Microbiology 150.2.3 (2011): 171-177.
- Martin KW., et al. “Evaluation of selective media for Campylobacter isolation when cycloheximide is replaced with amphotericin B”. Letter in Applied Microbiology 34.2 (2002): 124-129.
- Mary EP., et al. “Effects of Climate on Incidence of Campylobacter spp. in Humans and Prevalence in Broiler Flocks in Denmark”. Applied Environmental. Microbiology 70.12: 7474-7480.
- Ministry of Agriculture and Natural Resources (MANR): The Report, Nigeria Livestock Resources (1999).
- Nwankwo IO., et al. “Epidemiology of Campylobacter species in poultry and humans in the four agricultural zones of Sokoto State, Nigeria”. Journal of Public Health and Epidemiology8.9 (2016): 185-190.
- Odjugo PAO. “Regional evidence of climatic change in Nigeria”. Journal of Geography and Regional Planning 3.6 (2010): 142-150.
- Oluyemi JA and Roberts FA. “Poultry Production in Nigeria”. National Animal Production Research Institute Publication (1979): 163-186.
- Paul O. SAGE Research Method. The SAGE Dictionary of Social Research Methods (2006): 121-156.
- Robino P., et al. “Prevalence of Campylobacter jejuni, Campylobacter coli and enteric Helicobacter in domestic and free living birds in North-Western Italy”. Schweizer Archiv Fur Tierheilkunde152.9 (2010): 425-431.
- Rosef O., et al. “Thermophilic Campylobacter in surface water:a potential risk of Campylobacteriosis”. Internationa Journal of Environment Health Research11.4 (2001): 321-327.
- Salihu MD., et al. “Prevalence of Campylobacter in poultry meat in Sokoto, Northwestern Nigeria”. Journal of Public Health and Epidemiology 1.2 (2009): 41-45.
- Savill MG., et al. ”Elucidation of Campylobacter in New Zealand recreational and drinking waters”. Journal of Applied Microbiology 91.1 (2001): 38-46.
- Skirrow MB. “Diseases due to Campylobacter, Helicobacter and Related Bacteria”. Journal of Comparative Pathology 111.2 (1994): 113-214.
- Stanley KN., et al. “The seasonal variation of thermophilic Campylobacter in beef cattle, dairy cattle and calves”. Journal of Applied Microbiology 85.3 (1998): 472-480.
- Thrusfield M. Veterinary Epidemiology. 2nd ed. Blackwell publishing, 108 Cowley Road, Oxford OX4 IJF (2005): 180-198.
- Uaboi Egbenni PO., et al. “Epidemiological studies of the incidence of pathogenic Campylobacterspp. amongst animals in Lagos metropolis”. African Journal of Biotechnology7.16 (2008): 2852-2956.
- Valerie JE and John HM. Statistics Glossary 1.1(1997).
- Varslot M., et al. “Water-borne outbreaks of Campylobacter gastroenteritis due to pink-footed geese in Norway in 1994 and 1995”. Tidsskrift for Den Norske Laegeforening 116.28 (1996): 3366-3369.
- Waldenstrom J., et al. “Prevalence of Campylobacter jejuni, C. lari and C. coli in different ecological guids and taxa of migrating bird’s dagger”. Journal of Applied and Environmental Microbiology 68.12 (2002): 5917-5917.
- Wieczorek K., et al. “Prevalence, antimicrobial resistance and molecular characterization of Campylobacter jejuni and C. coli isolated from retail raw meat in Poland”. Journal of Veterinary Medicine Czech 57.6 (2012): 293-299.
- Workman NS., et al. “Pet dods and chicken meat as reservoir of Campylobacter spp. in Barbados”. Journal of Clinical Microbiology 43.6 (2005): 2642-2650.
Citation:
Abubakar SM Abba Maiha., et al. “Epidemiology of Thermophilic Campylobacter Species in Rural Poultry in Kebbi State,
Nigeria”. Multidisciplinary Advances in Veterinary Science 1.5 (2017): 219-226.
Copyright: © 2017 Abubakar SM Abba Maiha., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.