Research Article
Volume 2 Issue 1 - 2017
Effectiveness of the De-Escalation Regimen of Amtolmetin Guacyl in Patients with Active Ankylosing Spondylitis
1Department of therapy and rheumatology, North-Western State medical university named under I I Mechnikov, Saint Petersburg, Russia
2Saratov State Medical University named after V. I. Razumovsky. Saratov, Saratovskaya oblast', Russia, 410012
2Saratov State Medical University named after V. I. Razumovsky. Saratov, Saratovskaya oblast', Russia, 410012
*Corresponding Author: Prof. Andrey Petrovich Rebrov, Saratov State Medical University named after V. I. Razumovskyy, Saratov, Saratovskaya oblast, Russia.
Received: December 26, 2017; Published: December 30, 2017
Abstract
Background and purpose: Ankylosing spondylitis (AS) is a chronic inflammatory disease of axial skeleton, which commonly occurs in the second and third decade of life. Following study is conducted to evaluate efficacy of de-escalation regimen of amtolmetin guacyl (AMG) administration in active AS patients.
Patients and Methods: In a pilot open-label single-center study effectiveness of AMG was evaluated in 60 AS patients with total pain score ≥ 4.0 (Numeric Rating Scale (NRS)) and BASDAI ≥ 4.0. Patients received 1800 mg AMG per day for 2 weeks, switched to 600 mg per day for rest of the study period. Total study duration was 12 weeks. Ankylosing Spondylitis Disease Activity Score (ASDAS), Bath Ankylosing Spondylitis Activity Index (BASDAI), Bath Ankylosing Spondylitis Functional Index (BASFI) indexes, Low back pain (LBP), and C-reactive protein (CRP) level were measured at baseline, week 2, and week 12.
Results: The median of ASDAS decreased from 3.2 [2.8; 3.7] at baseline to 2.46 [2.1; 2.8] at week 2 and 1.79 [1.4; 2.3] at week 12 (p < 0.000 for all intergroup differences). The reduction in LBP intensity by ≥ 2.0 scores at week 2 was achieved in 85% patients. BASDAI, CRP levels decreased significantly at week 2 and week 12 (p < 0.000 for all intergroup differences). No major side effects were reported. Increase of arterial blood pressure was registered in 10% of patients at first 2 weeks of the treatment, requiring adjustment of antihypertensive therapy but not NSAID discontinuation. After dose de-escalation no changes of blood pressure were registered.
Interpretation: Use of AMG at a dose of 1800 mg/day substantially contributes to ameliorating back pain and disease activity in patients with AS. Subsequent administration of the drug at daily dose of 600 mg/day allows to maintain these results.
Keywords: Amtolmetin Guacyl; Ankylosing spondylitis; Non-steroidal anti-inflammatory drugs
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory disease of axial skeleton, which commonly occurs in the second and third decade of life. The global prevalence of AS is between 0.1% and 1.4% (Dean., et al. 2014).
Ankylosing spondylitis causes persistent inflammatory back pain, associated with structural and functional impairment, affecting the quality of life and socioeconomic abuse. Some patients suffer from extra-articular manifestations, which further add to the disease burden (Boonen and Linden 2006).
Non-steroidal anti-inflammatory drugs (NSAIDs) remain the cornerstone of treatment in patients with persistently active disease. Tumour necrosis factor blockers and Interleukin-17А inhibitors are preferred for patients, who are refractory to conventional treatment. However, insufficient control of symptoms, gastrointestinal, cardiovascular, and renal toxicities of current treatment drugs have restricted their use (Braun and Sieper 2007).
Majority of published data shows efficacy of NSAIDs in inflammation reduction in AS. Long courses of NSAIDs are preferred over short courses. It has been suggested, that continuous dosing of NSAIDS for long-term inhibits radiographic progression. However, data for preferred regimen of NSAIDs administration in long-term are limited and contravened. Few studies demonstrate preferred use of continuous NSAIDs intake over on-demand regimen (Kroon., et al. 2015) (Adams., et al. 2012). Sieper J., et al. had shown the equal effectiveness of continuous and on-demand regimens of 2 years diclofenac administration in AS (Sieper., et al. 2016). However, the majority of NSAIDs’ side effects are dose-dependent. It is interesting to explore the possibility of NSAIDs dose decrease in case of their long-term intake. It seems promising to achieve the remission of AS on high NSAIDs dosage and then maintain treatment results on lower and safer dosage of drug.
Amtolmetin is one such novel NSAID with good anti-inflammatory and analgesic properties with own gastroprotective activity. It was first approved for the treatment of painful disorders in Italy and India in 2008 and in Russia in 2014 (Garg., et al. 2016). Amtolmetin guacil (AMG) is a modification of tolmetine with increased gastrointestinal safety. The clinical efficacy and safety of tolmetin has been established earlier in AS. It has shown promising results with regard to relief of symptoms such as pain, stiffness, and restricted mobility with good tolerability. In several controlled studies, tolmetin had proven similar effectiveness as indomethacin, the mainstay of treatment in AS (Calin and Calif 1983). However, we could not find studies regarding AMG effectiveness in patients with AS.
The aim of the present study was to evaluate efficacy of de-escalation regimen of amtolmetin guacyl (AMG) in active ankylosing spondylitis (AS) patients.
Methods
Study population and Study Design
The present study was conducted in Russian Federation and registered with http://clinicaltrials.gov. number NCT02140073. Registration number in Unified State Information Accounting System: 115021010143 dated 10.02.2015.
The present study was conducted in Russian Federation and registered with http://clinicaltrials.gov. number NCT02140073. Registration number in Unified State Information Accounting System: 115021010143 dated 10.02.2015.
This was a pilot, open-label, single-center study to evaluate the effectiveness of de-escalation regimen of AMG treatment in AS patients. The study was conducted in the period between Jan-Oct 2015 in Russia. The study was performed in accordance with the Declaration of Helsinki and Good Clinical Practice (GCP). All patients provided signed informed consent prior to the start of the study in accordance with local regulations and standards of Bioethics.
Patients
Patients of age ≥ 18 years presenting with AS according to modified New-York criteria (Linden., et al. 1984) were included in the study. Other inclusion criteria were: if patients had arterial hypertension it has to be controlled by amlodipine monotherapy or its combination with other anti-hypertensive drugs; if patients were taking glucocorticoids, dose (prednisolone equivalent) did not exceed 10 mg per day and patients were stable for 2 weeks or more prior to screening; in case of methotrexate and sulfasalazine use: dose did not exceed 25 mg per week and 3 grams per day respectively and patients were stable for 4 weeks or more prior to screening. Lastly, in case of analgesics or antihypertensive drugs, dose was stable during 4 or more weeks prior to screening.
Patients of age ≥ 18 years presenting with AS according to modified New-York criteria (Linden., et al. 1984) were included in the study. Other inclusion criteria were: if patients had arterial hypertension it has to be controlled by amlodipine monotherapy or its combination with other anti-hypertensive drugs; if patients were taking glucocorticoids, dose (prednisolone equivalent) did not exceed 10 mg per day and patients were stable for 2 weeks or more prior to screening; in case of methotrexate and sulfasalazine use: dose did not exceed 25 mg per week and 3 grams per day respectively and patients were stable for 4 weeks or more prior to screening. Lastly, in case of analgesics or antihypertensive drugs, dose was stable during 4 or more weeks prior to screening.
Patients were excluded if they had traumatic or acute pain in the lower back (less than 3 month); if they had ulceration or gastrointestinal bleeding history; if they had ischemic heart disease, angina, myocardial infarction, stroke or transient disturbances of brain circulation, congestive heart failure (New York Heart Association [NYHA] class III - IV); if they had uncontrolled hypertension, manifested by an increase in systolic blood pressure ≥ 160 mm Hg and/or diastolic blood pressure ≥ 100 mm Hg during the screening and/or baseline visit or 3 month prior the screening; if they had inflammatory bowel disease (Crohn's disease, Ulcerative colitis) in the acute phase, hemophilia and other bleeding disorders, severe renal impairment (creatinine clearance less than 30 ml/min), progressive kidney diseases, or confirmed hyperkalemia.
Study Design and Treatment
All the patients received AMG orally in daily dose of 1800 mg on an empty stomach for 2 weeks, which was switched to 600 mg per day for rest of the study period.
All the patients received AMG orally in daily dose of 1800 mg on an empty stomach for 2 weeks, which was switched to 600 mg per day for rest of the study period.
Assessments
Study participants were evaluated at baseline and during the treatment visits at Week 2 and Week 12. The duration of study for each patient in this study was 12 weeks. Assessments of low back pain, nocturnal pain according to the numerical rating scales (NRS) were performed on all the treatment visits. To determine the AS activity, the AS Disease Activity Score (ASDASCRP) (Lukas C., et al. 2009), the Bath Ankylosing Spondylitis Activity Index (BASDAI) (Garrett S., et al. 1994) and to evaluate physical function status, the Bath Ankylosing Spondylitis Functional Index (BASFI) indices (Calin A., et al. 1994) were measured.
Study participants were evaluated at baseline and during the treatment visits at Week 2 and Week 12. The duration of study for each patient in this study was 12 weeks. Assessments of low back pain, nocturnal pain according to the numerical rating scales (NRS) were performed on all the treatment visits. To determine the AS activity, the AS Disease Activity Score (ASDASCRP) (Lukas C., et al. 2009), the Bath Ankylosing Spondylitis Activity Index (BASDAI) (Garrett S., et al. 1994) and to evaluate physical function status, the Bath Ankylosing Spondylitis Functional Index (BASFI) indices (Calin A., et al. 1994) were measured.
Other assessments were: Patient acceptable symptom state (PASS) (Maksymowych WP., et al. 2007) and parameters of vertebral mobility at Week 2 and Week 12 (Jenkinson TR., et al. 1994).
Study Endpoints
Primary Endpoint
Primary Endpoint
- Proportion of patients with reduction in low back pain intensity by ≥ 2.0 scores by numerical rating scale at Week 2
Secondary Endpoints
- Improvement of ASDAS, BASDAI, and BASFI at Week 2 and Week 12.
- Improvement of overall assessment of pain, nocturnal pain by numerical rating scale at Week 2 and Week 12.
- Number of patients achieved positive patient acceptable symptom state (PASS) at Week 2 and Week 12.
- Side effects were collected from baseline up to week 12 for safety reasons.
Statistics
Statistical analysis was performed applying Statistica SPSS17 and Statistica 8.0 software (StatSoft). To describe normally distributed quantitative attributes, mean value of the attribute and standard deviation (M ± SD) were used; to describe sampling distribution of attributes different from the normal one, median, upper and lower quartiles – Me [Q25;Q75] were used. To compare two groups with normal distribution of quantitative attribute, Student’s test was determined for independent groups. To compare significance of intergroup differences in quantitative values, Mann-Whitney test was used. The differences were deemed significant at p < 0.05.
Statistical analysis was performed applying Statistica SPSS17 and Statistica 8.0 software (StatSoft). To describe normally distributed quantitative attributes, mean value of the attribute and standard deviation (M ± SD) were used; to describe sampling distribution of attributes different from the normal one, median, upper and lower quartiles – Me [Q25;Q75] were used. To compare two groups with normal distribution of quantitative attribute, Student’s test was determined for independent groups. To compare significance of intergroup differences in quantitative values, Mann-Whitney test was used. The differences were deemed significant at p < 0.05.
Results
Sixty AS patients with the mean age of 37.58 ± 11.7 years were enrolled in the study. All patients completed the study. The patients’ characteristics are presented in Table 1.
| Parameter | All Patients (N = 60) |
| Average age, years | 37.58 ± 11.7 |
| Males, n (%) | 39 (65) |
| Disease duration, years | 10.2 ± 9.65 |
| Systemic COronary Risk Evaluation (SCORE) index | 0.31 [0.04; 0.90] |
| Smokers (%) | 22 (36.7) |
| Systolic BP, baseline, mm Hg | 129.8 ± 15.2 |
| Diastolic BP, baseline, mm Hg | 77±7.2 |
| Total cholesterol, mmol/L | 5.33 ± 1.18 |
| Body mass index (BMI), kg/m2 | 23.4 [20.07; 28.4] |
Table 1: Baseline Characteristics of Patients Enrolled in the Study with Ankylosing Spondylitis (N = 60).
| Baseline | Week 2 | Week 12 | P value Baseline/Week 2 | P value Baseline/ Week 12 |
|
| Low back pain, NRS | 5.5 [4.7;7.0] | 5.0 [4.0;7.0] | 4.0 [3.0;6.0] | 0.7 | <0.000 |
| Nocturnal low back pain, NRS | 5.0 [4.0;7.0] | 5.0 [4.0;6.0] | 3.0 [0.75;5.0] | 0.02 | <0.000 |
| BASDAI | 5.2 [4.6;6.3] | 3.6 [3.1;4.4] | 2.3 [1.22;3.1] | 0.000 | 0.000 |
| ASDAS | 3.2 [2.8;3.7] | 2.46 [2.1;2.8] | 1.79 [1.4;2.3] | 0.000 | 0.000 |
| BASFI | 4.25 [2.6;6.9] | 3.95 [2.4;5.3] | 2.95[1.4;4.2] | 0.000 | 0.000 |
| Lateral lumbar vertebral mobility, cm | 1.75 [0.3;7.5] | 2.25 [0.4;7.5] | 2.25 [0.4;7.5] | >0.05 | >0.05 |
| CRP, mg/L | 11.8 [5.23; 22.7] | 8.65 [4.4; 16.25] | 6.25 [2.87; 9.45] | 0.000 | 0.002 |
| ESR, mm/h | 13.5 [6.25; 23.0] | 9.5 [5.0; 17.0] | 10.0 [5.0; 15.0] | 0.026 | 0.000 |
NRS: Numerical Rating Scale
BASDAI: Bath Ankylosing Spondylitis Disease Activity Index
ASDAS: Ankylosing Spondylitis Disease Activity Score
BASFI: Bath Ankylosing Spondylitis Functional Index
CRP: C - reactive protein
ESR: Erythrocyte sedimentation rate
Table 2: Mean Low Back Pain, Nocturnal Pain, Ankylosing Spondylitis Activity and Functional Indexes, Vertebral Mobility in Ankylosing Spondylitis Patients Receiving Amtolmetin Guacyl.
BASDAI: Bath Ankylosing Spondylitis Disease Activity Index
ASDAS: Ankylosing Spondylitis Disease Activity Score
BASFI: Bath Ankylosing Spondylitis Functional Index
CRP: C - reactive protein
ESR: Erythrocyte sedimentation rate
Table 2: Mean Low Back Pain, Nocturnal Pain, Ankylosing Spondylitis Activity and Functional Indexes, Vertebral Mobility in Ankylosing Spondylitis Patients Receiving Amtolmetin Guacyl.
Efficacy Analysis
Primary Efficacy Analysis
The reduction in low back pain intensity by ≥ 2.0 scores by numerical rating scale at week 2 was achieved in 51/60(85%) patients.
Primary Efficacy Analysis
The reduction in low back pain intensity by ≥ 2.0 scores by numerical rating scale at week 2 was achieved in 51/60(85%) patients.
Secondary Efficacy Analysis
Mean back pain parameters, BASDAI, ASDAS, and BASFI reduced at week2 (during administration of maximal drug dose) regardless of hypertension presence. The decrease of AS activity continued after dose reduction to 600 mg daily, reaching minimal values at week 12. At baseline, the median of low back pain was 5.5 [IQR: 4.7; 7.0], which was reduced to 5.0 [IQR: 4.0; 7.0] at week 2 and 4.0 [IQR: 3.0; 6.0] at week 12 respectively. The observed decrease was statistically significant at week 12 (P < 0.000) (Figure 1). Similarly, the nocturnal low back pain was reduced from 5.0 [IQR: 4.0; 7.0] at baseline to 5.0 [IQR: 4.0; 6.0] at week 2 and 3.0 [IQR: 0.75; 5.0] at week 12. The observed difference was statistically significant (P = 0.02 at week 2 and P <0.000 at week 12) (Figure 2). Patient Acceptable Symptom State (PASS) was achieved in 19 patients at week 2 and 26 patients at week 12.
Mean back pain parameters, BASDAI, ASDAS, and BASFI reduced at week2 (during administration of maximal drug dose) regardless of hypertension presence. The decrease of AS activity continued after dose reduction to 600 mg daily, reaching minimal values at week 12. At baseline, the median of low back pain was 5.5 [IQR: 4.7; 7.0], which was reduced to 5.0 [IQR: 4.0; 7.0] at week 2 and 4.0 [IQR: 3.0; 6.0] at week 12 respectively. The observed decrease was statistically significant at week 12 (P < 0.000) (Figure 1). Similarly, the nocturnal low back pain was reduced from 5.0 [IQR: 4.0; 7.0] at baseline to 5.0 [IQR: 4.0; 6.0] at week 2 and 3.0 [IQR: 0.75; 5.0] at week 12. The observed difference was statistically significant (P = 0.02 at week 2 and P <0.000 at week 12) (Figure 2). Patient Acceptable Symptom State (PASS) was achieved in 19 patients at week 2 and 26 patients at week 12.
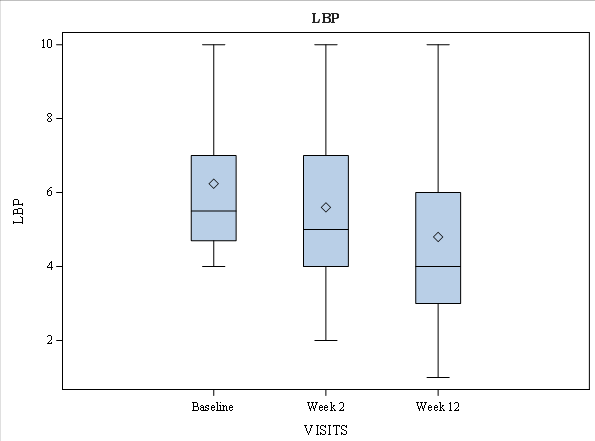
Figure 1: Low Back Pain According to Numeric Rating Scale over Time in Ankylosing Spondylitis Patients during Amtolmetin Guacyl Therapy.
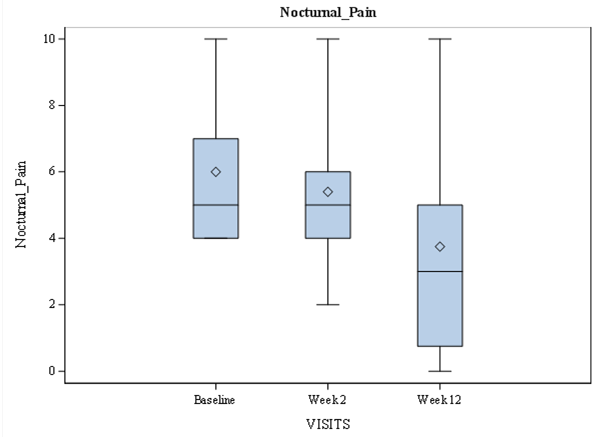
Figure 2: Nocturnal Low Back Pain According to Numeric Rating Scale over Time in Ankylosing Spondylitis Patients During Amtolmetin Guacyl Therapy.
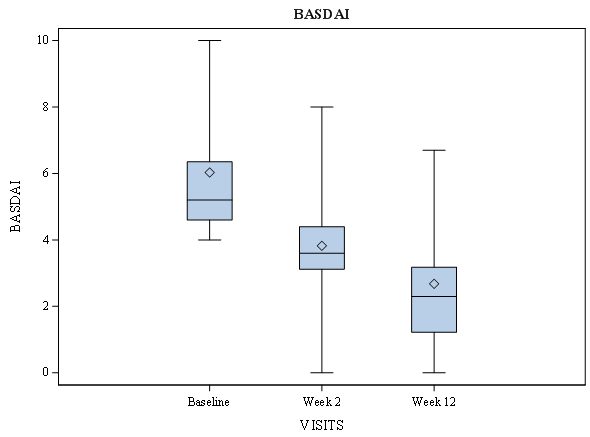
Figure 3: Bath Ankylosing Spondylitis Disease Activity Index (Basdai) Score According to Visual Analogue Scale over Time in Ankylosing Spondylitis Patients During Amtolmetin Guacyl Therapy.
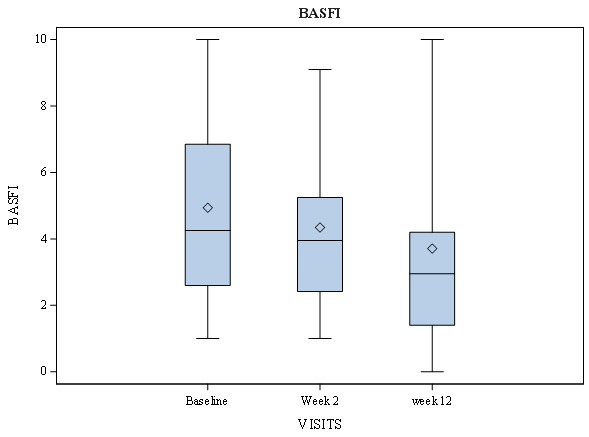
Figure 4: Bath Ankylosing Spondylitis Disease Functional Index (Basfi) Score According to Visual Analogue Scale over Time in Ankylosing Spondylitis Patients During Amtolmetin Guacyl Therapy.
Safety Analysis
No statistically significant changes in the laboratory parameters or major side effects were reported on the use of AMG during the study. Six (10%) patients showed small increase in blood pressure in the first two weeks of high-dose therapy, requiring adjustment of antihypertensive therapy but not NSAID discontinuation. The increased BP was observed in AS patients with high CV risk-older patients, smokers, patients with family history of arterial hypertension and coronary heart disease. After week 2, none of the patients demonstrated BP changes.
No statistically significant changes in the laboratory parameters or major side effects were reported on the use of AMG during the study. Six (10%) patients showed small increase in blood pressure in the first two weeks of high-dose therapy, requiring adjustment of antihypertensive therapy but not NSAID discontinuation. The increased BP was observed in AS patients with high CV risk-older patients, smokers, patients with family history of arterial hypertension and coronary heart disease. After week 2, none of the patients demonstrated BP changes.
Discussion
We conducted a pilot study in Russia to evaluate the effectiveness of de-escalation regimen of AMG in AS patients. It was shown that AMG at maximum daily dose reduces low back pain and activity parameters of ankylosing spondylitis. Reducing the maximal daily dose of AMG to the minimal maintaining dose is associated with retained analgesic and anti-inflammatory effect achieved in patients with AS. This may be associated with potentially lower risk of complications.
Non-steroidal anti-inflammatory drugs have been the standard treatment of AS for decades. In earlier open label trials, 32/34 patients showed excellent therapeutic efficacy and safety by the use of tolmetin (molecular precursor of AMG) over a period of 12 weeks. There was a statistically significant improvement in pain alleviation and limitation of motion. Further, the results of open label studies were confirmed by the double blind studies of tolmetin vs. placebo. For example, in a crossover study with 30 patients treated for four weeks, there was a significantly greater improvement in therapeutic response, patient assessment of spinal and nocturnal pain, duration of morning stiffness with tolmetin as compared to placebo (Calin and Calif 1983).
In six-weeks comparative studies with indomethacin, tolmetin had similar effectiveness to that of indomethacin in pain alleviation and providing increased mobility. A few other studies comparing tolmetin with indomethacin had the similar results (Calin and Calif 1983). In another 4 weeks single blind crossover study, it was found that tolmetin was superior to ibuprofen in AS patients (64% responders with tolmetin vs. 39% responders with indomethacin) (Calin and Calif 1983).
A recent study in knee osteoarthritis patients showed similar clinical benefit of AMG versus diclofenac; however, the probability of emerging gastrointestinal risk in the AMG group was comparatively less than that in diclofenac group (Povoroznyuk., et al. unpublished).
In our study, the possibility of AMG use in AS and its efficacy not only in maximal daily dosage, but its capacity to maintain the treatment result after dose decrease was shown. As we know, it is the first experience of de-escalation regimen of NSAIDs administration. Accordingly, we performed dose de-escalation at the fixed time-point (Week 2). At the same time, it seems that de-escalation of NSAIDs dose after reaching of low disease activity or remission will be more effective and has to be evaluated in the further studies. Maintenance of remission on lower NSAIDs dose, administered after the maximal dosage was the vital result of the present study. Our finding that all patients with destabilized hypertension were smokers is concordant with published literature (Bich., et al. 2010).
The possibility of improvement of NSAIDs safety profile was also shown. It is well-known, that NSAIDs are associated with GI, cardiovascular and renal toxicity, which limits their use. Amtolmetin is known to have the gastric sparing effect. This is mediated by the presence of vanillic moiety in the AMG, which stimulates capsaicin receptors and releases calcitonin gene related peptide (CGRP) and consequently increases nitric oxide (NO), hence counterbalancing the deleterious effects of prostaglandin depletion due to cyclooxygenase inhibition and providing mucosal protection (Pisano., et al. 1999). The cardiovascular safety of AMG is comparable to other non-selective COX inhibitors. In our study, administration of high doses of AMG for 2 weeks in AS patients did not reveal cardiovascular events (myocardial infarctions or sudden cardiac death, aggravation of circulatory failure). However, approximately 10% of patients showed increase in blood pressure in the first two weeks of high-dose therapy, requiring adjustment of antihypertensive therapy but not NSAID discontinuation. After dose de-escalation, no changes of blood pressure were registered. The achieved results could be explained with dose-dependence of NSAIDs side effects. The results of the present study demonstrate the possibility of NSAIDs safety improvement in case of NSAIDs dosage decrease. It seems promising in long-term perspective and has to be investigated in the future larger and longer studies with detailed safety analysis. The limitation of our study is that we could only evaluate clinical and laboratory efficacy and safety of the AMG regimen with the short term study period of 12 weeks without control. Long term controlled observations with larger population are required to study the effect of such dosing regimens in clinical, laboratory aspects of AS activity and structural progression.
Conclusion
Our study results suggest the effectiveness of AMG in reducing pain and other disease activity parameters of AS. Switching from high dose to low dose post 2 weeks was able to maintain this effectiveness. The de-escalation of AMG dosage is associated with stabilization of blood pressure in patients with destabilisation of the hypertension on high drug dosages. Efficacy and safety of prolonged administration of amtolmetin guacil in AS requires further studies. More studies are required to evaluate AMG potency to change the process of bone remodelling in AS.
References
- Adams K., et al. “Safety and efficacy of on-demand versus continuous use of nonsteroidal antiinflammatory drugs in patients with inflammatory arthritis: a systematic literature review”. The Journal of Rheumatology. Supplement 90 (2012): 56-58.
- Au Bich T., et al. “The association between smoking and hypertension in a population-based sample of Vietnamese men”. Journal of Hypertension 28.2 (2010): 245-250.
- Boonen A and Vander Linden SM. “The burden of ankylosing spondylitis”. The Journal of Rheumatology Supplement 78 (2006): 4-11
- Braun J., et al. “Ankylosing spondylitis”. Lancet 61.2 (2007): 1379-1390.
- Calin A and Calif S. “Clinical use of tolmetin sodium in patients with ankylosing spondylitis: A Review”. The Journal of Clinical Pharmacology 23.7 (1983): 301-308.
- Calin A., et al. “A new approach to defining functional ability in ankylosing spondylitis: the development of the Bath Ankylosing Spondylitis Functional Index”. The Journal of Rheumatology 21.12 (1994): 2281-2285.
- Dean LE., et al. “Global prevalence of ankylosing spondylitis”. Rheumatology 53.4 (2014): 650-657.
- Garg A., et al. “Amtolmetin: A Reappraisal of NSAID with Gastroprotection”. Arthritis (2016): 7103705.
- Garrett S., et al. “A new approach to defining disease status in ankylosing spondylitis: the Bath Ankylosing Spondylitis Disease Activity Index”. The Journal of Rheumatology 21.12 (1994): 2286-2291.
- Jenkinson TR., et al. “Defining spinal mobility in ankylosing spondylitis (AS). The Bath AS Metrology Index”. The Journal of Rheumatology 21.9 (1994): 1694-1698.
- Kroon FPB., et al. “Non-steroidal anti-inflammatory drugs (NSAIDs) for axial spondyloarthritis (ankylosing spondylitis and non-radiographic axial spondyloarthritis) (Protocol)”. Cochrane Database of Systematic Reviews 17.7 (2015): CD010952.
- Kroon FPB., et al. “Non-steroidal anti-inflammatory drugs (NSAIDs) for axial spondyloarthritis (ankylosing spondylitis and non-radiographic axial spondyloarthritis)”. Cochrane Database System Reviews 17.7 (2015): CD010952.
- Linden SV., et al. “Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis”. Arthritis and Rheumatism 27.4 (1984): 361-368.
- Lukas C., et al. “Assessment of SpondyloArthritis international Society. Development of an ASAS-endorsed disease activity score (ASDAS) in patients with ankylosing spondylitis”. Annals of the Rheumatic Diseases 68.1 (2009): 18-24.
- Maksymowych WP., et al. “Evaluation and validation of the patient acceptable symptom state (PASS) in patients with ankylosing spondylitis”. Arthritis & Rheumatology 57.1 (2007):133-139.
- Pisano C., et al. “Gastrosparing effect of new antiinflammatory drug amtolmetin guacil in the rat: involvement of nitric oxide”. Digestive Diseases and Sciences 44.4 (1999): 713-724.
- Povoroznyuk VV., et al. “A comparative study of amtolmetin guacil and diclofenac sodium in patients with knee osteoarthritis”. 25.1 (2017): S421.
- Sieper J., et al. “Effect of continuous versus on-demand treatment of ankylosing spondylitis with diclofenac over 2 years on radiographic progression of the spine: results from a randomised multicentre trial (ENRADAS)”. Annals of the Rheumatic Diseases 75.8 (2016): 1438-1443.
Citation:
Andrey Petrovich Rebrov., et al. “Effectiveness of the De-Escalation Regimen of Amtolmetin Guacyl in Patients with Active
Ankylosing Spondylitis”. Orthopaedic Surgery and Traumatology 2.1 (2017): 253-261.
Copyright: © 2017 Andrey Petrovich Rebrov., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.