Research Article
Volume 2 Issue 2 - 2018
Biodegradation of Paracetamol by Native Bacterial Strains Isolated from Yemeni Pharmaceutical Wastewater Plant in Sana’a
1Biology Department, Faculty of Science, Sana'a University, Yemen
2Medicinal Chemistry Department, Faculty of Pharmacy, Sana'a University, Yemen
3Chemistry Department, Faculty of Science, Sana'a University, Yemen
2Medicinal Chemistry Department, Faculty of Pharmacy, Sana'a University, Yemen
3Chemistry Department, Faculty of Science, Sana'a University, Yemen
*Corresponding Author: Wadhah H Edrees, Biology Department, Faculty of Science, Sana'a University, Yemen.
Received: March 27, 2018; Published: April 17, 2018
Abstract
Paracetamol has emerged as an important environment pollutant due to its extensive usage. This study amid to isolate, identify, and characterize of a native Paracetamol-degrading bacteria from the contaminated site of pharmaceutical wastewater at Sana'a City, Yemen. The bacteria were isolated and purified from wastewater samples by using the enrichment media and selective medium. The isolated bacteria were identified according to phenotypic and genotypic characterizations. The results showed that the two bacterial strains were isolated from a contaminated site. These strains were signed as an STB2 and STB4 and identified as Pseudomonas aeruginosa strain ATCC 10145 with similarity 88% and 97%, respectively. Also, the environmental factors, carbon, and energy sources were observed to influence the biodegradation rate of Paracetamol. However, it was found that the 79.4% and 88.4% of 3000 mg/L Paracetamol were degraded by strain STB2 and STB4, respectively, in 120 h. The results performed that the degradation potential of the two isolated bacterial strains possibly played a major active role in the Paracetamol removal processes.
Keywords: Bacteria; Biodegradation; Isolation; Paracetamol; Physiological Characterization
Introduction
Paracetamol or acetaminophen is a common analgesic and an anti-inflammatory that widely used as a non-prescription drug [1]. In the last decades, Paracetamol consumption has increased throughout the world. In the USA, it ranked as one of the top 200 prescriptions [2]. In Kuwait also it was consumed as the second prescription drugs during the year 2008 [3]. In Yemen, it is ranked as first of top ten drugs produced by local industrial and one of the top ten drugs imported [4].
Paracetamol is one of the most frequently detected pharmaceutical products in the aquatic environment. It was found with concentration of 0.298 μg/L in drinking water [5], 6.5 μg/L in groundwater [6], 15.7 µg/L in surface water [7], and 1368.47 μg/L in wastewater [8]. The frequent occurrence of this substance in the aquatic environments has raised a concern about their potential effects on the environment and human health [9].
Previous studies on removal of Paracetamol from wastewater mainly focused on chemical methods which are not a desirable choice for removing this substance due to the use of excessive chemicals and energy sources [10,11]. Therefore, the biodegradation of Paracetamol substance is being considered as an environmentally friendly and low-cost option [12].
This study describes the isolation, identification, and characterization of a native Paracetamol degrading bacteria that isolated from wastewater contaminated site of a Yemen Drug Company for Industrial and Commercial (YEDCO) at Sana'a City, Yemen. This wastewater was generated by the company containing a high concentration of Paracetamol through the production processes. The isolated bacterial strains were applied further study for physiological characterizations.
Materials and Methods
Chemicals and cultivation medium
Paracetamol ultrafine powder was obtained from Anqiu Lu'an Pharmaceutical Co. Ltd. (China) and all other chemicals were of the highest purity commercially available. The applied mineral salts medium (MSM) contained the following composition: per liter of distilled water, 0.5g of K2HPO4, 0.5g of K2HPO4, 0.01g of NaCl, 0.2g of MgCl2·6H2O, 0.02g of CaCl2, 0.339mg of MnSO4, 0.428mg of ZnSO4, 0.347mg of (NH4)6Mo7O24·4H2O, 0.4mg of CoCl2·6H2O, 10mg of EDTA. After adjusting the pH to 7.2–7.4 with 8M NaOH, the solution was autoclaved at 121˚C for 15 min. Paracetamol substance was added as the sole energy and carbon sources in different concentration after sterilized by filtration paper (0.22µm) [2].
Paracetamol ultrafine powder was obtained from Anqiu Lu'an Pharmaceutical Co. Ltd. (China) and all other chemicals were of the highest purity commercially available. The applied mineral salts medium (MSM) contained the following composition: per liter of distilled water, 0.5g of K2HPO4, 0.5g of K2HPO4, 0.01g of NaCl, 0.2g of MgCl2·6H2O, 0.02g of CaCl2, 0.339mg of MnSO4, 0.428mg of ZnSO4, 0.347mg of (NH4)6Mo7O24·4H2O, 0.4mg of CoCl2·6H2O, 10mg of EDTA. After adjusting the pH to 7.2–7.4 with 8M NaOH, the solution was autoclaved at 121˚C for 15 min. Paracetamol substance was added as the sole energy and carbon sources in different concentration after sterilized by filtration paper (0.22µm) [2].
Isolation and purification of bacteria and culture conditions
Different samples were collected from wastewater generated by YEDCO situated in Sana'a City, Yemen. One mL of each sample was transferred to bottle contained 90 mL of trypticase soy broth (TSB) and incubated at 30˚C for 3 days. Ten mL of incubated TSB media was inoculated into a bottle containing 90 mL of MSM with different concentration of paracetamol (250–2500 mg/L) and incubated at 30˚C for 5 days, as described by Ahmed., et al. [13] and Gusseme., et al. [14].
Different samples were collected from wastewater generated by YEDCO situated in Sana'a City, Yemen. One mL of each sample was transferred to bottle contained 90 mL of trypticase soy broth (TSB) and incubated at 30˚C for 3 days. Ten mL of incubated TSB media was inoculated into a bottle containing 90 mL of MSM with different concentration of paracetamol (250–2500 mg/L) and incubated at 30˚C for 5 days, as described by Ahmed., et al. [13] and Gusseme., et al. [14].
Subsequently, 1 mL of MSM was transferred to MSM agar containing 250 mg/L of Paracetamol and incubated several days at 30˚C. Plate colonies showing growth were purified on trypticase soy (TSA) and after several transfers on MSM agar containing Paracetamol and checked again on TSA to be pure. Pure colonies isolated were tested for growth on higher concentrations of Paracetamol (250–4000 mg/L) in MSM agar. The isolated strain showing best growth of the compounds under study was selected and characterized further [13].
Identification of isolated bacteria
The isolated bacteria were identified according to its phenotypic and genotypic characteristics. Preliminary characterization was based on morphological, physiological and biochemical tests[15].Further identification was completed by 16S rDNA sequencing.Genomic DNAs of the harvested cells were extracted using a DNeasy® Blood & Tissue kit (QIAGEN, Germany), and the 16S rRNA was amplified by means of PCR (Eppendrof, Germany) using the bacteria universal primer 27F (5′-AGAGTTTGATCMTGGCTCAG-3′ and 1492R (I) (5′-GGTTACCTTGTTACGACTT-3). PCR mixture contained 5 µL of 5X Q5 reaction buffer, 0.2 µL of 10 mM dNTPs, 100 pM of forward primer and 100 pM of reverse primer, 5 ng/uL of DNA template, 0.01 µL/µL of Q5 high-fidelity DNA polymerase (NEB, M041S) and the volume completed up to 25 µL by nuclease-free water [14].
The isolated bacteria were identified according to its phenotypic and genotypic characteristics. Preliminary characterization was based on morphological, physiological and biochemical tests[15].Further identification was completed by 16S rDNA sequencing.Genomic DNAs of the harvested cells were extracted using a DNeasy® Blood & Tissue kit (QIAGEN, Germany), and the 16S rRNA was amplified by means of PCR (Eppendrof, Germany) using the bacteria universal primer 27F (5′-AGAGTTTGATCMTGGCTCAG-3′ and 1492R (I) (5′-GGTTACCTTGTTACGACTT-3). PCR mixture contained 5 µL of 5X Q5 reaction buffer, 0.2 µL of 10 mM dNTPs, 100 pM of forward primer and 100 pM of reverse primer, 5 ng/uL of DNA template, 0.01 µL/µL of Q5 high-fidelity DNA polymerase (NEB, M041S) and the volume completed up to 25 µL by nuclease-free water [14].
Amplification was started with an initial denaturation at 98˚C for 5 min, followed by 35 cycles at 98˚C for 20 s, 58˚C for 25 s, and 72˚C for 30 s/kb, followed by 8 min at 72˚C for a final extension. The resulting 16s rRNA gene sequences were compared to database NCBI using BLAST program (http://www.ncbi.nlm.nih.gov). The multiple alignments of the sequence were performed, and a neighbor-joining phylogenetic tree was constructed using the latest version (2.6.1) of the BLAST program.
Physiological characterization
The microorganism’s physiological characterization play an important role on Paracetamol degradation rate. Though, the optimization of the degrading bacteria to the tested Paracetamol was necessary to enable the isolated strains to utilize it effectively as a carbon and energy source [13]. The physiological characterizations include the following:-
The microorganism’s physiological characterization play an important role on Paracetamol degradation rate. Though, the optimization of the degrading bacteria to the tested Paracetamol was necessary to enable the isolated strains to utilize it effectively as a carbon and energy source [13]. The physiological characterizations include the following:-
a)Effect of temperature and pH on Paracetamol biodegradation
The effect of the range and optimum of temperature and pH on the biodegradation rate of Paracetamol (1500 mg/L) was determined within 72h. For the temperature effect, each bacterium strain was cultivated independently in a volume of 100 mL of MSM media containing Paracetamol and incubated at different levels of temperature (15, 20, 25, 30, 35, 40 and 45˚C) for 72h. However, in the pH effect study, the bacteria strains were cultivated individually in a volume of 100 mL of MSM media containing Paracetamol and having different initial pH values (4, 5, 6, 6.5, 7, 7.5, 8, 9, and 10) and incubated at 30˚C for 72h. For rate degradation, 5 mL of each culture medium was withdrawn and centrifuged separately at 8000 rpm 30 min. The supernatant was collected in separate clean test tubes and analyzed for residual Paracetamol content [13,17].
The effect of the range and optimum of temperature and pH on the biodegradation rate of Paracetamol (1500 mg/L) was determined within 72h. For the temperature effect, each bacterium strain was cultivated independently in a volume of 100 mL of MSM media containing Paracetamol and incubated at different levels of temperature (15, 20, 25, 30, 35, 40 and 45˚C) for 72h. However, in the pH effect study, the bacteria strains were cultivated individually in a volume of 100 mL of MSM media containing Paracetamol and having different initial pH values (4, 5, 6, 6.5, 7, 7.5, 8, 9, and 10) and incubated at 30˚C for 72h. For rate degradation, 5 mL of each culture medium was withdrawn and centrifuged separately at 8000 rpm 30 min. The supernatant was collected in separate clean test tubes and analyzed for residual Paracetamol content [13,17].
b)Effect of contact time on Paracetamol biodegradation
The effect of contact time on Paracetamol biodegradation was performed on MSM media for each bacterium strain. Each bacterium isolated was cultured individually on MSM media containing a different concentration of Paracetamol (1000‒4000 mg/L) and incubated in different contact time with a label (24, 48, 72, 96, and 120h) at 30˚C. After each time designated, the sample was analyzed for residual Paracetamol [18].
The effect of contact time on Paracetamol biodegradation was performed on MSM media for each bacterium strain. Each bacterium isolated was cultured individually on MSM media containing a different concentration of Paracetamol (1000‒4000 mg/L) and incubated in different contact time with a label (24, 48, 72, 96, and 120h) at 30˚C. After each time designated, the sample was analyzed for residual Paracetamol [18].
c) Effect of glucose concentration on Paracetamol biodegradation
The effect of glucose on Paracetamol biodegradation, as a source of energy and carbon for bacteria growth, was evaluated with different concentration of glucose (0, 1, 3, and 5 mg/L). Each concentration of glucose was added separately to MSM media (100 mL) containing-paracetamol (2500 mg/L) inoculated by each bacterium strain and incubated at 30˚C for 120h. sampling was done at regular time intervals for residual Paracetamol measuring [19].
The effect of glucose on Paracetamol biodegradation, as a source of energy and carbon for bacteria growth, was evaluated with different concentration of glucose (0, 1, 3, and 5 mg/L). Each concentration of glucose was added separately to MSM media (100 mL) containing-paracetamol (2500 mg/L) inoculated by each bacterium strain and incubated at 30˚C for 120h. sampling was done at regular time intervals for residual Paracetamol measuring [19].
D)Effect of bacteria concentration on Paracetamol biodegradation
The effect of bacteria concentration on paracetamol degradation was studied by cultivated each bacterial strain individually in paracetamol (2000 mg/L) - containing MSM medium with different initial bacterial concentration (1.0 × 104, 1.0 × 106, and 1.0 × 108 CFU/mL) and incubated at 30˚C for 72h. The residual Paracetamol content was analyzed for each experimental individually [18].
The effect of bacteria concentration on paracetamol degradation was studied by cultivated each bacterial strain individually in paracetamol (2000 mg/L) - containing MSM medium with different initial bacterial concentration (1.0 × 104, 1.0 × 106, and 1.0 × 108 CFU/mL) and incubated at 30˚C for 72h. The residual Paracetamol content was analyzed for each experimental individually [18].
Chemical analysis methods
300 mg of working standard of Paracetamol was weighed and dissolved in 100 mL of methanol in a 100 mL volumetric flask. 5 mL of solution was transferred to a 100 mL volumetric flask containing 47 mL of methanol and 53 mL of purified water and mixed well.Then, 10 mL of this solution was filtered by 0.22 μm nylon membrane filter before used [20].
300 mg of working standard of Paracetamol was weighed and dissolved in 100 mL of methanol in a 100 mL volumetric flask. 5 mL of solution was transferred to a 100 mL volumetric flask containing 47 mL of methanol and 53 mL of purified water and mixed well.Then, 10 mL of this solution was filtered by 0.22 μm nylon membrane filter before used [20].
Sample analyzed
The residual concentration of Paracetamol was determined by using HPLC (PerkinElmer, USA). The mobile phase with a flow rate of 1.0 mL/min was composed of acetonitrile: water (47: 53 v/v). The separation was performed at 30˚C by RP–8 column with dimension (5 μm, 4.6 × 250 mm). The injection volume was 20 μm, retention time 5 min, and detection wavelength was fixed at 275 nm [20].
The residual concentration of Paracetamol was determined by using HPLC (PerkinElmer, USA). The mobile phase with a flow rate of 1.0 mL/min was composed of acetonitrile: water (47: 53 v/v). The separation was performed at 30˚C by RP–8 column with dimension (5 μm, 4.6 × 250 mm). The injection volume was 20 μm, retention time 5 min, and detection wavelength was fixed at 275 nm [20].
Statistical analysis
The obtained data were subjected to statistical analysis using IBM SPSS Statistics software (version 18.0, 2009). The statistical analysis of variance between difference isolated microorganisms and biodegradation efficiency were compared by Kruskal-Wallis test. Also, the analysis of variance between biodegradation efficiency and physiological characterizations (glucose and microbial concentration) were compared by Friedman test. However, the correlation coefficient between degradation and physiological characterizations (pH and temperature) were performed by using Pearson test. Values (P < 0.01) was considered statistically significant.
The obtained data were subjected to statistical analysis using IBM SPSS Statistics software (version 18.0, 2009). The statistical analysis of variance between difference isolated microorganisms and biodegradation efficiency were compared by Kruskal-Wallis test. Also, the analysis of variance between biodegradation efficiency and physiological characterizations (glucose and microbial concentration) were compared by Friedman test. However, the correlation coefficient between degradation and physiological characterizations (pH and temperature) were performed by using Pearson test. Values (P < 0.01) was considered statistically significant.
Results
Two bacterial strains that capable of degrading Paracetamol in different concentration were isolated from the wastewater samples and assigned an STB2 and STB4 and identified by using 16s rRNA sequencing. These bacteria were gram-negative, slimy colonies, and non-spore forming. All strains were motile rods and positive for the catalase, oxidase, and nitrate reduction test. While, the citrate utilization, Vogues Proskauer, in dole production, H2S production, urease, DNase, and methyl-red test were negative.
The results of 16S rRNA sequence alignment and phylogenetic tree analysis revealed that the 16S rRNA sequence of strains of STB2 and STB4 belonged to the genus P. aeruginosa. Also, the taxonomic characteristics of isolated bacterial strain were mostly the same as those of P. aeruginosa.
However, a 1324 base per (bp) 16S rRNA gene sequence that obtained from strain STB2 and analyzed shows 88% similarity to the 16S rRNA gene sequence of P. aeruginosa strain ATCC 10145 as shown in Figure [1]. A 1306 bp 16S rRNA gene sequence was obtained from strain STB4 and analyzed. The 16S rRNA gene sequence of strain STB4 shows 97% similarity to the 16S rRNA gene sequence of P. aeruginosa strain ATCC 10145 as shown in Figure [2].
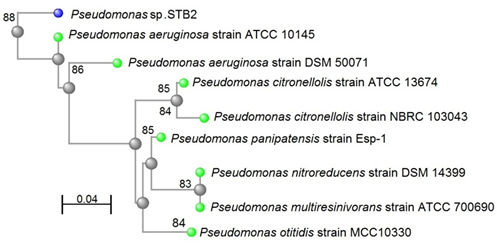
Figure 1: Neighbor-joining phylogenetic tree derived from 16S rRNA
gene sequence data showing the positions of Pseudomonas sp. STB2
and related organisms. Bar, 0.04 changes per nucleotide position.
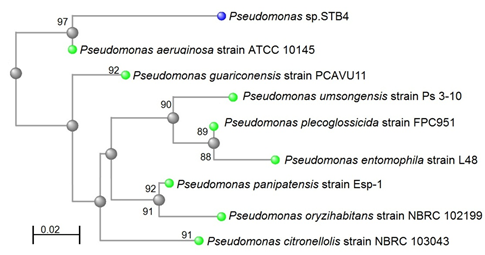
Figure 2: Neighbor-joining phylogenetic tree derived from 16S rRNA
gene sequence data showing the positions of Pseudomonas sp. STB4
and related organisms. Bar, 0.02 changes per nucleotide position.
Optimum conditions for isolated bacteria results
Effect of temperature and pH results
Table [1] shows the effect of temperature on the biodegradation rate of Paracetamol (1500 mg/L) by STB2 and STB4 strains within 72 h of incubation. Also, the decreasing and increasing effect on Paracetamol degradation were recorded at temperatures greater than 40˚C or smaller than 20˚C for both strains, STB2 and STB4.
Effect of temperature and pH results
Table [1] shows the effect of temperature on the biodegradation rate of Paracetamol (1500 mg/L) by STB2 and STB4 strains within 72 h of incubation. Also, the decreasing and increasing effect on Paracetamol degradation were recorded at temperatures greater than 40˚C or smaller than 20˚C for both strains, STB2 and STB4.
| Temperature (˚C) | The Paracetamol reduction (%) after 72h | P value | |
| STB2 | STB4 | ||
| 15 | 24 | 29 | < 0.01* |
| 20 | 44.11 | 51.19 | |
| 25 | 76 | 78.12 | |
| 30 | 97.73 | 100 | |
| 35 | 80 | 83 | |
| 40 | 44 | 53 | |
| 45 | 24.68 | 28 | |
Table 1: The effect of different temperatures on Paracetamol
Biodegradation by isolated bacterial strains.
*. Correlation is significant at the 0.01 level (2-tailed).
*. Correlation is significant at the 0.01 level (2-tailed).
However, the effect of pH on the biodegradation rate of Paracetamol (1500 mg/L) by STB2 and STB4 strains within 72 h are listed in Table [2].
| pH | The Paracetamol reduction (%) after 72h | P value |
|
| STB2 | STB4 | ||
| 4 | 18 | 21 | <0.01* |
| 5 | 40 | 44 | |
| 6 | 60 | 64 | |
| 6.5 | 93.89 | 97.4 | |
| 7 | 97.73 | 100 | |
| 7.5 | 92 | 96 | |
| 8 | 83 | 87 | |
| 9 | 34 | 40 | |
| 10 | 11 | 16 | |
Table 2: The effect of different levels of pH on Paracetamol biodegradation by isolated bacterial strains.
*. Correlation is significant at the 0.01 level (2-tailed).
*. Correlation is significant at the 0.01 level (2-tailed).
Effect of contact time results
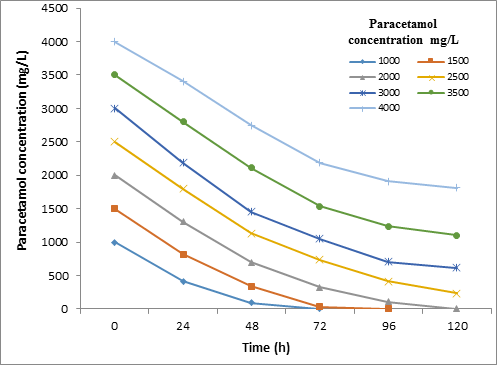
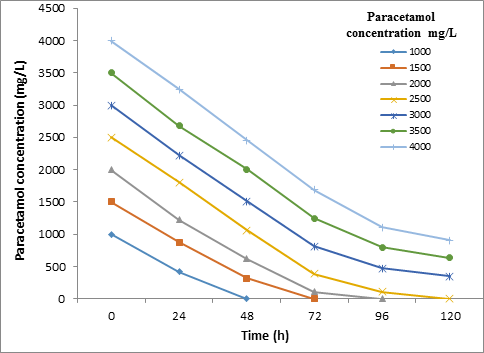
The results of contact time influence on Paracetamol degradation by STB2 and STB4 strains at a different concentration of Paracetamol within 120h are shown in Figure 3 and Figure 4. The Kruskal-Wallis test showed that there are not a significant difference (P = > 0.01) in the biodegradtion effeciency between the STB2 and STB4 starins.
The results of contact time influence on Paracetamol degradation by STB2 and STB4 strains at a different concentration of Paracetamol within 120h are shown in Figure 3 and Figure 4. The Kruskal-Wallis test showed that there are not a significant difference (P = > 0.01) in the biodegradtion effeciency between the STB2 and STB4 starins.
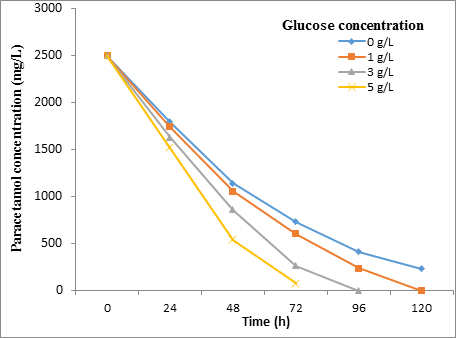
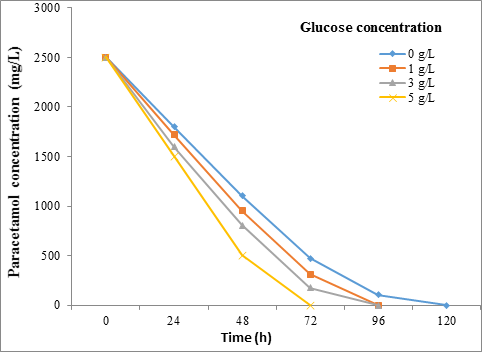
Effect of glucose concentration results
Figure [5] and Figure [6] show the effect of different concentration of glucose on the degradation rate of paracetamol (2500 mg/L) by STB2 and SBT4 strains at 30˚C for 120h. Also, the Friedman test showed a significant difference (P = < 0.01) between the glucose concentration and biodegradation effecincy of all tested strains.
Figure [5] and Figure [6] show the effect of different concentration of glucose on the degradation rate of paracetamol (2500 mg/L) by STB2 and SBT4 strains at 30˚C for 120h. Also, the Friedman test showed a significant difference (P = < 0.01) between the glucose concentration and biodegradation effecincy of all tested strains.
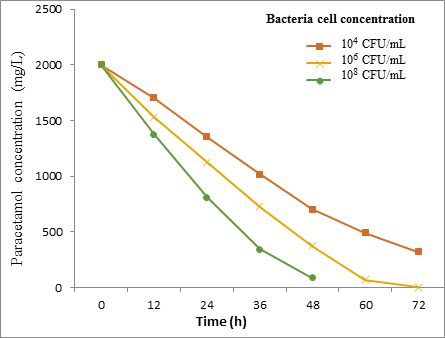
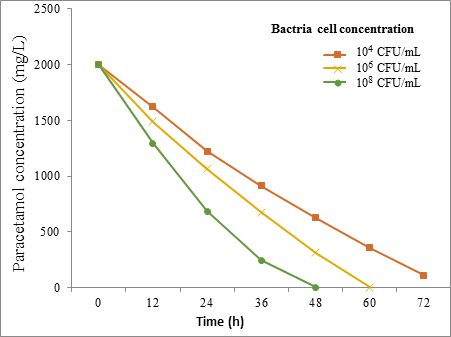
Effect of bacterial cells concentration results
The results obtained from the effect of bacteria cell concentrations on biodegradation of paracetamol (2000 mg/L) at 30˚C for 72h are shown in Figure [7] and Figure [8].
The results obtained from the effect of bacteria cell concentrations on biodegradation of paracetamol (2000 mg/L) at 30˚C for 72h are shown in Figure [7] and Figure [8].
However, the Friedman test showed a significant difference (P = < 0.01) between the bacterial concentration and biodegradation effecincy of all tested strains.
Discussion
Microbial degradation plays an important role in removing paracetamol from the environment [21]. The results of the current study indicate that the site contaminated with the pharmaceutical effluent containing paracetamol is rich with diversity microorganisms which tolerate and use it as the sole source of carbon and energy for growth. Two bacteria strain signed as an STB2 and STB4 were isolated from this contaminated site and identified as P. aeruginosa strain ATCC 10145 with similarity 88% and 97%, respectively.
In similar, several studies were successfully isolated the Pseudomonas species degrading Paracetamol from a different site contaminated with pharmaceutical wastewater containing Paracetamol [13, 22-24].
The pH plays a vital role in biodegradation and gives an understanding of degradation process of paracetamol. The optimum pH for Paracetamol degradation was found between6.5–7.5 on both bacteria stains. Whereas, the decreasing on Paracetamol degradation was observed at pH more than 8.0 or less than 6.0. This result coincides with Hu., et al. [23] who reported that the Paracetamol (1200 mg/L) was completely degraded by P. aeruginosa strain HJ1012 at around pH 7.0 on 48 h. Also, the optimum pH for the Pseudomonas sp. growth on media was reported to be between 7.0-8.0 [12, 22, and 24]. A reduction in Paracetamol biodegradation in more acidic and alkaline media is due to the extreme pH levels which inhibit the bacterial cells growth.
The temperature plays a significant role in microbial growth rate and can affect the degradation of organic compounds through direct effects on enzyme activity [25]. In this study, the effect of temperature was detected on bacterial strains and 30˚C was found to be the most effective temperature for Paracetamol biodegradation. This result is in agreement with the findings Hu., et al. [23] who reported that the optimum temperature was appeared to be at around 30˚C for Paracetamol biodegradation by P. aeruginosa strain HJ1012.
Moreover, a study by Fang., et al. [12] and Yun., et al. [24] observed that the optimum temperature for the Pseudomonas sp. growth on media was recorded at 30˚C. It was established that growth and metabolic activity of Pseudomonas sp. Ap-3 was maximum at 30˚C [26]. Some of the experimental studies on the Paracetamol biodegradation by Pseudomonas sp. were carried out at a temperature of 30˚C and pH of 7.0 [23].
The current study was observed that the degradation rate of Paracetamol was affected by cell concentrations which play a significant role in transport elements. An increase in the bacterial cell concentration resulted in increasing the rate of Paracetamol degradation.
Biodegradation of organic substrates provides microorganisms with energy and building materials that are used for growth of new cells, cell maintenance and co-metabolism of other less degradable substances [27].
The result of this study was revalued that the biodegradation on Paracetamol (2500 mg/L) in the presence of glucose was increased with increasing glucose amounts. A study by Ahmed., et al. [13] observed that the glucose enhance the Pseudomonas sp. strain ST1 growth in media containing Paracetamol (500-5000 mg/L). It indicated that the glucose acts as a facilitator as it provides additional energy supplement to bacteria for Paracetamol biodegradation. The presence of glucose provides a source of carbon and energy for the microbes, thus enhancing their activity to utilize the resistant aromatic amines [19].
The result showed that the STB2 and STB4 could perform complete degradation of Paracetamol at concentrations of 2000 and 2500 mg/L or below, respectively. At 4000 mg/L, 54.62% and 77.4% of Paracetamol concentration were degraded by STB2 and STB4, respectively, within 120 h of reaction. In a similar study by Ahmed., et al. [13] who observed that the isolated Pseudomonas sp. strain ST1 was able to degrade 76.8% of Paracetamol at an initial concentration of 4000 mg/L within 72 h.
Another study by Fang., et al. [12] who reported that the Cupriavidus sp. strain F1, Lysobacter sp. strain F2 and Pseudomonas sp. strain (Figure-2) were able to degrade Paracetamol up to 400 mg/L, 2500 mg/L, and 2000 mg/L, respectively. In a similar study by Hu., et al. [23] who recorded that the P. aeruginosa strain HJ1012 could completely degrade Paracetamol as high as 2200 mg/L within 75 h of reaction.
A study by Zhang., et al. [28] who observed that the Stenotrophomonas sp. strain (Figure 2) and Pseudomonas sp. strain (F1 and F2) were able to complete degradation of Paracetamol at concentrations of 2000, 400, and 2500 mg/L or below in 116h, 60h, and 45h, respectively. Also, it was reported that the Pseudomonas sp. degraded 96% of 1500 mg/L Paracetamol in 12h [24].
Pseudomonas species are environmental organisms known for their ability to degrade aromatic compounds of environmental concern [29]. The adaptability of Pseudomonas species to different organic compounds makes it an attractive organism for its use in biodegradation for wide ranges of organic substances that present in wastewater.
Conclusion
Pharmaceutical wastewater is the most potential sources to isolate high-performance Paracetamol degrading microbes. The isolated bacterial strains were showed a high degradation efficiency of Paracetamol and could tolerate Paracetamol at 4000 mg/L. The rate of Paracetamol degradation was also affected by the environmental factors. Therefore, P. aeruginosa may prove to be important in bioremediation and wastewater treatment. A further investigation on Paracetamol metabolic pathways may increase further knowledge about the abilities of these bacterial strains towards biodegradation process.
References
- Martindale W. “The complete drug reference, cough suppressants, expectorants, mucolytics and nasal decongestants 36th edition.” The pharmaceutical Press London UK (2009).
- Zhang X., et al. “Photo degradation of acetaminophen in TiO2 suspended solution”. Journal of Hazardous Materials 157.2.3 (2008): 300-307.
- Alajmi HM. “Effect of physical, chemical and biological treatment on the removal of five pharmaceuticals from domestic wastewater in laboratory-scale reactors and a full-scale plan”. University of Newcastle upon Tyne (2014): 50-87.
- Edrees WH et al. “A review on comparative study between the physicochemical and biological processes for Paracetamol degradation”. Universal Journal of Pharmaceutical Research2.2 (2017): 9-13.
- Kleywegt S., et al. “Pharmaceuticals, hormones and bisphenol an in untreated source and finished drinking water in Ontario, Canada: Occurrence and treatment efficiency”. Science of the Total Environment 409.8 (2011): 1481-1488.
- Zimmerman MJ. “Occurrence of organic wastewater contaminants, pharmaceuticals, and personal care products in selected water supplies. Cape Cod, Massachusetts, June 2004”. US Geological Survey Open-File Report 2005-1206 (2005): 1-16.
- Lin YA and Tsai T. “Occurrence of pharmaceuticals in Taiwan's surface waters: Impact of waste streams from hospitals and pharmaceutical production facilities”.Science of the Total Environment 407.12 (2009): 3793-3802.
- Thomas KV., et al. “Source to sink tracking of selected human pharmaceuticals from two Oslo city hospitals and a wastewater treatment works”. Journal of Environmental Monitoring 9.12 (2007): 1410-1418.
- Wu S., et al. “Paracetamol in the environment and its degradation by microorganisms”. Applied Microbiology and Biotechnology 96.4 (2012): 875-884.
- Brillas E., et al. “Mineralization of Paracetamol in aqueous medium by anodic oxidation with a boron-doped diamond electrode”. Chemosphere 58.4 (2005): 399-406.
- Trovo AG., et al. “Photo degradation of the pharmaceuticals amoxicillin, bezafibrate and Paracetamol by the photo-Fenton process: Application to sewage treatment plant effluent”. Journal of Photochemistry and Photobiology A: Chemistry 198.2.3 (2008): 215-220.
- Fang W., et al. “Study on bacterial function of high-efficiency Paracetamol-degrading aerobic granule. Master's thesis”. Zhejiang University of Technology Microbiology, China (2011): 1-15.
- Ahmed S., et al. “Isolation and characterization of a Pseudomonas strain that degrades 4-acetamidophenol and 4-aminophenol”. Biodegradation 12.5 (2001): 303-309.
- Chen L., et al. “Rapid sanger sequencing of the 16S rRNA gene for identification of some common pathogens”. PLoS ONE 9.2 (2014).
- Gusseme BD., et al. “Degradation of acetaminophen by Delftia tsuruhatensis and Pseudomonas aeruginosa in a membrane bioreactor”. Water Res 45.4 (2011): 1829-1837.
- Bergey’s Manual of Systematic Bacteriology Second edition. Michigan State University; East Lansing, USA (2004).
- Khan AS., et al. “Degradation of 4-aminophenol by newly isolated Pseudomonas sp. strain ST-4”. Enzymatic Microbiology and Technology 38.1.2 (2006): 10-113.
- Shourian M., et al. “Efficient phenol degradation by a newly characterized Pseudomonas sp. SA01 isolated from pharmaceutical wastewaters”. Desalination 246.1.3 (2009): 577-594.
- Khan SA., et al. “Influence of pH, temperature and glucose on biodegradation of 4-aminophenol by a novel bacterial strain, Pseudomonas sp. ST-4”. African Journal of Biotechnology 8.16 (2009): 3827-3831.
- United States Pharmacopoeia 30-NF/25: Monograph: Paracetamol (acetaminophen): The United States Pharmacopeial Convention, Rockville, USA; (2005).
- Edrees WH., et al. “Occurrence of Paracetamol in aquatic environments and transformation by microorganisms: A review”. Chronicles of Pharmaceutical Science 1.6 (2017): 341-355.
- Edrees WH., et al. “Isolation and identification of a new bacterial strains degrading paracetamol isolated from Yemeni environment”. Clinical Biotechnology and Microbiology 1.6 (2018): 257-270.
- Hu J., et al. “Degradation of Paracetamol by Pseudomonas aeruginosa strain HJ1012”.Journal of Environmental Science and Health, Part A: Toxic/Hazardous Substances and Environmental Engineering 48.7 (2013): 791-799.
- Yun MA., et al. “Isolation and characterization of a acetaminophen degrading bacterium”. Journal of Zhejiang University of Technology 1 (2013): 35-38.
- Pettersson M and Baath E. “Temperature-dependent changes in the soil bacterial community in limed and unlimed soil”. FEMS Microbiology Ecology 45.1 (2003): 13-21.
- Takenaka S., et al. “Complete nucleotide sequence and functional analysis of the gene for 2-aminophenol metabolism from Pseudomonas sp. AP-3”. Archives of Microbiology 174.4 (2000): 265-272.
- Cornelissen G and Sijm DT. “An energy budget model for the biodegradation and co-metabolism of organic substances”. Chemosphere 33.5 (1996): 817-830.
- Zhang L., et al. “Degradation of paracetamol by pure bacterial cultures and their microbial consortium”. Applied Microbiology and Biotechnology 97.8 (2013): 3687-3698.
- Neumann G., et al. “Simultaneous degradation of atrazine and phenol by Pseudomonas sp. strain ADP: Effect of toxicity and adaptation”. Applied and Environmental Microbiology 70.4 (2004):1907-1912.
Citation:
Wadhah H Edrees., et al. “Biodegradation of Paracetamol by Native Bacterial Strains Isolated from Yemeni Pharmaceutical
Wastewater Plant in Sana’a”. Chronicles of Pharmaceutical Science 2.2 (2018): 512-522.
Copyright: © 2018 Wadhah H Edrees., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.