Case Report
Volume 1 Issue 3 - 2018
Synchronous Primary Carcinoma Breast and Primary Malignant Mixed Mullerian Tumor of the Uterus- An Extremely Rare Presentation
1Associate professor of surgical oncology, Department of surgical oncology, Sri Ramachandra University, Chennai - 600116, India
2Associate Professor of Obstetrics & Gynaecology, Department of Obstetrics & Gynaecology, Sri Ramachandra University, Chennai-600116, India
3Professor of Obstetrics&Gynaecology, Department of Obstetrics & Gynaecology, Sri Ramachandra University, Chennai-600116, India
4Associate Professor of Pathology, Department of Pathology, Sri Ramachandra University, Chennai-600116, India
2Associate Professor of Obstetrics & Gynaecology, Department of Obstetrics & Gynaecology, Sri Ramachandra University, Chennai-600116, India
3Professor of Obstetrics&Gynaecology, Department of Obstetrics & Gynaecology, Sri Ramachandra University, Chennai-600116, India
4Associate Professor of Pathology, Department of Pathology, Sri Ramachandra University, Chennai-600116, India
*Corresponding Author: Gouthaman Shanmugasundaram, Associate professor of surgical oncology, Department of surgical oncology, Sri Ramachandra University, Chennai - 600116, India.
Received: December 20, 2017; Published: January 26, 2018
Abstract
Synchronous primary carcinoma of the breast and Malignant Mixed Mullerian Tumor (MMMT) of the uterus in the absence of tamoxifen use is an extremely rare scenario. The synchronous presentation could be due to common risk factors for both malignancies. Malignant mixed Mullerian tumor is a rare tumor that shows both epithelial and stromal malignant differentiation. Malignant mixed Mullerian Tumor (MMMT) is subdivided into the homologous and heterologous type, based on the histopathologic differentiation of the stromal component of the tumor. Whole body PET-CT facilitates staging of these synchronous lesions. Inview of early lesions in both uterus and the breast, we proceeded with surgery for both in the same sitting. Adjuvant treatment guidelines and prognosis are not clear in view of the rare presentation of such a combination.
Keywords: Malignant Mixed Mullerian Tumor (MMMT); Tamoxifen; Nulliparity, PET-CT; Vimentin, Desmin
Introduction
Malignant Mixed Müllerian tumor (MMMT) is a rare aggressive malignant tumor, which demonstrates both malignant epithelial (carcinoma) and mesenchymal (sarcoma) components [1]. Nulliparity and obesity are associated with increased risks for both breast and uterine cancers [2]. Few cases of Malignant mixed mullerian tumor of the uterus following tamoxifen therapy for carcinoma breast has been described since 1988 [3]. Tamoxifen also has an estrogenic effect on the uterus, thereby increasing the risk of endometrial hyperplasia, endometrial polyp and endometrial neoplasms such as endometrial adenocarcinoma and malignant mixed Mullerian tumour (MMMT) [4]. Only one case of synchronous primary carcinoma of the breast and Malignant mixed tumor of the uterus without the use of tamoxifen has been described in the world literature so far [5]. Here we describe the second case in the world literature.
Case Report
A 65 year old postmenopausal multiparous lady presented with bleeding pervaginum of 1 month duration. She had multiple co-morbidities - diabetes, systemic hypertension, hypothyroidism and hypercholesterolemia. Abdominal examination revealed a small reducible umbilical hernia. On pelvic examination, she had bulky uterus and bilateral parametrium were supple. No significant inguinal nodes were palpable. Incidentally on examination of the breast, we found a 4*3 cm mass in the upper inner quadrant of the left breast. Examination of left axilla revealed few nonmatted mobile axillary nodes. With this background, we proceeded with the evaluation of the patient. Haematological and biochemical investigations were within normal limits. X-Ray Mammogram of both the breasts with Ultrasound correlation showed an irregular spiculated lesion with micro and macro calcifications noted in the upper central quadrant (1.5*1.5*1.7cm)-BIRADS V lesion left breast. Endometrial biopsy done at an outside center was suggestive of High grade stromal sarcoma. Ultrasound guided trucut biopsy from the breast lesion was done at our center. To our surprise, it was suggestive of invasive mammary carcinoma with adjacent low grade DCIS.
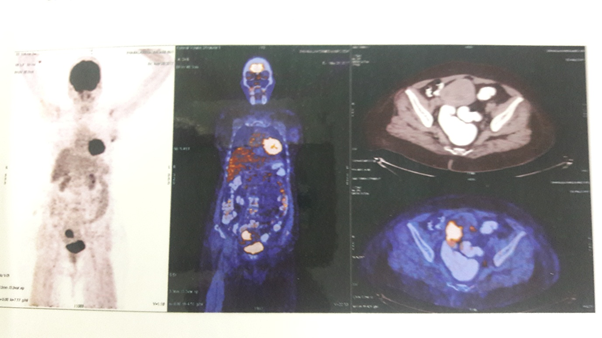
Inview of two synchronous primary lesions in the breast and the uterus, we proceeded with whole body PET-CT. Whole body PET CT showed hypermetabolic activity of the irregular hypodense lesion measuring 26.4mm*49.4mm*40.8mm in the endometrial cavity infiltrating the myometrium of uterus (SUV max 22.5) and in the irregular lobulated soft tissue density lesion measuring 14.7mm*13.2mm*17.4mm in the lower inner quadrant of left breast (SUV max 6.2). No significant metabolic activity in the other parts of the body (Figure 1).
Figure 1: Whole Body PET-CT showing significant FDG uptake in the left breast (SUV max 22.5) and the endometrial cavity (SUV max 6.2).
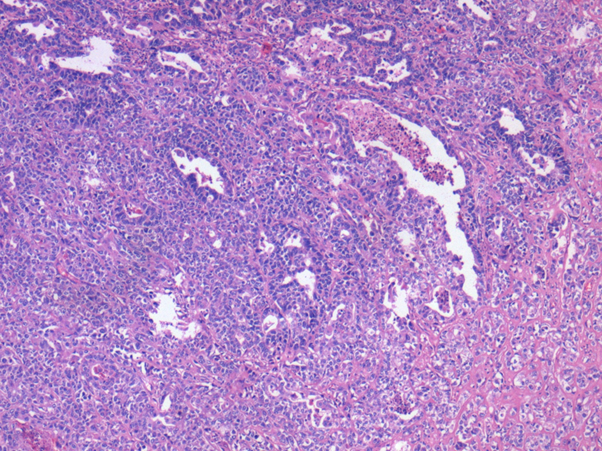
With this background, we proceeded with Total Abdominal Hysterectomy with Bilateral salphingo-oophorectomy and Left Modified radical mastectomy by two simultaneous surgical teams. Pelvic nodal dissection was not done as the patient could not withstand prolonged anesthesia due to perioperative ECG changes. Postoperative period was uneventful. Histopathology was suggestive of malignant mixed mullerian tumor with heterologous elements in the uterus and invasive mammary carcinoma carcinoma with micro papillary features in the left breast. Macroscopically, the uterus showed a solid, firm, grey-white lesion in the fundus measuring 7.5*5.5*3.5cm.Microscopic features were typical of high grade malignant mullerian tumor with heterologous elements (osteoid and chondroid elements) (Figure 2).
Figure 2: Microscopic appearance showing endometrial adenocarcinoma with intervening pleomorphic stromal cells and atypical mitosis (sarcomatous component).
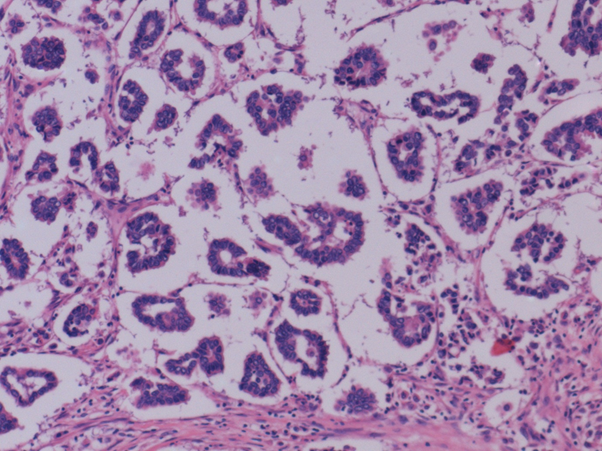
The tumor was limited to the endometrium without invasion of myometrium and designated as pT1a.Immunohistochemistry was not done as the microscopy was typical of malignant mixed mullerian tumor. Macroscopic examination of the left breast specimen revealed 4.4*3.2*2.2cm solid, firm infiltrative lesion. Microscopic features are that of grade I invasive mammary carcinoma with micro papillary features (20%) (pT2 lesion) (Figure 3).
Figure 3: Microscopic appearance showing breast tissue with infiltrating tumour arranged in micro papillary pattern.
17 out of 31 left axillary nodes dissected showed malignant features (pN3a). Patient is planned for adjuvant chemotherapy and adjuvant radiation therapy to both the chest wall and pelvis.
Discussion
Malignant Mixed Mullerian Tumor, otherwise called as Carcinosarcoma, is a rare tumor that shows both epithelial and stromal malignant differentiation. The risk factors for uterine carcinosarcoma include obesity, exogenous estrogen, exposure to radiation and tamoxifen [6]. Carcinosarcoma shares risk factors with breast cancer such as obesity and null parity. Recent study indicated that nulliparity and overweight had a synergistic effect on breast cancer risk in elderly women [7]. Tamoxifen associated with 14 fold increased risk of carcinosarcoma has been documented [8]. Only obesity was a risk factor in our case.
Carcinosarcomas are subdivided into the homologous and heterologous type, based on the histopathologic differentiation of the stromal component of the tumor. Immunohistochemical analyses of various antigens have been performed to evaluate the nature of different malignant components of the uterine carcinosarcoma. Vimentin and Desmin were positive in the sarcomatous portion of carcinosarcoma, focally in the stromal component.CD10 was sensitive immunohistochemical marker of neoplastic endometrial stromal component [9].
Positive staining with CD10 in the fusiform cells which were negative with muscle marker SMA might indicate endometrial stromal sarcoma [10]. Therefore, immunohistochemical staining panel of CD10, Desmin, Vimentin and SMA can be used to support making a definitive diagnosis for carcinosarcoma. Immunohistochemical studies were not done in our case as the pathological features were typical of heterologous type of carcinosarcoma with osteoid and chondroid elements.
The primary treatment for Carcinosarcoma of the uterus includes total abdominal hysterectomy with bilateral salphingo-oophorectomy with surgical staging, peritoneal lavage for cytology, omental and peritoneal biopsies and consideration of maximal tumor debulking for gross disease [11]. Lymphadenectomy as part of surgical staging was not done in our case in view of co-morbidities and a perioperative cardiac event. In order to reduce the operative time, the modified radical mastectomy and Total abdominal hysterectomy with bilateral-salphingo-oophorectomy were done by two surgical teams simultaneously.
For women with carcinosarcoma that invades less than half of the myometrium (stage IA), there are low quality data to inform the benefit of adjuvant treatment. Some experts prefer observation rather than adjuvant treatment because women with stage IA carcinosarcoma have a better prognosis compared with those with IB or later stage disease [12]. In addition, it is unclear if their prognosis is improved with any form of adjuvant treatment [13-15]. For patients with stage IA without myometrial invasion, options include observation, systemic therapy with or without vaginal brachytherapy or External Beam Radiotherapy (EBRT) with or without brachytherapy.
For all other patients with more advanced disease, systemic therapy with or without tumor-directed radiotherapy is the preferred option [16]. Our patient is planned for adjuvant radiation therapy to the pelvis as the pelvic lymph nodes were not addressed as part of staging. Malignant Mixed Mullerian Tumor (Carcinosarcoma) following tamoxifen therapy for carcinoma breast has been reported in few case reports [17,18] and case series [19]. But the synchronous presentation of carcinosarcoma of the uterus and carcinoma breast has been reported in only one case in the world literature [5]. This is the second case reported in the world literature.
The treatment of breast cancer includes the treatment of local disease with surgery, radiation therapy, or both and systemic therapy with chemotherapy, endocrine therapy, biologic therapy, or combination of these [20]. Several randomized trials document that mastectomy is equivalent to breast conservation therapy as primary local treatment for the majority of women with stage I and stage II breast cancers [21]. Our patient opted for Modified radical Mastectomy (Mastectomy with level I,II,III axillary nodal dissection) though the lesion was suitable for Breast conservation therapy.
Several combination chemotherapy regimens are appropriate to consider when adjuvant cytotoxic chemotherapy for the treatment of breast cancer is planned. The preferred regimens include dose – dense doxorubicin and cyclophosphamide (AC) with dose-dense sequential paclitaxel, dose-dense AC followed by weekly paclitaxel and docetaxel plus cyclophosphamide (TC). Randomized controlled trials have shown that a diseasefreesurvival (DFS) and Overallsurvival (OS) advantage is conferred by the irradiation of chest wall and regional lymphnodes in women with positive axillary lymphnodes after mastectomy and axillary lymph node dissection [19].
Our patient is planned for adjuvant chemotherapy and radiation to the chestwall. The prognosis of the patients with combination of mixed mullerian tumor denovo without tamoxifen exposure and Carcinoma breast with high nodal positivity is probably poor inview of the aggressive nature of both malignancies. However, there are no robust data as the combination is extremely rare.
Conclusion
Synchronous presentation of breast and uterine malignancies is rare. Meticulous clinical breast examination of the breast is mandatory in all cases of uterine malignancies to detect early breast lesions. Evaluation of both the abdomen and breast by imaging, as clinically indicated, would help identifying synchronous lesions. Synchronous breast and uterine lesions should be borne in mind in cases with uterine malignancies.
References
- Ling Shen., et al. “Synchronous uterine carcinosarcoma and contralateral breast cancer after tamoxifen therapy: a case report”. International Journal of Clinical and Experimental Pathology 7.8 (2014): 5295–5301.
- Gleicher N. “Why are reproductive cancers more common in nulliparous women?” Reproductive BioMedicine Online 26.5 (2013): 416–419.
- Saim M., et al. “Malignant mixed müllerian tumor of the uterus following tamoxifen for breast cancer: case report”. Gynécologie Obstétrique & Fertilité 36.2 (2008):166-168.
- Gupta M and Kiruthiga KG. “Malignant mixed Mullerian tumour of uterus secondary to tamoxifen therapy for hormone responsive breast cancer”. BMJ Case Reports (2015):
- Tsekeris P and Dimou S. “Synchronous endometrial carcinosarcoma and breast carcinoma: a case report”. European Journal of Gynaecological Oncology 21.3 (2000): 309-310.
- Kanthan R and Senger JL. “Uterine carcinosarcomas (malignant mixed mullerian tumours): a review with special emphasis on the controversies in management”. Obstetrics and Gynecology International 2011(2011): 1-13.
- Opdahl S., et al. “Joint effects of nulliparity and other breast cancer risk factors”. British Journal of Cancer 105.5 (2011): 731–736.
- Swerdlow AJ and Jones ME. “Tamoxifen treatment for breast cancer and risk of endometrial cancer: a case-control study”. The Journal of the National Cancer Institute 97.5 (2005): 375–384.
- Abeler VM and Nenodovic M. “Diagnostic immunohistochemistry in uterine sarcomas: a study of 397 cases”. International Journal of Gynecological Pathology 30.3 (2011): 236–243.
- McCluggage WG., et al. “CD10 is a sensitive and diagnostically useful immunohistochemical marker of normal endometrial stroma and of endometrial stromal neoplasms”. Histopathology 39.3 (2001):273–278.
- NCCN Guidelines Version 2.2016- Endometrial carcinoma
- Page BR., et al. “Does the FIGO 2009 endometrial cancer staging system more accurately correlate with clinical outcome in different histologies? Revised staging, endometrial cancer, and histology”. International Journal of Gynecological Cancer 22 (2012): 593.
- Reed NS., et al. “Phase III randomized study to evaluate the role of adjuvant pelvic radiotherapy in the treatment of uterine sarcomas stages I and II: an European Organization for Research and Treatment of Cancer Gynaecological Cancer Group Study (protocol 55874)”. European Journal of Cancer 44.6 (2008): 808-818.
- Omura GA., et al. “A randomized clinical trial of adjuvant adriamycin in uterine sarcomas: a Gynecologic Oncology Group Study”. Journal of Clinical Oncology 3.9 (1985): 1240-1245.
- Cantrell LA., et al. “A multi-institutional cohort study of adjuvant therapy in stage I-II uterine carcinosarcoma”. Gynecologic Oncology 127.1 (2012): 22-26.
- NCCN GUIDELINES Version 2.2017- Endometrial carcinoma
- Gupta M and Kiruthiga KG. “Malignant mixed Mullerian tumour of uterus secondary to tamoxifen therapy for hormone responsive breast cancer”. BMJ Case Reports (2015):
- Magnani KK., et al. “Malignant mixed Müllerian tumor of the uterus associated with tamoxifen therapy for breast cancer”. Indian Journal of Pathology and Microbiology 53.4 (2010): 886-887.
- Behtash N., et al. “Uterine malignancy following tamoxifen use in breast cancer patients in Iran: case series and literature review”. Asian Pacific Journal of Cancer Prevention 10.1 (2009):163-166.
- NCCN guidelines Version 3.2015
- Veronesi U., et al. “Twenty –year follow-up of a randomized study comparing breadt –conserving surgery with radical mastectomy for early breast cancer”. The New England Journal of Medicine 347.16 (2002): 1227-1232.
Citation:
Gouthaman Shanmugasundaram., et al. “Synchronous Primary Carcinoma Breast and Primary Malignant Mixed Mullerian
Tumor of the Uterus- An Extremely Rare Presentation”. Gynaecology and Perinatology 1.3 (2018): 152-157.
Copyright: © 2018 Gouthaman Shanmugasundaram., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.