Research Article
Volume 1 Issue 2 - 2017
Relationship between Viscosity and Some Chemical Characteristics of Frying Oils
1Biochemisrty Dept., Faculty of Agriculture Beni-Suef University
2Oils & Fats Research Dept., Food Technology Research Institute, Agricultural Research Center
3Oils & Fats Research Dept., National Research Center. Egypt
2Oils & Fats Research Dept., Food Technology Research Institute, Agricultural Research Center
3Oils & Fats Research Dept., National Research Center. Egypt
*Corresponding Author: Shaker M Arafat, Oils & Fats Research Dept., Food Technology Research Institute.
#Corresponding Author: Amany M Basuny, Biochemisrty Dept., Faculty of Agriculture Beni-Suef University. Agricultural Research
Center.
Received: April 29, 2017; Published: May 15, 2017
Abstract
The viscosity and chemical characteristics (acidity, peroxide value, polar content, polymer content and oxidized fatty acids) of sunflower
oil, cottonseed oil and palm olein during deep fat frying at 180ºC ± 5ºC for 10 hrs were investigated. Results showed a relative
between viscosity and chemical properties. Measurements of viscosity can provide a good overall estimate of frying oil quality.
Keywords: Sunflower oil; Cottonseed oil; Palm olein; Deep-fat frying; Viscosity
Introduction
Deep frying is one of the most common methods used for the preparation of food. Repeated frying causes several oxidation and thermal reactions which results in change in physico-chemical, nutritional and sensory properties of the oil [1]. During frying, due to hydrolysis, oxidation and polymerization process the composition of oil changes which in turn changes the flavor and stability of its compounds [2]. During deep frying different reactions depend on some facts such as replenishment of fresh oil, frying condition, original quality of frying oil and decrease in their oxidative stability [3]. Atmospheric oxygen reacts instantly with lipid and other organic compounds of the oil to cause structural degradation in the oil which leads to loss of quality of food and is harmful to human health [4]. Therefore, it is essential to monitor the quality of oil to avoid the use of abused oil due to the health consequences of consuming foods fried in degraded oil, to maintain the quality of fried foods and to minimize the production costs associated with early disposal of the frying medium [5]. Different physical and chemical parameters of edible oil were used to monitor the compositional quality of oils [6,7].
These physico-chemical parameters include iodine value, saponification value, viscosity, density and peroxide value. Edible oils are one of the main constituents of the diet used for cooking purposes. Several researchers studied the impact of temperature on the stability, viscosity, peroxide value and iodine value to assess the quality and functionality of the oil [8,9,10]. Viscosity measurements can provide a good overall estimate of frying fat or oil qual [11]. The viscosity of vegetable oils is affected by a number of factors. These include the physical and chemical properties of oil such as the density, molecular weight, melting point and degree of unsaturation. A factor that greatly affects the viscosity of oils is temperature. It has been reported that the viscosity of oils and fats decreased linearly with temperature [12]. In vegetable oils, viscosity increase with chain lengths of triglyceride fatty acids and decrease with unsaturation in other words, increase with hydrogenation. So, viscosity is a function of molecules dimension and orientation [13]. The sole objective of the present study was to explore the effect of frying on viscosity and physico-chemical properties of sunflower, cotton seed and palm olein during frying of potato chips.
Materials and Methods
Materials: Sunflower oil, cottonseed oil palm olein and potato tubers (Solanum tubersum) were purchased from local market in Giza, Egypt. All solvents and reagents from various suppliers were of the highest purity needed for each application and used without further purification.
Sample preparation: Potatoes were peeled and then sliced using a Mandolin Slicer (Matfer model 2000, France) to a thickness gage (Mi tutoyo thickness Gage, Japan), and cut into a diameter of 5.08 cm using a cylindrical metal cutter. The potato slices were rinsed with distilled water to eliminate starch material on the surface and then blotted with paper towels before each experiment. The samples were placed in aluminum foil to avoid any moisture loss before further processing.
Frying process: A known amount (Ca. 2 kg) of each of the sunflower oil, cottonseed, and palm olein were placed separately in a stainless steel pan fryer (40 cm diameter X 10 cm height). The aforementioned oil were separately heated at 180°C ± 5°C, then 100 gm of slices potato were fried every 30 min, for 10 hrs during two consecutive days. The fryer was left unconverted during the frying period. The fryer was turned off at the end of the frying experiment for each day and the oil was allowed to cool to 60°C then stored at room temperature during the night till next day to continue the frying. Oil samples were taken every 2 hrs and the entire continuous frying period was 10 hrs. The oil samples were left to cool down and then stored at-18°C for physic-chemical properties.
Quality assurance of fried oil: Viscosity measurement of oil at 30°C was carried out the Brookfield LV viscometer Mode3l T.C.500 (Brookfield Engineering Laboratories Stoughton, MA, USA) [14]. Methods in the [15] were used to determine acid value and peroxide value. Insoluble oxidized fatty acids and insoluble polymer contents of sunflower oil samples were determined according to the methods of [16]. Polar and non-polar components in oil samples were separated by column chromatography according to the method described by [17]. Column-packing material used was silica gel (60-120) mesh and the polar components were eluted with a solvent mixture composed of 87% light petroleum and 13% diethyl ether.
Results and Discussion
Acid value was used to assess frying oil degradation and is related to fried food quality [18]. The changes in the acid values of cottonseed,
sunflower and palm oil during deep-fat frying at 180°C ± 5°C are given in Figure 1. Acid value of sunflower and cottonseed oil
increased strongly after 10 hrs during frying compared with palm oil which increased slightly after 10 hrs. The higher oxidative stability
of palm oil compared to cottonseed and sunflower oil is due to the high oleic acid (monounsaturated) and low polyunsaturated fatty
acid content of the triacylglycerols. Hydroperoxides are the primary products of lipid oxidation; therefore, determination of peroxide
value can be used as an oxidation index for the early stages of lipid oxidation. During frying of potato chips in cottonseed and sunflower
oil, peroxide value increased strongly with frying time Figure 2. While palm oil after 10 hrs increased slightly. Cottonseed oil the highest
values of peroxide at the end of frying period (10 hrs). Unsaturated fatty acids easily react with oxygen to form peroxides, whereas, the
lowest value was recorded for palm oil. This finding is line with the degree of oil unsaturation.
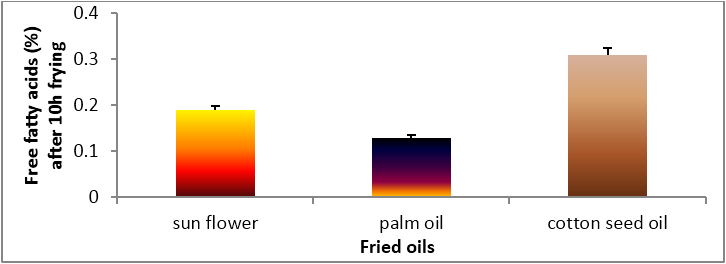
Figure 1: Changes in free fatty acids of sunflower oil, palm olein and cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
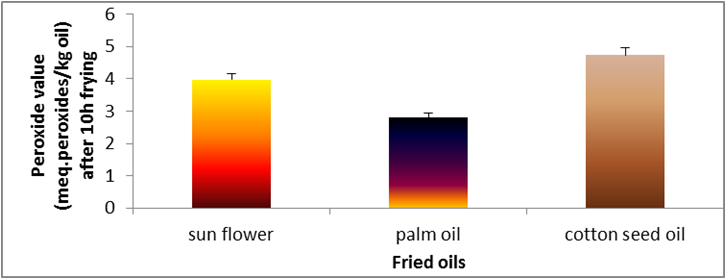
Figure 2: Changes in free peroxide value of sunflower oil, palm olein and cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
The level of polar compounds is a good indicator of the overall quality of frying oils, providing critical information about the amount
of newly formed compounds having higher polarity than triacylglycerols [19]. Figure 3 shows the total polar compounds of cottonseed,
palm and sunflower during deep-fat frying 180°C ± 5̊°C. The lowest value of total polar compounds was observed for palm oil, while the
highest value of total polar compounds was observed for cottonseed oil at the end of frying period 10 hrs. Decreasing the linoleic and
increasing the oleic acid of palm oil produced good frying stability as measured by total polar compounds [20].
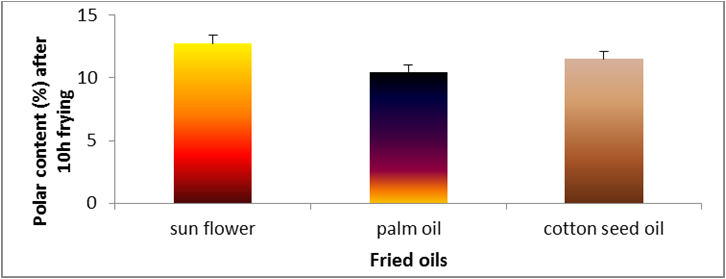
Figure 3: Changes in polar content (%) of sunflower oil, palm olein and cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
The changes in polymer content of cottonseed, palm and sunflower oil during deep-fat frying at 180̊°C ± 5̊°C are shown in Figure
4. The highest polymer content was recorded for sunflower oil at the end of frying process. On the other hand, palm oil had the lowest
value at the end of frying period. Figure 5 demonstrates the oxidized fatty acids of oils (cottonseed, palm and sunflower) during deep-fat
frying at 180̊°C ± 5̊°C for 10 hrs. The highest value of oxidized fatty acids was recorded for sunflower oil at the end of frying period. On
the other hand, palm had the lowest value of oxidized fatty acids.
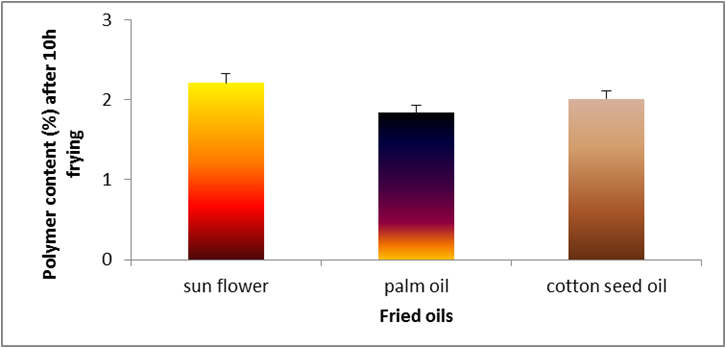
Figure 4: Changes in free polymer content (%) of sunflower oil, palm olein and cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
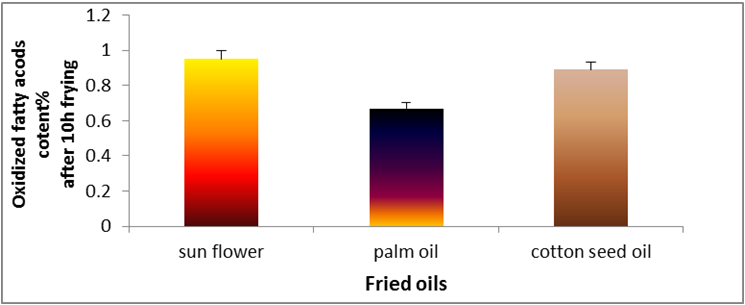
Figure 5: Changes in free oxidized fatty acids (%) of sunflower oil, palm olein and cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
Viscosity of the frying oil is an important factor in determining the total value of oil sticking in the large cavities in the crust of the food product [21]. Viscosity of oils increased during frying and was influenced by frying time and the changes in viscosity indicate structural change. The tending of viscosity to increase during frying of the oil has been found to correlate with formation of polymers. The increasing in viscosity indicates that polymers, which are the higher molecular weight fraction of the degradation products, are increased by increasing frying time; also, increasing in viscosity may be due to a hidden effect of free fatty acids produced by hydrolysis reactions and other small molecular weight decomposition products during frying [2].
Figures 6 to 20 show relationship between viscosity and other properties of oil (cottonseed, palm and sunflower) during frying at 180̊°C ± 5̊°C for 10hrs. The dependence of viscosity on the oxidative changes in polymerization period of vegetable oils was investigated by correlating the oil viscosity with other properties. So, an effort was made to investigate how the degree of chemical properties affected oil viscosity. Highly positive correlation was observed between viscosity and acid value, peroxide value, polar content, polymer content and oxidized fatty acids in palm oil (Figure 6-10). Also, highly positive correlation was observed between viscosity and chemical properties in cottonseed oil (Figures 11-20).
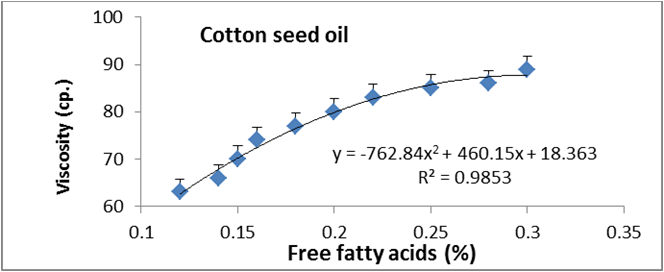
Figure 6: Relationship between viscosity and free fatty acid of cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
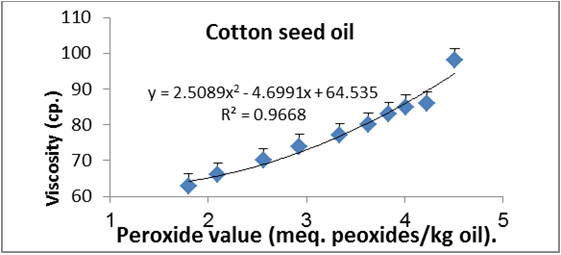
Figure 7: Relationship between viscosity and peroxide value of cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
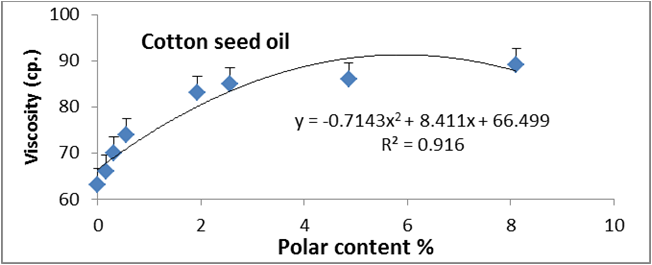
Figure 8: Relationship between viscosity and polar content (%) of cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
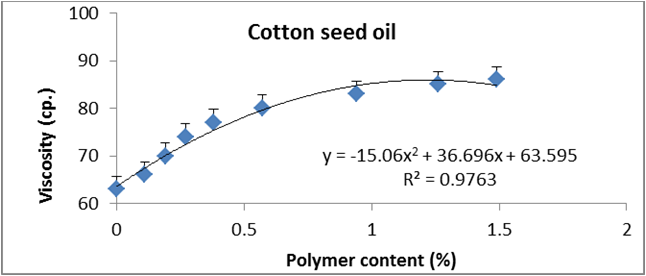
Figure 9: Relationship between viscosity and polymer content (%) of cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.

Figure 10: Relationship between viscosity and oxidized fatty acids of cottonseed oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
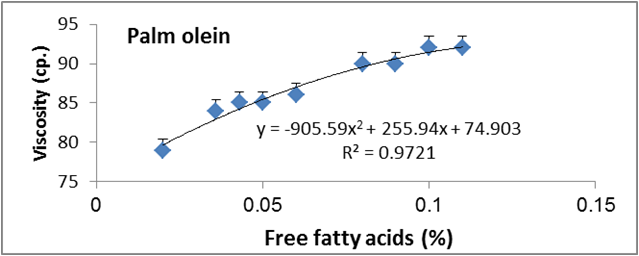
Figure 11: Relationship between viscosity and free fatty acid of palm olien.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
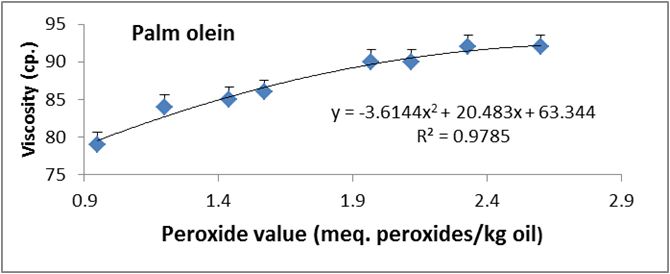
Figure 12: Relationship between viscosity and peroxide value of palm olien.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
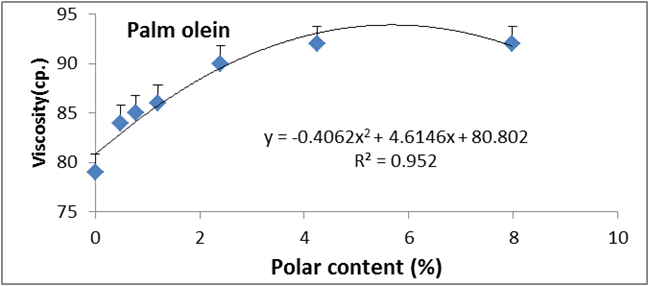
Figure 13: Relationship between viscosity and polar content (%) of palm olien.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
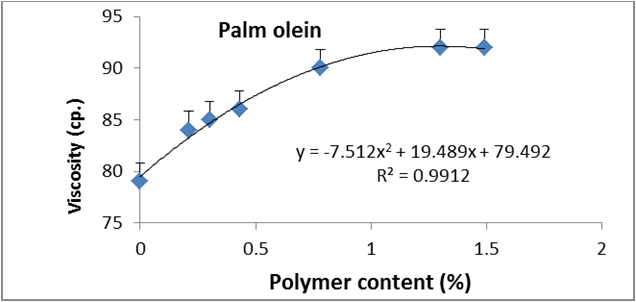
Figure 14: Relationship between viscosity and polymer content (%) of palm olien.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
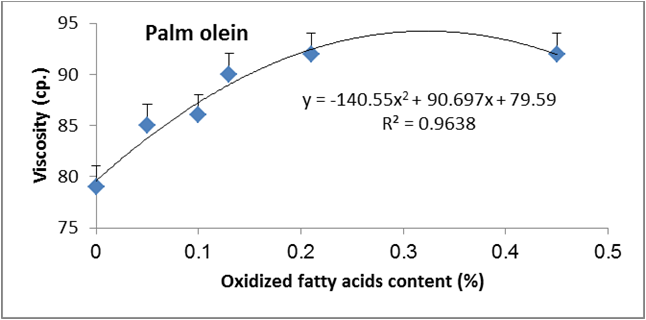
Figure 15: Relationship between viscosity and oxidized fatty acids content (%) of palm olien.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
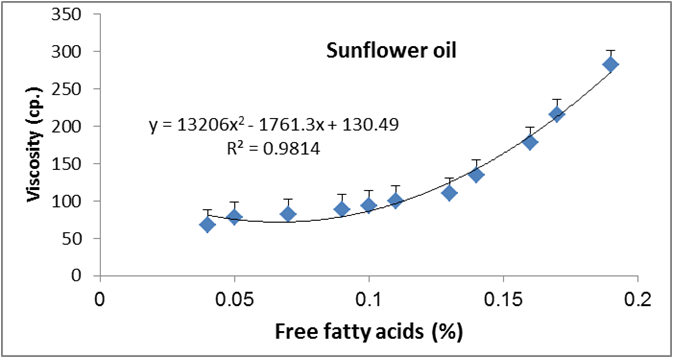
Figure 16: Relationship between viscosity and free fatty acid of sunflower oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
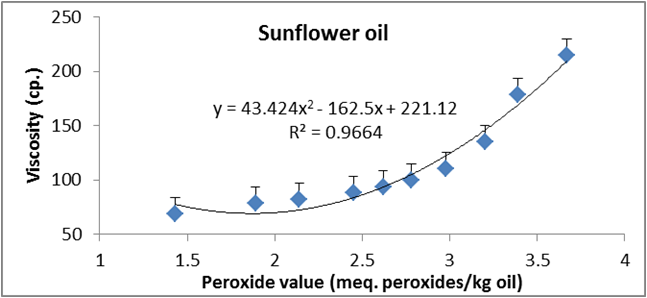
Figure 17: Relationship between viscosity and peroxide value of sunflower oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
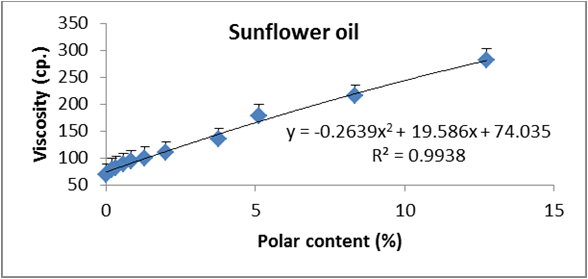
Figure 18: Relationship between viscosity and polar content (%) of sunflower oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
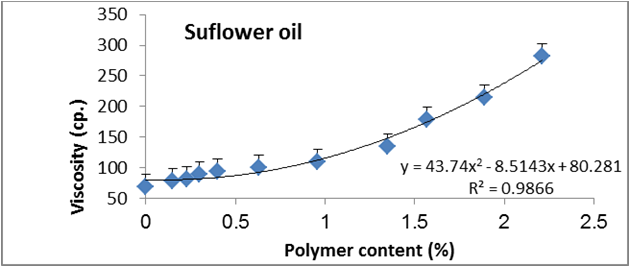
Figure 19: Relationship between viscosity and polymer content (%) of sunflower oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
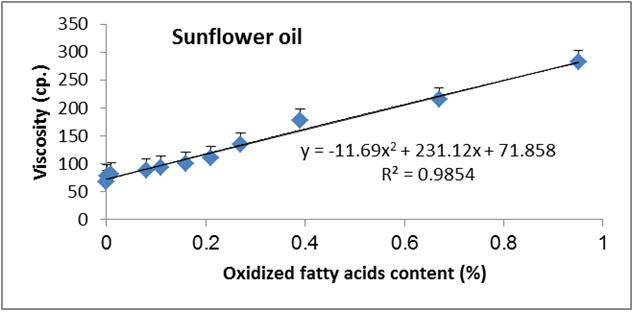
Figure 20: Relationship between viscosity and oxidized fatty acid content (%) of sunflower oil.
Data are expressed as mean ± SD values given represent means of three determinations.
Data are expressed as mean ± SD values given represent means of three determinations.
Conclusions
From these results of this study we can see all the characteristics of oils depending on the viscosity. This depends on the resulting
curve and equation from the determined of viscosity and other properties of oils; therefore, we can know the viscosity values referee on
oil quality and knowledge of the other properties of oil because rheology plays a very important role in oils quality during deep frying
process.
References
- Che Man YB and Jasvir I. “Effect of rosemary and sage extracts on frying performance of refined, bleached and deodorized (RBD) palm olein during deep-fat frying”. Food Chemistry 69.3 (2000): 301-367.
- Gloria H and Aguilera JM. “Assessment of the quality of heated oils by differential scanning calorimetry”. Journal of Agricultural and Food Chemistry 46.4 (1998): 1363-1368.
- Choe E and Min DB. “Chemistry of deep-fat frying oils”. Journal of Food Science 72.5 (2007): 77-86.
- Bhattacharya AB., et al. “Regeneration of thermally polymerized frying oils with adsorbents”. Food Chemistry 110.3(2008): 562-570.
- Vijayan J., et al. “Optional properties of corn oil during frying”. International Journal of Food Science & Technology 31 (1996): 353-358.
- CerianiR., et al. “Densities, and viscosities of vegetable oils of nutritional value”. Journal of Chemical & Engineering Data 53.8 (2008): 1846-1853.
- Mousavi K. et al. “Effects of storage conditions and PET packaging on quality of edible oils in Iran”. Advances in Environmental Biology 6.2 (2012): 694-701.
- Farhoosh R., et al. “Investigation on frying oils quality in terms of color index, refractive index and viscosity during frying process”. Journal of Food Science and Technology 5.1(2008): 13-19.
- Li H., et al. “Chemistry constituents and biological activities of saponin from the seed of Camellia Olefera”. Scientific Research and Essays 5.25 (2010): 4088-4092.
- Jenfeng P., et al. “Changes in physicochemical properties of Myotibrillar protein from Silver Carp (Hypophthalmichthys mollitrix) during heat treatment”. Journal of Food Biochemistry 35.3 (2011): 939-925.
- Jacobson G.A. “Quality control in deep-fat frying operations”. Food Technology 45 (1991): 72-74.
- Igwe OI. “The effect of temperature on the viscosity of vegetable oils in solution”. Industrial Crops and Products 19.2 (2004): 185-190.
- Santos JC. et al. “Thermoanalytical kinetic and rheological parameters of commercial edible vegetable oils”. Journal of Thermal Analysis and Calorimetry 75.2 (2004): 419-479.
- Saguy S and Dana D. “Integrated approach to deep fat frying engineering, nutrition, health and consumer aspects”. Journal of Food Engineering 56.2-3 (2003): 143-152.
- A.O.A.C. “Association of Official Agriculture Chemists, Official Methods of Analysis of 18th Edition.” (2005): D.C. USA
- Wu PF and Nawar WW. “A technique for monitoring his quality of used frying oils”. Journal of the American Oil Chemists Society 63.10 (1986): 1363-1367.
- Waltking AE and Wessels H. “Chromatographic separation of polar and non-polar components of frying oils”. Journal of the Association of Official Analytical Chemistry 64 (1981): 1329-1330.
- Melton S L., et al. “Review of stability measurements for frying oils and fried food flavor”. Journal of the American Oil Chemists Society 71.12 (1994): 1301-1308.
- Blumenthal MM. “A new look at the chemistry and physics of deep-fat frying”. Food Technology 45.2 (1991): 68-94.
- Warner K., et al. “Effect of frying oil composition on potato chip stability”. Journal of the American Oil Chemists' Society 71.10 (1994): 1117-1121.
- Clark W and Serbia GW. “Safety aspects of frying fats and oils”. Food Technology 45 (1991): 84-89.
Citation:
Shaker M Arafat., et al. “Relationship between viscosity and some chemical characteristics of frying oils”. Nutrition and Food
Toxicology 1.2 (2017): 44-53.
Copyright: © 2017 Shaker M Arafat., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.