Review Article
Volume 1 Issue 1 - 2016
Thromboaspiration during Primary PCI. When, Where and How?
1Department of Cardiology, Elpis General Hospital, Athens, Greece
2Cardiology Division, Ospedale Faenza (RA), Italy
2Cardiology Division, Ospedale Faenza (RA), Italy
*Corresponding Author: Athanassios Antonopoulos MD, FESC, Cardiology Division, Ospedale Faenza (RA), Italy.
Received: August 31, 2016; Published: November 18, 2016
Introduction
It is well established that the therapeutic gold standard for patients with ST-elevation infarction (STEMI) is mechanical revascularization of the responsible coronary artery: a technique that has been improved with stent implantation. Over the last decade, the use of primary PCI has spread and as a result the mortality rate for this group of patients has reduced significantly [1]. Despite this fact, complete reperfusion, epicardial and myocardial, is not achieved in 1/3 of these patients [2,3]. Since it has a predictive value for possible future cardiovascular events [4], the fundamental goal of primary PCI is tissue reperfusion of the myocardium. Many pathophysiological mechanisms have been described as being responsible for unsuccessful myocardial reperfusion: peripheral embolism by thrombotic micromolecules (debris); platelet activation; and inflammatory processes during primary PCI that lead to microvascular occlusion, which may contribute to the activation of bio-pathological procedures that lead to the phenomenon of non-reflow [5,7]. The use of thromboaspiration for the prevention of peripheral embolism, which, according to literature, is the inevitable result of the thrombus breakdown, is based on the judgment of the physician. However, in the presence of a great thrombotic burden, it could be used for the improvement of epicardial and, most of all, myocardial reperfusion. Based on the above data, there are many systems for coronary thromboaspiration, although they can generally be said to be of two types: manual and mechanical (rheolytic) thromboaspiration catheters. The first kind (manual) is based on a simple dual lumen catheter that is made up of a guide wire lumen and a thrombus extraction lumen. In practice, the guide wire is placed distally to the lesion and thrombus, and the aspiration catheter is placed just proximally to the thrombotic portion of the coronary artery. Negative pressure is applied via a syringe as the catheter is advanced and withdrawn over the thrombotic area. Once the vacuum syringe is full of blood, the catheter is removed as follows: the aspiration catheter is withdrawn over the wire, and the guide wire is then back bled to minimise the possibility of any residual thrombus remaining in the catheter. The aspirate is injected into a filter to examine the success of the thrombectomy. The second kind (Angiojet) employs the venturi effect via three high pressure saline jets directed from distal to proximal within the shaft of the catheter. A powerful suction effect is generated as each jet passes a narrow aperture that draws the thrombus into the catheter shaft where it is disrupted by the water jets and extracted. The system consists of a drive unit that generates the jet pressure, the Spiroflex coronary catheter, and a pump that connects the drive unit to the catheter.
In order to evaluate the efficacy of primary PCI, many multi-centre studies have been designed, with variable end-points using:
- Angiographical criteria, with the use of TIMI and BLUSH scores.
- ECG criteria using the time for the ST elevation and restoration.
- Some functional tests aimed at evaluating the restriction of microcirculatory damage at the site of infraction and left ventricle remodelling with imaging techniques and
- Clinical criteria that evaluate decreases in mortality. However, it is remarkable how the studies published on the issue of manual thromboaspiration during primary PCI have, in many cases, conflicting results.
The main purpose of this article is to describe a 'correct therapeutic method' on the basis of the published evidence worldwide and our personal experience on how, where, and when manual thromboaspiration should be used.
Evidence on manual thromboaspiration
According to the ESC 2012 and the ACC/AHA 2013 guidelines, routine thromboaspiration with a manual catheter during primary PCI is considered reasonable (recommendation IIa, LoE B) in order to prevent peripheral embolism [8,9]. The efficacy and safety of manual thromboaspiration during primary PCI in order to act on the affected artery has been demonstrated by many randomised studies. Moreover, on a global scale the majority of randomised studies, simple registries, and meta-analysis, all concur that thrombectomy improves the parameters of tissue reperfusion, with a more rapid and effective restoration of ST elevation on the ECG. Furthermore, the TAPAS study as well as EXPIRA trial, and some meta-analysis (although the latter did not have any statistical value on the prediction of major cardiac events), proved that in patients with STEMI who underwent primary PCI, thrombectomy improves the long-term clinical outcome by decreasing cardiac death and major cardiac events [10,11]. Moreover, in the EXPIRA trial, the group that underwent thrombectomy [12] were assessed with an MRI using paramagnetic substances.
According to the ESC 2012 and the ACC/AHA 2013 guidelines, routine thromboaspiration with a manual catheter during primary PCI is considered reasonable (recommendation IIa, LoE B) in order to prevent peripheral embolism [8,9]. The efficacy and safety of manual thromboaspiration during primary PCI in order to act on the affected artery has been demonstrated by many randomised studies. Moreover, on a global scale the majority of randomised studies, simple registries, and meta-analysis, all concur that thrombectomy improves the parameters of tissue reperfusion, with a more rapid and effective restoration of ST elevation on the ECG. Furthermore, the TAPAS study as well as EXPIRA trial, and some meta-analysis (although the latter did not have any statistical value on the prediction of major cardiac events), proved that in patients with STEMI who underwent primary PCI, thrombectomy improves the long-term clinical outcome by decreasing cardiac death and major cardiac events [10,11]. Moreover, in the EXPIRA trial, the group that underwent thrombectomy [12] were assessed with an MRI using paramagnetic substances.
After quantification, a decrease in microvascular damage in the infracted area was shown. The above results, were confirmed in the INFUSE-AMI trial, which was carried out on a larger sample of patients [13], with the best results being observed only in the group who underwent thrombectomy and endovascular administration of Abciximab. On the other hand, different results were recently published by a randomised Scandinavian trial (TASTE), which used a clinical registry of 7,244 patients with classical primary PCI, or primary PCI and manual thrombectomy as a basis. As a primary endpoint, the above study had an ambitious reduction in all causes of mortality in 30 days and as a result, there was no improvement with the use of a manual thrombectomy [14]. The aim of this study was over ambitious because expecting a reduction of all causes of mortality in 30 days is too much and therefore it might have been more appropriate to use a primary endpoint with a longer follow-up observation (of at least one year), as seen in the TAPAS study. Furthermore, it must be underlined that the design of a study that is based on a secondary randomised registry has a greater risk of error in the estimation of the final results. This is due to unavoidable mistakes being made in the beginning (bias), and also having no mention of the angiographic data after the thromboaspiration, such as TIMI or BLUSH scores, or the restoration time of ST segment elevation. Moreover, the frequency of high thrombotic burden (thrombus score > 3) was very low (around 25%) in the total population studied.
Besides, during the analysis of the inclusion criteria in the studies TAPAS, INFUSE-AMI, and EXPIRA, it was clear that all the patients showed an increase in the endovascular thrombotic burden. In EXPIRA, this was most evident (thrombus score > 3). As a result, these studies suggest that an embolectomy must carried out on patients with a high risk of peripheral embolism, i.e. in those for whom a thrombotic mass is evident. Consequently, according to universal references and guidelines, manual aspiration during primary PCI has to be performed where there is a large thrombotic endovascular burden (thrombus score > 3) and an increased possibility of peripheral embolism. In addition, another meta-analysis, which includes TASTE results and all randomised controlled trials on aspiration thrombectomy to date, demonstrates a significant reduction in adverse clinical outcomes that include stent thrombosis compared with only conventional PCI [15].
The other issue that is very important to discuss concerns the method and type of thromboaspiration that is used. So, based on the two great meta-analyses, that of Burzotta (with the participation of 2686 patients); and that of Kumbhani (with the participation of 5534 patients) [16,17], we can say that manual thromboaspiration was used more than mechanical thrombo-aspiration. According to the RETAMI trial [18], new devices for manual thromboaspiration are better than first generation manual devices. The use of mechanical- rheolytic thrombectomy must be limited to only those cases where the thrombotic endovascular mass is too large and the use of a manual device is inadequate. Among the few studies on mechanical-rheolytic thromboaspiration during primary PCI, the most well-structured is the JET-STENT Trial [19]. This trial, in which 501 patients participated, showed that the recovery of ST segment elevation on the ECG was more frequently observed in the randomised branch of the mechanical thrombectomy than the control group, and it was accompanied by a clear decrease of MACE at 1, 6, and 12 months. Additionally, it is necessary to mention that in the aforementioned study the primary endpoints that were set by the investigators (i.e. improved myocardial reperfusion, increased myocardial salvage, decreased infarct size, and better clinical outcome) were not achieved.
The need to decrease peripheral micro-embolism and the non-reflow phenomenon during primary PCI, both with or without mechanical thrombectomy, led to research into ways of finding better and more effective restrictions of this adverse event. As a result, the use of the endovascular administration of IIb/IIIa glycoprotein inhibitors seems to be promising in these kinds of situations [2,20,22].
Among IIb/IIIa inhibitors used in this way is, one of the most popular is Abciximab. Despite the fact that AHA/ACC guidelines recommend intracoronary Abciximab administration in patients with STEMI undergoing primary coronary angioplasty for class IIb inhibitors, randomised trials have shown mixed results for a variety of surrogate and combined clinical end points. [10,23,24]. Furthermore, in a recent study, pooled (individual patient-level) analysis of randomised trials comparing intracoronary and intravenous Abciximab bolus use in STEMI patients undergoing primary angioplasty, revealed that, comparatively, intracoronary Abciximab does not provide any additional benefit over the standard intravenous one [25].
On the other hand, a small molecule IIb/IIIa inhibitor (tirofiban) used endovascularly, showed better results than classic drugs, such as verapamil, adenosine, or sodium nitroprusside, for the prevention of the non-reflow phenomenon [26]. Furthermore, the SUIT-AMI trial based on 203 patients with STEMI compared the elective endovascular infusion of tirofiban after thrombectomy (10 μg/kg of body weight, followed by a 12-hour infusion of 0.1 μg/kg/min) through the aspiration catheter with the usual infusion through the guide catheter. The results of the study did not show any statistically significant difference in the primary end-points considered (CTFC, corrected thrombolysis count frame και MACE) in 30 days and 6 months and the major and minor bleeding incidence in the hospital [27].
Tirofiban’s mechanism of action
Tirofiban is a small molecule with an elimination half-life of 2 to 3 hours and its activity as an antiplatelet is of a short duration compared to Abciximab, which is used in many studies as an adjuvant treatment of a manual thromboaspiration. Moreover, from an immunological point of view, the use of Abciximab is associated with a highly increased risk of severe thrombocytopenia, which makes it necessary to closely monitor the platelet count shortly after administration of Abciximab. Tirofiban’s mechanism of action is multifactorial. It is clear that its endovascular bolus infusion, in contrast with the intravenous administration, increases the levels of the medicine locally.
Tirofiban is a small molecule with an elimination half-life of 2 to 3 hours and its activity as an antiplatelet is of a short duration compared to Abciximab, which is used in many studies as an adjuvant treatment of a manual thromboaspiration. Moreover, from an immunological point of view, the use of Abciximab is associated with a highly increased risk of severe thrombocytopenia, which makes it necessary to closely monitor the platelet count shortly after administration of Abciximab. Tirofiban’s mechanism of action is multifactorial. It is clear that its endovascular bolus infusion, in contrast with the intravenous administration, increases the levels of the medicine locally.
This, on one hand, facilitates tirofiban’s bonding with more IIb/IIIa platelet receptors and, on the other hand, more effectively inhibits the de-novo thrombin induced platelet aggregation. Consequently, the direct and effective separation of the platelets occurs in the epicardial artery, where they are responsible for the infarct, and in the capillaries. As a result of this, a remarkable improvement in the microcirculation, a decrease of the non-reflow phenomenon, and a decrease of the infarct size are demonstrated [28,29]. Moreover, tirofiban shows a much higher affinity with GPIIb/IIIa receptors than other GPIIb/IIIa inhibitors [30], and it also has the ability to react with both the latent and active states of GPIIb/IIIa receptors. Additionally, tirofiban is characterised by a rapid on-rate and off-rate and it dissociates from the receptor at a half-life of 11 seconds [31].
Another advantage of tirofiban use over other GPIIb/IIIa inhibitors, is considered to be the induction of effects based on a dose-concentration dependent manner, which is important for both local (intracoronary) and systemic environments [30,32]. Furthermore, the endovascular administration of tirofiban increases the concentration of nitric oxide, which is an endothelium protector [33] that inhibits the peripheral microembolism at multiple levels. Consequently, the endovascular bolus infusion of tirofiban during primary PCI prevents pro-thrombotic and pro-inflammatory factors such as P-selectin, factor von Willebrand, CD40 ligand, and factor A amyloid in coronary sinus [34] from being activated. As a consequence of this,a reduction in platelet aggregation, both regionally and at the inflammatory sites, was observed in atherothrombotic lesions [35,36].
Our Practice
In agreement with the aforementioned studies, we are trying to apply the combined technique of thromboaspiration and endovascular administration with a reversible IIb/IIIa inhibitor such as tirofiban. During this procedure two items were taken into consideration:
In agreement with the aforementioned studies, we are trying to apply the combined technique of thromboaspiration and endovascular administration with a reversible IIb/IIIa inhibitor such as tirofiban. During this procedure two items were taken into consideration:
- An estimate of the angiographical TIMI score. If this is 0 to 1 the classic angioplasty is preferred, if it is > 1 the manual aspiration is preferred. In the case that the first effort is not successful, the catheter is rewashed and the procedure repeated.
- An estimate of the thrombus score [37]: If it is < 3 the classic angioplasty is preferred, if it is > 3 manual thromboaspiration is preferred. Lastly, if the thrombus is 3-4 times larger than the diameter of the vessel, in our opinion, mechanical-rheolytic thrombectomy represents the first choice.
In all of the situations mentioned above, tirofiban is administered either through a catheter guide or through a manual catheter for thromboaspiration (when this is already in the vessel). Up until now, the second administration choice seems to have better results due to catheter size. It has in fact a small diameter and thus the drug is diffused with a higher velocity from the intraluminal tip of the catheter and it pervades a greater amount of thrombus. Afterwards the stent is being placed.
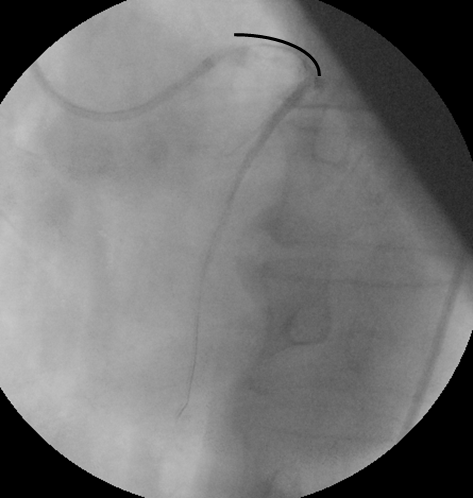
Figure 1: Patient with cardiac shock caused by anterior myocardial infarction due to acute occlusion of left main artery. Manual thromboaspiration was performed and tirofiban was infused in the vessel through the same catheter. Flow TIMI I. The great amount of thrombus from the left main coronary artery to the outgrowth of the septal branch of the left anterior descending artery is obvious.
The length of the endovascular thrombus is underlined by the black curve.
The length of the endovascular thrombus is underlined by the black curve.
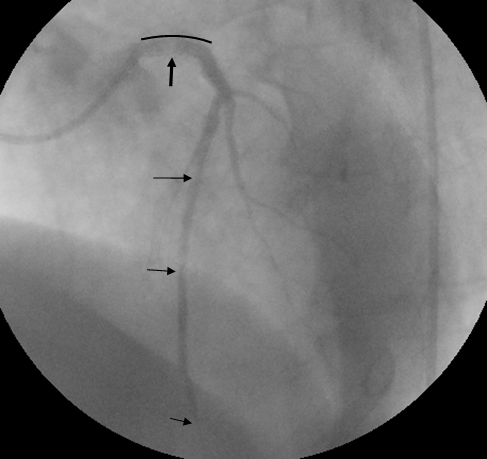
Figure 2: A second attempt of manual thromboaspiration was made and tirofiban was infused in the coronary artery through the same catheter, followed by a direct stent implantation in the left main artery. Afterwards an improvement of the flow TIMI III and sufficient outline of the diagonals and the first septal was noted. Based on protocol tirofiban was administered intravenously for the next 12 hours.
The black curve outlines the length of the stent implanted while thrombus remnants are seen (pointed with arrows) at the beginning of the left main and distally that do not disturb the satisfactory flow of the vessel.
The black curve outlines the length of the stent implanted while thrombus remnants are seen (pointed with arrows) at the beginning of the left main and distally that do not disturb the satisfactory flow of the vessel.
Conclusion
To sum up, manual thromboaspiration in primary coronary angioplasty is easier and more efficient. However, in order to achieve optimal results, some rules must be followed. Indeed, strong evidence suggests that, during primary angioplasty in patients with STEMI, the local delivery of IIb/IIIa receptor antagonists directly into the coronary circulation may improve microvascular function and clinical outcomes. The reason for this therapeutic choice is the possibility of reaching a high concentration of the aforementioned drugs at the thrombus site without significantly increasing the risk of bleeding. Thus, a high concentration of antithrombotic agents at the site of a thrombotic lesion may facilitate the diffusion of the drug into acute thrombus and consequently promote clot dissolution in the epicardial and micro-vessels. As described above, on the one hand intracoronary administration of Abciximab (used in most of the trials) did not show a clear advantage compared to the intravenous administration. On the other hand, there is clear evidence that endovascular coronary administration of tirofiban (a small molecule that is a reversible IIb/IIIa antagonist), either through the catheter of thromboaspiration or the guide catheter, can improve the results of primary PCI in patients with an acute myocardial infarction. Lastly, the mechanical-rheolytic aspiration being a more complex procedure, its use may be preferred only in the context of a huge thrombotic burden.
References
- Grines CL., et al. “Coronary angioplasty with or without stent implantation for acute myocardial infarction. Stent Primary Angioplasty in Myocardial Infarction Study Group”. The New England Journal of Medicine 341.26 (1999): 1949-56.
- Montalescot G., et al. “Platelet glycoprotein IIb/IIIa inhibition with coronary stenting for acute myocardial infarction”. The New England Journal of Medicine 344.25 (2001): 1895-903.
- Stone GW., et al. “Comparison of angioplasty with stenting, with or without abciximab, in acute myocardial infarction.” The New England Journal of Medicine 346.13 (2002): 957-66.
- van`t Hof AW., et al. “Clinical value of 12-lead electrocardiogram after successful reperfusion therapy for acute myocardial infarction. Zwolle Myocardial infarction Study Group.” The Lancet 350.9078 (1997): 615-9.
- White CJ., et al. “Coronary thrombi increase PTCA risk. Angioscopy as a clinical tool”. Circulation 93.2 (1996): 253-8.
- Kotani J., et al. “Plaque gruel of atheromatous coronary lesion may contribute to the no-reflow phenomenon in patients with acute coronary syndrome”. Circulation 106.13 (2002): 1672-7.
- Henriques JP., et al. “Incidence and clinical significance of distal embolization during primary angioplasty for acute myocardial infarction”. European Heart Journal 23.14 (2002): 1112-7.
- O` Gara PT., et al. “2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines”. Circulation 127.4 (2013): 529-55.
- Steg PG., et al. “ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation”. European Heart Journal 33.20 (2012): 2569-619.
- Sardella G., et al. “Impact of thrombectomy with EXPort Catheter in Infarct-Related Artery during Primary Percutaneous Coronary Intervention (EXPIRA Trial) on cardiac death”. American Journal of Cardiology 106.5 (2010): 624-9.
- Vlaar PJ., et al. “Cardiac death and reinfarction after 1 year in the Thrombus Aspiration during Percutaneous coronary intervention in Acute myocardial infarction Study (TAPAS): a 1-year follow-up study”. The Lancet 371.9628 (2008): 1915-20.
- Sardella G., et al. “Thrombus aspiration during primary percutaneous coronary intervention improves myocardial reperfusion and reduces infarct size: the EXPIRA (thrombectomy with export catheter in infarct-related artery during primary percutaneous coronary intervention) prospective, randomized trial”. Journal of the American College of Cardiology 53.4 (2009): 309-15.
- Stone GW., et al. “Intracoronary abciximab and aspiration thrombectomy in patients with large anterior myocardial infarction: the INFUSE-AMI randomized trial”. JAMA 307.17 (2012): 1817-26.
- Frobert o., et al. “Thrombus aspiration during ST-segment elevation myocardial infarction”. The New England Journal of Medicine 369.17 (2013): 1587-1597.
- Kumbhani DJ., et al. “Aspiration thrombectomy in patients undergoing primary angioplasty: totality of data to 2013”. Catheterization and Cardiovascular Interventions 84.6 (2014): 973-977.
- Burzotta F., et al. “Clinical impact of thrombectomy in acute STelevation myocardial infarction: an individual patient-data pooled analysis of 11 trials”. European Heart Journal 30 (2009): 2193-203.
- Kumbhani DJ., et al. “Role of aspiration and mechanical thrombectomy in patients with acute myocardial infarction undergoing primary angioplasty: an updated meta-analysis of randomized trials”. Journal of the American College of Cardiology 62.16 (2013): 1409-18.
- Sardella G., et al. “The effect of thrombectomy on myocardial blush in primary angioplasty: the Randomized Evaluation of Thrombus Aspiration by two thrombectomy devices in acute Myocardial Infarction (RETAMI) trial”. Catheterization and Cardiovascular Intervention 71.1 (2008): 84-91.
- Migliorini A., et al. “Comparison of AngioJet rheolytic thrombectomy before direct infarct artery stenting with direct stenting alone in patients with acute myocardial infarction. The JETSTENT trial”. Journal of the American College of Cardiology 56.16 (2010): 1298-306.
- Tao Qin., et al. “Meta-analysis of randomized controlled trials on the efficacy and safety of intracoronary administration of tirofiban for no-reflow phenomenon”. BMC Cardiovascular Disorders 13 (2013): 68.
- Romagnoli E., et al. “Rationale for intracoronary administration of Abciximab”. Journal of Thrombosis and Thrombolysis 23.1 (2007): 57-63.
- De Luca G., et al. “Benefits from intracoronary as compared to intravenous Abciximab administration for STEMI patients undergoing primary angioplasty: a meta-analysis of 8 randomized trials”. Atherosclerosis 222.2 (2012): 426-33.
- Shimada YJ., et al. “Meta-analysis of prospective randomized controlled trials comparing intracoronary versus intravenous abciximab in patients with ST-elevation myocardial infarction undergoing primary percutaneous coronary intervention”. American Journal of Cardiology 109.5 (2012): 624-8.
- Hansen PR., et al. “Improved clinical outcomes with intracoronary compared to intravenous abciximab in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a systematic review and meta-analysis”. Journal of Invasive Cardiology 22.6 (2010): 278-282.
- Hansen PR., et al. “Improved clinical outcomes with intracoronary compared to intravenous abciximab in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a systematic review and meta-analysis”. Journal of Invasive Cardiology 22.6 (2010): 278-82.
- Piccolo R., et al. “Intracoronary versus intravenous bolus abciximab administration in patients undergoing primary percutaneous coronary intervention with acute ST-elevation myocardial infarction: a pooled analysis of individual patient data from five randomised controlled trials”. EuroIntervention 9.9 (2014): 1110-1120.
- Natarajan D. “Combined Intracoronary Glycoprotein Inhibitors And Manual Thrombus Extraction In Patients With Acute ST-Segment Elevation Myocardial Infarction - Does Incorporation Of Both Have A Legitimate Role?” Interventional cardiology 6.2 (2011): 182-5.
- Yi Chen., et al. “Impact of selective infarct-related artery infusion of tirofiban on myocardial reperfusion and bleeding complications in patients with acute myocardial infarction: the SUIT-AMI trial”. Journal of Invasive Cardiology 25.8 (2013): 376-382.
- Deibele AJ., et al. “Intracoronary eptifibatide bolus administration during percutaneous coronary revascularization for acute coronary syndromes with evaluation of platelet glycoprotein IIb/IIIa receptor occupancy and platelet function: the Intracoronary Eptifibatide (ICE) Trial”. Circulation 121.6 (2010): 784-791.
- Gurbel PA and Tantry US. “Delivery of glycoprotein IIb/IIIa inhibitor therapy for percutaneous coronary intervention. Why not take the intracoronary highway?” Circulation 121 (2010): 739-741.
- Schrör K and Weber AA. “Comparative pharmacology of GP IIb/IIIa antagonists”. Journal of Thrombosis and Thrombolysis 15.2 (2003): 71-80.
- Shanmugam G. “Tirofiban and emergency coronary surgery”. European Journal Cardio-Thoracic Surgery 28.4 (2005): 546-550.
- Hashemzadeh M., et al. “Chemical structures and mode of action of intravenous glycoprotein IIb/IIIa receptor blockers: A review”. Experimental & Clinical Cardiology 13.4 (2008): 192-197
- Yang YJ., et al. “Different effects of tirofiban and aspirin plus clopidogrel on myocardial no‐reflow in a mini‐swine model of acute myocardial infarction and reperfusion”. Heart 92.8 (2006): 1131-1137.
- Zhu TQ., et al. “Beneficial effects of intracoronary tirofiban bolus administration following upstream intravenous treatment in patients with ST-elevation myocardial infarction undergoing primary percutaneous coronary intervention: the ICT-AMI study”. International Journal of Cardiology 165.3 (2013): 437-43.
- Huber K., et al. “Use of glycoprotein IIb/IIIa inhibitors in primary percutaneous coronary intervention: insights from the APEX-AMI trial”. European Heart Journal 31.14 (2010): 1708-1716.
- Mater W., et al. “Inflammatory markers at the site of ruptured plaque in acute myocardial infarction: locally increased interleukin-6 and serum amyloid A but decreased C-reactive protein”. Circulation 111.11 (2005): 1355-1361.
- Topaz O., et al. “Thrombus Grading for Coronary Interventions. The Role of Contemporary Classifications”. Interventional Cardiology 3.6 (2011): 705-712.
Citation:
Athanassios Antonopoulos and Evangelia Sergi. “ Thromboaspiration during Primary PCI. When, Where and How?” Therapeutic Advances in Cardiology 1.1 (2016): 5-11.
Copyright: © 2016 Athanassios Antonopoulos and Evangelia Sergi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.