Research Article
Volume 2 Issue 1 - 2018
Low Integrated Area of Desaturation Index after Adaptive Servo-Ventilation Therapy is a Strong Indicator to Predict Fatal Cardiovascular Events in Heart Failure Patients
1Department of Cardiovascular Medicine, Gunma University Graduate School of Medicine, Maebashi, Japan
2Division of Cardiology, Isesaki Municipal Hospital, Isesaki, Japan
2Division of Cardiology, Isesaki Municipal Hospital, Isesaki, Japan
*Corresponding Author: Atsushi Umeyama, MD, Department of Cardiovascular Medicine, Gunma University Graduate School of Medicine, 3-39-15 Showamachi, Maebashi, Gunma, Japan.
Received: May 16, 2018; Published: May 26, 2018
Abstract
Background: Adaptive servo-ventilation (ASV) has been used to treat heart failure (HF) with sleep-disordered breathing. However, there are no polysomnographic data correlated with HF prognosis. Therefore, the present study aimed to establish whether integrated area of desaturation (IAD) index, immediately after ASV therapy, was effective in predicting the incidence of fatal cardiovascular events in patients with HF, over a five-year follow-up period.
Methods: Fifty-seven patients with HF were enrolled for ASV treatment. Patients were categorised into 4 groups based on IAD index before (pre-IAD index) and after (post-IAD index) ASV therapy: Group A, pre-IAD index ≥ 50% sec/min and post-IAD index < 15% sec/min (n = 5); Group B, pre-IAD index ≥ 50% sec/min and post-IAD index ≥ 15% sec/min (n = 22); Group C, pre-IAD index < 50% sec/min, post-IAD index < 15% sec/min (n = 15) and Group D, pre-IAD index < 50% sec/min, post-IAD index ≥ 15% sec/min (n = 15). We observed patients over a five-year follow-up period, and noted fatal cardiovascular events (death from progressive HF, cardio-embolic stroke or fatal cardiac arrhythmias).
Results: Incidence of fatal cardiovascular events significantly decreased in low post-IAD index groups compared with high post-IAD index groups (Group A and C versus Group B and D; P = 0.034).
Conclusions: Results of the present study suggest that low IAD index after ASV therapy may be a useful parameter to assess the efficacy of ASV therapy in HF patients.
Keywords: Adaptive servo-ventilation; Integrated area of desaturation index; Sleep-disordered breathing; Heart failure
Abbreviations: AHI: Apnea–Hypopnea Index; ASV: Adaptive Servo-Ventilation; CT90%: Cumulative Percentage Time at Oxygen Saturation below 90%; HF: Heart Failure; HFrEF: Heart Failure with Reduced Ejection Fraction; IAD: Integrated Area of Desaturation; LVEF: Left Ventricular Ejection Fraction; ODI: Oxygen Desaturation Index; PSG: Polysomnography; SDB: Sleep-Disordered Breathing
Introduction
Adaptive servo-ventilation (ASV) has been originally used to treat sleep-disordered breathing (SDB), including central sleep apnoea, Cheyne–Stokes respiration [1-3] and obstructive sleep apnoea [4]. Furthermore, ASV has been recently shown to be effective for heart failure (HF) treatment in patients with SDB [1, 5-13]. However, Cowie., et al. [14] reported that ASV therapy has adverse effects in patients with HF with reduced ejection fraction (HFrEF) and predominant central sleep apnoea (SERVE-HF study). Despite these conflicting results, several studies have reported that ASV was an effective device for HF treatment [1, 5-13]. Based on these results, we deemed it necessary to examine which HF patients can be effectively treated with ASV.
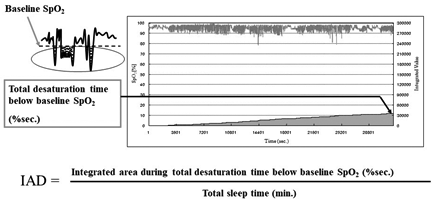
It is common to evaluate the severity and prognosis of SDB using apnoea-hypopnoea index (AHI), cumulative percentage time at oxygen saturation below 90% (CT90%), or 3% oxygen desaturation index (3%ODI). However, little is known about the relationship between these polysomnographic data and fatal cardiovascular events in patients with HF undergoing ASV therapy [10,11,15,16]. Therefore, the objective of the present study was to establish whether integrated area of desaturation (IAD) index was effective in predicting the incidence of a fatal cardiovascular event in patients with HF, over a five-year follow-up period. IAD index, devised by the Tokyo Medical University, is calculated by dividing the total area of desaturation by the total sleep time using polysomnography (PSG) [15]. Therefore, in comparison to other PSG data, the IAD index can more accurately measure SDB severity, as it takes into account the duration and degree of hypoxia.
Materials and Methods
Study design and ethical considerations
The present study was a two-centre, prospective cohort study. We enrolled 57 patients with HF, classified into New York Heart Association classes II–IV and used ASV for HF treatment. All the patients were treated at Gunma University Hospital and Isesaki Municipal Hospital between February 2009 and April 2011. The research protocol was approved by the Institutional Review Board for Human Research of both the hospitals, and written informed consent was obtained from each patient before the study.
The present study was a two-centre, prospective cohort study. We enrolled 57 patients with HF, classified into New York Heart Association classes II–IV and used ASV for HF treatment. All the patients were treated at Gunma University Hospital and Isesaki Municipal Hospital between February 2009 and April 2011. The research protocol was approved by the Institutional Review Board for Human Research of both the hospitals, and written informed consent was obtained from each patient before the study.
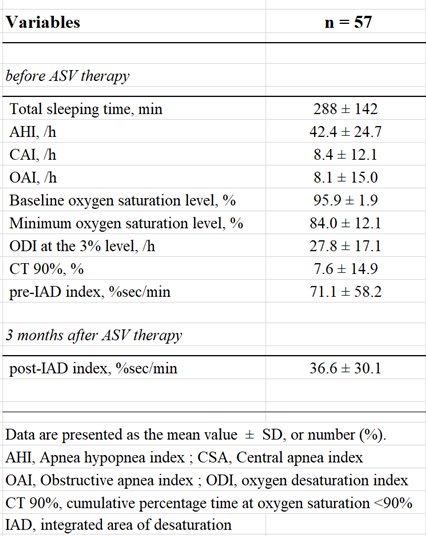
After medical therapy was optimised, all the patients were examined overnight by PSG, and IAD index was calculated to assess the severity of SDB before ASV therapy. Successive ASV treatment was administered for three months. Immediately thereafter, patients were re-examined overnight by PSG, and IAD index was calculated. Patients were classified into 4 groups based on their IAD index before (pre-IAD index) and after (post-IAD index) ASV therapy. Patients with pre-IAD index ≥ 50% sec/min and post-IAD index <15% sec/min were designated as Group A (n = 5), those with pre-IAD index ≥ 50% sec/min and post-IAD index ≥ 15% sec/min were designated as Group B (n = 22), those with pre-IAD index < 50% sec/min and post-IAD index < 15% sec/min were designated as Group C (n = 15), and those with pre-IAD index < 50% sec/min and post-IAD index ≥ 15% sec/min were designated as Group D (n = 15) (Figure 1). We observed the patients over a 5 year follow-up period and noted any fatal cardiovascular events (e.g., death from progressive HF, cardio-embolic stroke or fatal cardiac arrhythmias) to establish long-term prognosis. Non-cardiovascular related deaths were excluded from the present study.
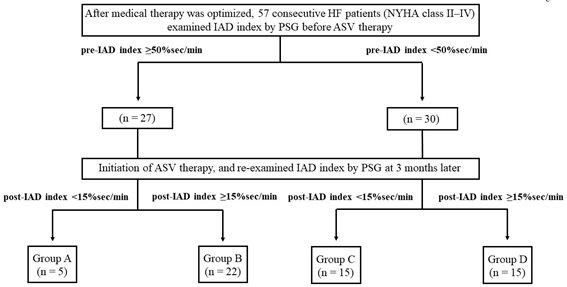
Figure 1: The present study protocol. HF, heart failure; NYHA, New York Heart Association
IAD, integrated area of desaturation; ASV, adaptive servo-ventilation.
Sleep evaluation and treatment devices
To assess the severity of SDB, all the patients underwent overnight PSG using digital polygraph (E-Series Plus; Compumedics, Abbotsville, Australia). We measured chest and abdominal movement using two bands (Inductive Respiratory Bands). Airflow was measured using a thermistor and arterial oxygen saturation was measured using pulse oximetry with a finger probe (Nonin 8000J Adult Flex Sensor). Electroencephalography, electrooculography and chin electromyography were also conducted. We scored sleep stage and arousal according to standard criteria [17,18]. We defined apnoea as the absence of airflow for > 10 sec and defined hypopnoea as > 50% decrease in airflow accompanied by >3% desaturation. We investigated PSG data, AHI, central apnoea index, obstructive apnoea index, 3% ODI, CT90%, minimum oxygen saturation level and baseline oxygen saturation level. IAD index was calculated by dividing the total area of desaturation by total sleep time, measured in minutes. Baseline oxygen saturation level was determined using the average of 10 min in the sleeping position, before the patients fell asleep (Figure 2).
To assess the severity of SDB, all the patients underwent overnight PSG using digital polygraph (E-Series Plus; Compumedics, Abbotsville, Australia). We measured chest and abdominal movement using two bands (Inductive Respiratory Bands). Airflow was measured using a thermistor and arterial oxygen saturation was measured using pulse oximetry with a finger probe (Nonin 8000J Adult Flex Sensor). Electroencephalography, electrooculography and chin electromyography were also conducted. We scored sleep stage and arousal according to standard criteria [17,18]. We defined apnoea as the absence of airflow for > 10 sec and defined hypopnoea as > 50% decrease in airflow accompanied by >3% desaturation. We investigated PSG data, AHI, central apnoea index, obstructive apnoea index, 3% ODI, CT90%, minimum oxygen saturation level and baseline oxygen saturation level. IAD index was calculated by dividing the total area of desaturation by total sleep time, measured in minutes. Baseline oxygen saturation level was determined using the average of 10 min in the sleeping position, before the patients fell asleep (Figure 2).
For HF treatment, we used the ASV device (AutoSet-CS; ResMed, Sydney, Australia) with a full facemask (ResMed). Expiratory positive airway pressure was set to 4 cm H2O, and optimum inspiratory support pressure was set to a minimum of 3-8 cm H2O. The backup respiratory rate for SDB was 15 breaths/min. We performed ASV titration for reduced SBD when required.
Data collection
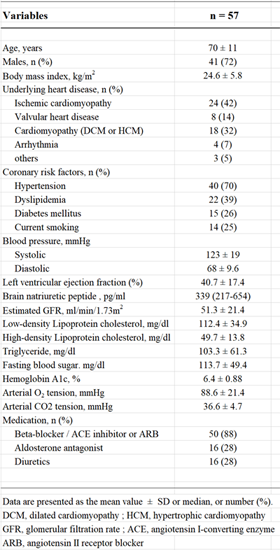
We assessed physical examination, medical history, oral agents, venous and arterial blood samples, and transthoracic echocardiography data as baseline data, prior to ASV therapy. Venous blood samples were examined after overnight fasting. Arterial blood samples were examined with patients in the supine position ensuring sufficient room air at rest. Hypertension was defined as blood pressure ≥ 140/90 mmHg or the use of antihypertensive drugs. Dyslipidemia was defined as low-density lipoprotein cholesterol ≥ 140 mg/dL, triglycerides ≥ 150 mg/dL, high-density lipoprotein cholesterol < 40 mg/dL or the use of cholesterol-lowering drugs. Diabetes mellitus was defined as fasting blood glucose ≥ 126 mg/dL, HbA1c ≥ 6.5% or the use of insulin or oral antihyperglycemic drugs. Left ventricular ejection fraction (LVEF) was examined by transthoracic echocardiography using Simpson’s method.
We assessed physical examination, medical history, oral agents, venous and arterial blood samples, and transthoracic echocardiography data as baseline data, prior to ASV therapy. Venous blood samples were examined after overnight fasting. Arterial blood samples were examined with patients in the supine position ensuring sufficient room air at rest. Hypertension was defined as blood pressure ≥ 140/90 mmHg or the use of antihypertensive drugs. Dyslipidemia was defined as low-density lipoprotein cholesterol ≥ 140 mg/dL, triglycerides ≥ 150 mg/dL, high-density lipoprotein cholesterol < 40 mg/dL or the use of cholesterol-lowering drugs. Diabetes mellitus was defined as fasting blood glucose ≥ 126 mg/dL, HbA1c ≥ 6.5% or the use of insulin or oral antihyperglycemic drugs. Left ventricular ejection fraction (LVEF) was examined by transthoracic echocardiography using Simpson’s method.
Statistical analysis
Continuous data were expressed as mean ± standard deviation (SD) or median (25%–75% range). Categorical data were expressed as a percentage. To analyse the incidence of fatal cardiovascular events between 4 groups, we used Kaplan–Meier analysis and log-rank test. In all analyses, P < 0.05 was considered to be statistically significant.
Continuous data were expressed as mean ± standard deviation (SD) or median (25%–75% range). Categorical data were expressed as a percentage. To analyse the incidence of fatal cardiovascular events between 4 groups, we used Kaplan–Meier analysis and log-rank test. In all analyses, P < 0.05 was considered to be statistically significant.
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan). EZR is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria) and modified version of R designed to add statistical functions frequently used in biostatistics.
Results
We enrolled 57 patients with HF New York Heart Association classes II–IV in the present study. Table 1 shows the clinical characteristics of the patients. The mean age was 70 ± 11 years, and 72% of the patients were male. The average body mass index was 24.6 ± 5.8 kg/m2, average LVEF was 40.7 ± 17.4% and median brain natriuretic peptide level was 339 (217–654) pg/ml. Table 2 shows PSG data before and after three months of ASV therapy. The average AHI was 42.4 ± 24.7 /h, 3%ODI was 27.8 ± 17.1 /h and CT90% was 7.6 ± 14.9%. The average pre-IAD index was 71.1 ± 58.2% sec/min, the mean post-IAD index was 36.6 ± 30.1% sec/min.
Figure 3 shows a scatter plot depicting the patients who incurred a fatal cardiovascular event. Data points uniformly distributed in the vertical axis represent the pre-IAD index, whereas those inclined to the right in the horizontal axis represent the post-IAD index. Groups with lower than average post-IAD index experienced fewer fatal cardiovascular events than those with high post-IAD index, regardless of the severity of pre-IAD index (Group A; 0.0%, Group B; 40.9%, Group C; 13.3%, Group D; 40.0%). Figure 4 shows the Kaplan–Meier analysis for the incidence of fatal cardiovascular events between the four groups. The incidence of fatal cardiovascular events was significantly different between the low post-IAD index groups (Group A and C) and the high post-IAD index groups (Group B and D) (Group A and C versus Group B and D; log-rank test: P = 0.034).
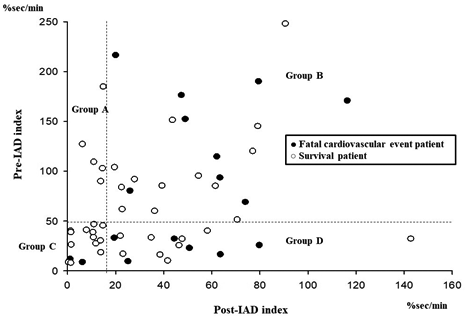
Figure 3: Scatter plot for the fatal cardiovascular events about all patients.
IAD, integrated area of desaturation.
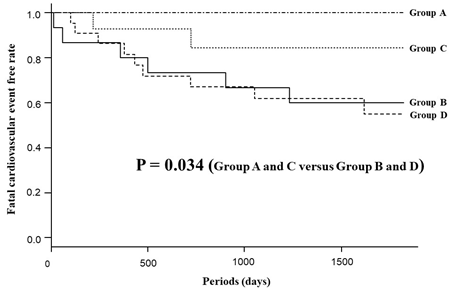
Figure 4: Kaplan-Meier analysis for the fatal cardiovascular event-free rate between the four groups.
Discussion
In the present study, 57 patients with HF were enrolled for ASV treatment. These patients were classified into 4 groups based on IAD index before and after ASV therapy. We observed patients over a 5 year follow-up period and noted any fatal cardiovascular events (e.g., death from progressive HF, cardio-embolic stroke or fatal cardiac arrhythmias). The results showed that low IAD index after ASV therapy was correlated with a decrease in fatal cardiovascular events in HF patients, regardless of severity of SDB as indicated by the IAD index before ASV therapy. ASV therapy could also be performed safely regardless of the severity of IAD index before ASV therapy.
A previous report compared IAD indexes between patients with cardiovascular events and non-cardiovascular events retrospectively, and focussed only on SDB patients with obstructive sleep apnoea [15]. Another report targeted only patients with acute or old myocardial infarction [19]. In contrast, the present study investigated the long-term prognosis of patients with various types and stages of HF after ASV therapy. Furthermore, the present study targeted patients with any SDB related conditions associated with HF. To our knowledge, the present study is the first to investigate the prognosis of HF after ASV therapy, according to IAD index prospectively.
It is well known that SBD increases cardiovascular and all-cause mortality [20-23]. However, conventional indexes of SDB, including AHI, 3%ODI, CT90%, are only measured as a frequency or ratio, and these indexes may not accurately assess the severity of SDB. Conversely, IAD index considers the duration and degree of hypoxia, thereby measuring the severity of SDB quite accurately. It is our opinion that this is the reason that the patients with high IAD index after ASV therapy experienced fatal cardiovascular events more often.
Until now, ASV has been commonly used for HF treatment, and studies have shown that ASV is an effective treatment for HF. However, recently, the SERVE-HF study reported that ASV therapy had adverse effects in patients with HFrEF. Based on this result, it is particularly important to consider how ASV therapy affects HFrEF patients. The present study showed good prognosis if the IAD index after ASV therapy was low, regardless of the severity of the IAD index before ASV therapy.
The present study has limitations; most importantly, it included only a small number of patients from only two centres. These limitations may have biased the results, and it is necessary to conduct a larger, multicenter study in the future.
Conclusion
In conclusion, the present study showed that low IAD index after ASV therapy in HF patients could reduce fatal cardiovascular events despite IAD index prior to ASV treatment being severe. Furthermore, our results suggest that low IAD index after ASV therapy may be a useful parameter to assess the long-term efficacy of ASV therapy in HF patients.
References
- H Teschler., et al. “Adaptive pressure support servo-ventilation: a novel treatment for Cheyne-Stokes respiration in heart failure”. American Journal of Respiratory and Critical Care Medicine 164 (2001): 614-619.
- JC Pepperell., et al. “A randomized controlled trial of adaptive ventilation for Cheyne-Stokes breathing in heart failure”. American Journal of Respiratory and Critical Care Medicine 168 (2003): 1109-1114.
- JS Allam., et al. “Morgenthaler, Efficacy of adaptive servo-ventilation in treatment of complex and central sleep apnea syndromes”. Chest 132 (2007): 1839-1846.
- AS Jordan., et al. “Adult obstructive sleep apnoea”. Lancet 383 (2014): 736-747.
- Oldenburg., et al. “Adaptive servoventilation improves cardiac function in patients with chronic heart failure and Cheyne-Stokes respiration”. European Journal of Heart Failure 10 (2008): 581-586.
- BK Sharma., et al. “Adaptive servoventilation for treatment of sleep-disordered breathing in heart failure: a systematic review and meta-analysis”. Chest 142 (2012): 1211-1221.
- PC Hastings., et al. “Adaptive servo-ventilation in heart failure patients with sleep apnea: a real world study”. International Journal of Cardiology 139 (2010): 17-24.
- S Momomura., et al. “Adaptive servo-ventilation therapy for patients with chronic heart failure in a confirmatory, multicenter, randomized, controlled study”. Circulation Journal 79 (2015): 981-990.
- T Kasai., et al. “Adaptive servo-ventilation in cardiac function and neurohormonal status in patients with heart failure and central sleep apnoea nonresponsive to continuous positive airway pressure”. JACC Heart Failure 1 (2013): 58-63.
- N Takama and M Kurabayashi. “Safety and efficacy of adaptive servo-ventilation in patients with severe systolic heart failure”. Journal of Cardiology 63 (2014): 302-307.
- N Takama and M Kurabayashi. “Effect of adaptive servo-ventilation on 1-year prognosis in heart failure patients”. Circulation Journal 76 (2012): 661–667.
- A Yoshihisa., et al. “Adaptive servo ventilation improves cardiac dysfunction and prognosis in chronic heart failure patients with Cheyne-Stokes respiration”. International Heart Journal 52 (2011): 218-223.
- T Imamura., et al. “Long-Term Adaptive Servo-Ventilator Treatment Prevents Cardiac Death and Improves Clinical Outcome”. International Heart Journal 57 (2016): 47–52.
- MR Cowie., et al. “Adaptive Servo-Ventilation for Central Sleep Apnea in Systolic Heart Failure”. The New England Journal of Medicine 373 (2015): 1095-1105.
- K Asano., et al. “New index for analysis of polysomnography, ‘integrated area of desaturation’, is associated with high cardiovascular risk in patients with mild to moderate obstructive sleep apnea”. Respiration 78 (2009): 278-284.
- B Gellen., et al. “Apnea-hypopnea and desaturations in heart failure with reduced ejection fraction: Are we aiming at the right target?” International Journal of Cardiology 203 (2016): 1022–1028.
- Rechtschaffen A and Kales A. “A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects”. Brain Information Service/Brain Research Institute, UCLA (1968):
- “EEG arousals: scoring rules and examples: a preliminary report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders Association”. Sleep 15 (1992): 174-184.
- N Takama. “Integrated area of desaturation index in patients with myocardial infarction”. International Journal of Cardiology 143 (2010): 425-428.
- NM Punjabi., et al. “Sleep-disordered breathing and mortality: a prospective cohort study”. PLoS Medicine 6 (2009): e1000132.
- N Takama and M Kurabayashi. “Influence of untreated sleep-disordered breathing on the long-term prognosis of patients with cardiovascular disease”. American Journal of Cardiology 103 (2009): 730-734.
- P Lavie., et al. “All-cause mortality in males with sleep apnoea syndrome: declining mortality rates with age”. European Respiratory Journal 25 (2005): 514–520.
- P Lavie., et al. “Mortality in sleep apnea patients: a multivariate analysis of risk factors”. Sleep (1995): 149-157.
Citation:
Atsushi Umeyama., et al. “Low Integrated Area of Desaturation Index after Adaptive Servo-Ventilation Therapy is a Strong
Indicator to Predict Fatal Cardiovascular Events in Heart Failure Patients”. Therapeutic Advances in Cardiology 2.1 (2018): 219-226.
Copyright: © 2018 Atsushi Umeyama., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.